CHEM121 Notes - Limiting Reactants
Concept of Limiting Reactant
Definition:
The limiting reactant (LR) is the reactant that runs out first in a chemical reaction, limiting the amount of product that can be formed.
Example of Limiting Reactant with Ammonia (NH₃)
Scenario:
Calculating product formation with 5 grams of N₂ and an excess of H₂. Both reactants are needed to produce NH₃.
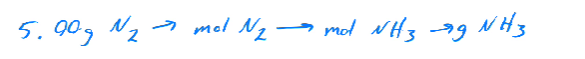
Outcome:
Once N₂ runs out, no more NH₃ can be produced regardless of the amount of H₂ left.
Calculation Steps:
Convert grams of N₂ to moles using the molar mass.
Use stoichiometry to convert moles of N₂ to moles of NH₃.
Convert moles of NH₃ back to grams using its molar mass.
Key Point:
N₂ is the limiting reactant in this scenario.
Example with Pizza Ingredients
Ingredients:
4 crusts, 15 ounces of sauce, 10 cups of cheese.
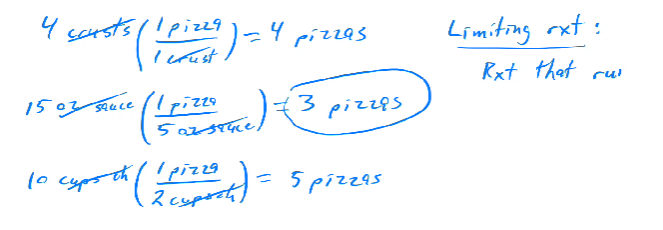
Calculating Maximum Pizzas:
Crust: One crust = one pizza (4 pizzas).
Sauce: 5 ounces per pizza (Can make 3 pizzas from 15 ounces).
Cheese: 2 cups per pizza (Can make 5 pizzas from 10 cups).
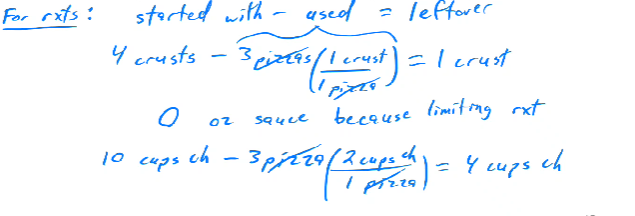
Conclusion:
The maximum number of pizzas is 3, limited by the sauce.
Definition:
The limiting reactant (sauce) restricts the product amount.
Leftover Ingredients Calculation
To find how much of each ingredient remains after maximum pizzas are made:
Crust (4 initial - 3 used) = 1 leftover.
Sauce (15 initial - 15 used) = 0 leftover (limiting reactant).
Cheese (10 initial - 6 used) = 4 leftover.
Terminology:
Reactants in Excess: Ingredients that remain after the reaction.
Theoretical Yield: Maximum amount of product that can be formed. In the pizza example, it was 3 pizzas.
Actual Yield: The measured amount of product actually obtained. For example, if only 2 pizzas were salvaged after cooking.
Percent Yield Formula: Percent Yield = (Actual Yield / Theoretical Yield) x 100%.
Example: (2 pizzas / 3 pizzas) x 100% = 67% yield.
Applying Concepts to Chemical Reactions
Example Reaction with N₂ and H₂
Reactants: 2 molecules of N₂ and 8 molecules of H₂.
Determining Limiting Reactant:
N₂ reacts with H₂ in a ratio according to the balanced equation. After calculations, O₂ was determined to be the limiting reactant because it would run out first, preventing further production of NH₃.
Detailed Example with Chemical Reaction Calculations
Given: 21 g of N₂ and 1.5 g of H₂.Objective: Identify the limiting reactant through stoichiometry and compute leftover amounts.
Steps to Solution:
Convert grams of N₂ and H₂ to moles using their respective molar masses.
Calculate the theoretical yields for ammonia (NH₃) derived from each reactant.
Identify the limiting reactant by determining which reactant yields the least amount of NH₃ (in this case, it may be found that H₂ is the limiting reactant).
Calculate the remaining amounts for N₂ and H₂ after the reaction completion.
Key Terms:
Limiting Reactant: The reactant that limits the amount of product made.
Reactants in Excess: The reactants left over after the reaction.
Theoretical Yield: The maximum calculated amount of product.
Actual Yield: The measured amount of product after the reaction.
Percent Yield: A measure of the efficiency of a reaction.
Common Reasons for Discrepancies:
Loss of product, incomplete reactions, side reactions, impurities.