Stoichiometric Relationships (The Mole)
Seven Molecular Elements (Diatomic):
- Hydrogen (H2)
- Nitrogen (N2)
- Oxygen (O2)
- Fluorine (F2)
- Chlorine (Cl2)
- Iodine (I2)
- Bromine (Br2)
Seven Complex Ions:

Introduction to Chemical Change
Types of Chemical Reactions:
Synthesis: 2 or more reactants combine and form one product (A + B → AB)
Decomposition: 1 reactant breaks down into 2 or more products (AB → A+B)
Single Displacement Reaction: More reactive element replaces less reactive element in a compound
(A + XY → AY + X)
Double Displacement Reaction: 2 compounds react to form 2 new compounds (usually involves aqueous solutions and 1 product is a precipitate)
(AB + XY → XB + AY)
The Mole Concept
Mole: 6.02 x 10^23
Avogadro’s Number (NA) or L = 6.02 x 10^23
Number of Fundamental Particles (N)
Number of Moles (mol) Present (n)
Formula: N = nNA
- To calculate # of molecules when mol is given, multiply mol by 6.02 x 10^23 (N = nNA)
- To calculate # of atoms when mol and molecules are given, multiply number of atoms by N of previous number
Molar Mass (g/mol)
Mass of atoms relative to carbon (12 amu)
Mole: Number of atoms in 12 grams of Carbon
Refer to Periodic Table
Example: Molar Mass of H2O
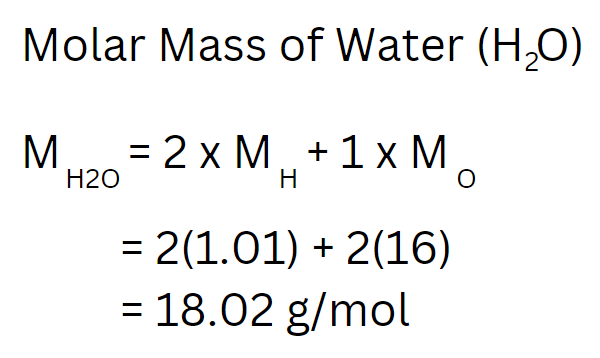 Relating Mass and Moles
Relating Mass and Moles
Calculate mass from the moles, and moles from the mass using MOLAR MASS
Problem Type #1
 Problem Type #2:
Problem Type #2:

Percentage Composition of a Compound
Problem Type #3:
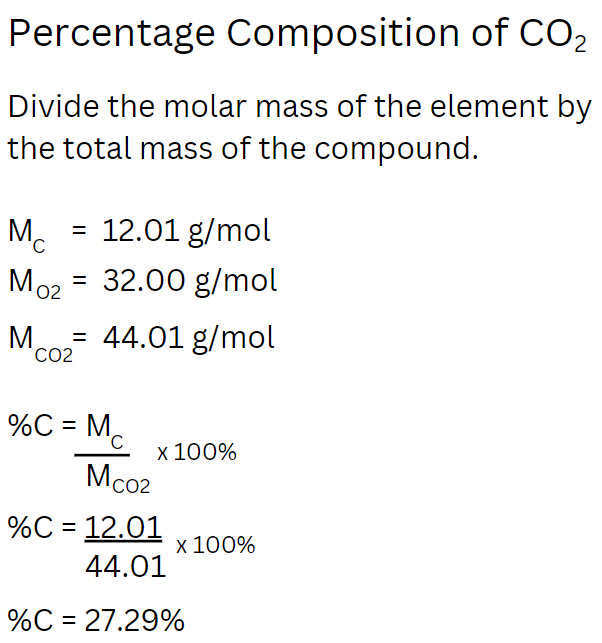
Empirical and Molecular Formula
Empirical Formula: Simplest Ratio
Example: Benzene (C6H6 → CH)
Molecular Formula: Actual number of atoms of each element making up molecule
Problem Type #4:
Finding Empirical Formula
- Convert percentage to mass (assuming 100g of compound)
- Find # of moles (n) using mass (m) and molar mass M [n = m/M]
- Compare ratios
Molecular Formula:
Let x be (EF)x
x = Actual Molar Mass/Empirical Molar Mass
Multiply EF formula (subscripts) by x
Gravimetric Stoichiometry
Mass and mole relationships
- Balance Chemical Reaction (find missing precipitates, combustions, subscripts)
- Find mol by dividing mass/molar mass (n=m/M)
- Find mass by multiplying mol by molar mass (m=nM)
- Follow coefficients (don’t include them in molar mass) and multiply/divide accordingly to the ratios to get mols of other elements/compounds
Both sides should be balanced
Hydrocarbons always produce CO2 and H2O (carbon dioxide and water vapor)
Percentage Yield
% Yield = Actual Yield/Theoretical Yield x 100%
- Actual Yield is less due to incomplete reaction, side reaction, loss of product, etc.
Limiting Reagents
Limiting Reagent: Reactant that is COMPLETELY consumed (controls how much product produced)
Excess Reagent: Reactant NOT completely consumed
- Find LR (smaller number)
- Find ER (bigger number)
- Find amount of product produced (Compare LR to ratio of Product)
- Find excess reagent remaining (Compare LR to ratio of ER a and subtract original ER to a)