Atomic Structure
Objectives
\
- Protons and neutrons exist in the nucleus, electrons exist outside the nucleus
- Protons are positively charged, neutrons are neutral, and electrons have a negative charge
- Relative means considered in relation or proportion to something else
- All atomic mass is relative
- Characterize protons, neutrons, electrons by location, relative charge, relative mass
%%Atom%%- the smallest particle of an element that retains its identity in a chemical reaction
Democritus
- believed that atoms were indivisible and indestructible
John Dalton

Dalton’s Atomic Theory
- states all elements are composed of tiny indivisible particles called atoms
- atoms of the same element are identical. Atoms from one element are different from atoms from another element
- atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds
- chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element, however, are never changed into atoms of another element as a result of a chemical reaction
%%Subatomic Particles%%- atoms can be broken down into smaller objects, which are called subatomic particles
- Proton
- Neutron
- Electron
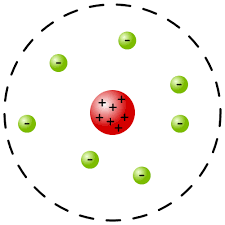
Rutherford’s gold foil experiment
- 1911; Fired alpha particles (Helium atoms without electrons) into a sheet of gold foil
- Most of the alpha particles went straight through, some deflected at large angles
- Led to the discovery of the nucleus
https://www.youtube.com/watch?v=B-k_kMwB1zM
Atomic Mass
- Atomic mass is based on a Carbon-12 isotope
- An atomic mass unit (amu) is defined as one twelfth of the mass of a carbon-12 atom
%%Neutron%%- subatomic particles with no charge that are located in the nucleus of an atom
-==Discovered by Chadwick in 1932==
Plum Pudding Model/ Chocolate Chip Model- Thompson’s theory that subatomic particles were evenly distributed throughout the nucleus of an atom
-proven to be incorrect
%%Protons%%- positively charged subatomic particles, that are located in the nucleus of an atom
-==discovered by Goldstein in 1886==
\
The atomic model
- states that the . The electrons are distributed around the nucleus and occupy almost all of the volume of an atom.
- improved the “chocolate chip cookie” model but was still incomplete
\