Lecture 4 - Macronutrients (Carbohydrates)
Carbohydrates: An Overview
Introduction to Carbohydrates
Carbohydrates are composed of carbon, hydrogen, and oxygen atoms.
They are often referred to as sugars.
Carbohydrates are hydrophilic, meaning they interact well with water.
Smaller carbohydrates are soluble in aqueous environments and dissolve.
Larger carbohydrates (polymers) are insoluble but still interact with water.
Monosaccharides: Simple Sugars
Monosaccharides are the simplest carbohydrates and serve as fuel molecules for energy production in the body.
They can be as small as three carbons long.
Deoxyribose is a five-carbon monosaccharide found in the backbone of DNA; it has a cyclic structure.
Common six-carbon sugars:
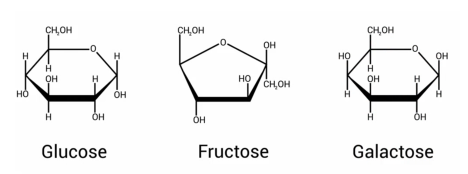
Glucose: An essential energy source for virtually all forms of life.
Fructose: A six-carbon sugar with a five-membered ring structure; used as a sweetener and can be converted into glucose.
Galactose: Very similar to glucose with a minor change in the position of a hydroxyl group; can be converted into glucose.
The only difference between glucose and galactose is the position of the hydroxyl group. In glucose, its position is "down", while in galactose, it is "up".
Mannose: Another six-carbon sugar.
Joining glucose, galactose, and mannose in different ways can produce over 12,000 different structures in a lab.
Disaccharides: Two Sugars Joined
Disaccharides consist of two monosaccharides joined together by an O-glycosidic bond (or O-glycosidic link).
Examples:
Sucrose (table sugar): Formed by joining a glucose molecule to a fructose molecule.
The enzyme sucrase breaks the bond between glucose and fructose to release them for use in the body.
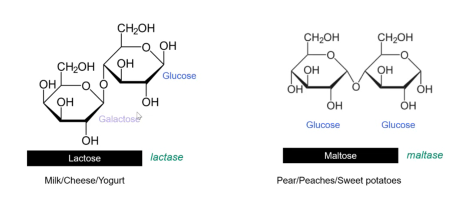
Lactose: Made up of galactose and glucose molecules joined by an O-glycosidic bond; found in milk, cheese, and yogurt.
The enzyme lactase is required to break the bond between galactose and glucose.
Lactose intolerance occurs when individuals do not produce enough lactase enzyme.
Lactose intolerance is more prevalent in certain populations, such as those of Asian descent, who do not traditionally consume large amounts of dairy.
Maltose: Composed of two glucose molecules joined together; found in fruits like pears and peaches, as well as sweet potatoes and pumpkin.
The enzyme maltase is needed to break the bond between the two glucose molecules.
Polysaccharides: Many Sugars Joined
Polysaccharides are formed when many monosaccharides are joined together.
They can be made up of the same monosaccharide (e.g., many glucose molecules) or different types of monosaccharides in various combinations.
Starch: A long polymer of glucose molecules; more than half of the carbohydrates ingested by humans is starch.
Amylose: A long, unbranched chain of glucose molecules.
Amylopectin: A branched structure of glucose molecules, allowing for more glucose to be added to the ends.
Starch is found in foods like wheat, potato, rice, and pasta.
The enzyme amylase breaks down the bonds between glucose molecules during digestion.
Cellulose: Found in plants and provides structural support for plant cell walls; it is not typically broken down for energy.
Cellulose is an insoluble fiber that aids in the digestion process in mammals.
Fiber Types and Digestion
Soluble Fibers:
Pectin is an example.
Slow down the movement of food in the small intestines, allowing for effective breakdown and nutrient absorption.
Insoluble Fibers:
Cellulose is an example.
Help move food quickly through the large intestine, reducing exposure to potential toxins.
Humans cannot break down cellulose because they lack the enzyme cellulase needed to break the bonds between glucose molecules.
Clinical Insight: Human Milk Oligosaccharides (HMOs)
Oligosaccharides are carbohydrates with 3-10 sugars joined together.
Human milk contains over 150 different oligosaccharides that protect newborns from infections.
These oligosaccharides are not digested by the child but appear to offer protection against bacterial infections.
HMOs are not found in infant formula.
Theory on how HMOs work:
Certain bacteria (e.g., Streptococcus) can colonize the vaginal epithelium and be transferred to the child during vaginal birth, potentially causing pneumonia, septicemia, or meningitis.
HMOs may prevent the growth of these bacteria by acting as food for the good bacteria.
The good bacteria then outcompete the bad bacteria, preventing them from growing on intestinal epithelial walls.
Research is ongoing to explore the therapeutic potential of HMOs as a new type of antibiotic.
Roles of Carbohydrates
Carbohydrates can attach to proteins (glycoproteins) and lipids (glycolipids), influencing cell adhesion, recognition, signaling, lubrication, and structural components.
In cell adhesion:
Carbohydrates play a role in sperm binding to the egg.
Lubricant or structural components:
Mucus contains carbohydrates that aid in lubrication.
Hormones:
Erythropoietin (EPO) is a hormone secreted by the kidneys that stimulates bone marrow to produce red blood cells.
The addition of a carbohydrate component to EPO stabilizes it, allowing it to remain in the blood longer.
EPO increases oxygen-carrying capacity by stimulating the production of red blood cells.
Recombinant EPO has been used by endurance athletes to enhance performance, but is banned in most sports.
EPO is also used to treat anemia, where individuals have low red blood cell counts.
Individuals living in high-altitude areas tend to produce higher levels of EPO due to lower oxygen concentrations.
Storage of Glucose
After digestion, carbohydrates are broken down into monosaccharides (mainly glucose), which are then picked up by tissues and organ cells for energy.
Excess glucose is stored as glycogen for later use.
Glycogen is a homopolymer made up of glucose molecules and has a branched structure similar to amylopectin.
Glycogen is primarily stored in skeletal muscles and the liver.
There is a limited amount of glycogen that can be stored.
Excess glucose, after glycogen stores are full, is converted into fats for storage.
Carb Loading
Carb loading involves maximizing glycogen stores before an endurance event.
Athletes consume a large amount of carbohydrates before, during, and after the event to ensure glycogen supplies are full.
Glycogen helps maintain blood sugar levels.
During endurance sports, glycogen stores can be depleted within a few hours.
Simple vs. Complex Carbohydrates
Simple Carbohydrates:
Made up of one or two sugar units (monosaccharides or disaccharides).
Broken down quickly and easily, causing a rapid increase in blood glucose levels.
Provide a sudden burst of energy followed by a quick drop.
Examples: chocolate, cookies, candies, processed foods.
Complex Carbohydrates:
Made up of many sugars joined together (polysaccharides).
Take longer to break down, resulting in a slower release of glucose into the bloodstream.
Provide a prolonged release of energy.
Help maintain stable blood glucose levels and promote a feeling of fullness.
Examples: vegetables, brown bread, wheat.
It is recommended to consume more complex carbohydrates and moderate amounts of simple carbohydrates.