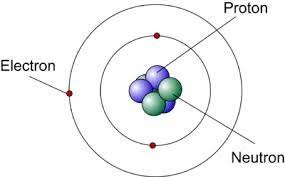
Atomic Structure
The timeline:
500BC - Democritus
 - they are indivisible: there are different types
- they are indivisible: there are different types
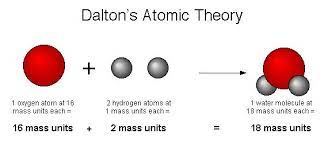
1808 - Dalton
1897 - Thompson: Plum Pudding Model
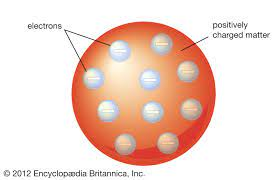 - electrons embedded in a solid atom of positive charge
- electrons embedded in a solid atom of positive charge
1911 - Rutherford
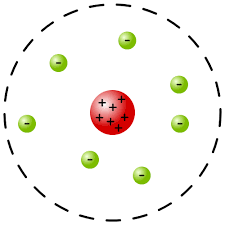 - discovered in alpha scattering experiment that the middle was positive and electrons were around the outside, most of the atom is space
- discovered in alpha scattering experiment that the middle was positive and electrons were around the outside, most of the atom is space
1913 - Bohr
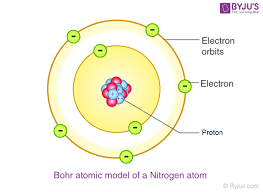 - electrons arranged in shells
- electrons arranged in shells
Chadwick and Rutherford: