module 5
5.1 static and dynamic equilibrium
enthalpy (ΔH): energy required by/released in a reaction.
exothermic (-ΔH): energy released/graph ends lower than start
endothermic (+ΔH): energy absorbed/graph ends higher than start
ΔH reaction = ΔH products - ΔH reactants
entropy (ΔS): amount of disorder in system, increases as particles become more randomly dispersed
solid → liquid, solid → gas, liquid → gas | increase in ΔS (more freedom to move) |
|---|---|
produce an increase in number of particles/more particles in products than reactants | increase in ΔS |
increasing volume of a gas sample (at constant temp) | increase in ΔS |
increasing temperature of a gas = increases dispersion of kinetic energy of the gas | increase in ΔS |
2nd law of thermodynamics: total entropy of Universe is increasing
Gibbs Free Energy (ΔG): thermodynamically favoured/spontaneous
calculated by ΔG = ΔH - TΔS, where T = temperature
ΔG < 0, products are favoured, spontaneous reaction
ΔG > 0, reactants are favoured, non spontaneous
ΔG = 0 is equilibrium
+ΔH | -ΔH | |
|---|---|---|
+ΔS | depends on temp | -ΔG |
-ΔS | ΔG | depends on temp |
chemical equilibrium: rate of forward reaction equal to rate of reverse reaction
static equilibirum: very rare, forward and back rates both equal to 0. equilibirum of irreversible reactions
dynamic equilibrium: forward and reverse reaction rates equal, not equal to 0. can appear unchanging
a system at equilibrium can’t produce any energy. a non-equilibrium system is deliberately and permanently prevented from reaching equilibrium eg. combustion
all reactions will move towards equilibrium
homogenous equilibrium: all same state
collision theory
collision theory: particles must collide to react with each other
successful collision: must collide with force higher than required activation energy, and at correct orientation for bonds to be formed
rate of reaction = number of successful reactant collisions = Δconc/time
conc, pressure, volume, temp, SA, catalysts affect rate
activation energy = Ea
Maxwell-Boltzmann distribution curve: probability curve. shows number of particles that have a particular kinetic energy.
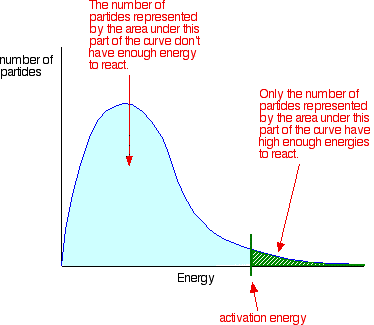
small area under curve = small number of molecules
reaction rate
temperature: increase = increase in rate, increases kinetic energy
concentration: increase = increase, more particles increase chance of collision
pressure: increase = increased reaction rate as greater collisions with walls. increasing temp/conc is same as increasing pressure
surface area: more exposed surface area = more successful collisions = greater reaction rate
catalysts: alternative pathway for reaction to occur, reduces activation energy required. therefore more successful collisions, but not more overall collisions = increased reaction rate. often also impact orientation, therefore more likely to be correct collision
known non-equilibrium reactions
generally non-equilibrium systems are open and consist of irreversible processes under the conditions at which they operate
non-equilibrium systems are perpetually moving away from equilbrium. ΔG is positive
combustion: reverse reaction has a highly positive ΔG at any temperature; therefore reverse reaction won’t occurr
photosynthesis: large overall negative change in enthalpy and large positive change in entropy = overall negative gibbs free energy, can occur spontenously
5.2 factors that affect equilibrium
dynamic equilibrium: ΔG=0. constant concentrations, but not necessarily equal
Le Chatelier’s Principle
stress: change imposed on equilibrium that changes conditions of the system and alters position
equilibrium position: relative amounts of reactants/products
if stress disturbs equilibrium balance, it will shift to restore balance (in direction that counteracts the stress)
any stress applied to system at equilibrium can be viewed as an effect on concentration or temperature
le chatlier’s principle states the system will shift to restore balance/shift in the direction that will counteract the stress
temperature: depends on the forward/reverse enthalpies. if temperature is increased, the reaction will move in the direction of +ΔH to ‘cool’ and vice versa
concentration: if concentration of all gases increases (eg. volume decrease, pressure increase), will shift to produce less gas. if concentration of one gas increases, will shift to produce less of that specific gas. if inert gas is added, no change due to no impact on concentration overall
eg. N2 + 3H2 → 2NH3
increase volume = shift left
N2/H2 added = shift right
equilibrium & collision theory
collision theory allows an explanation of why le chatlier’s principle occurs
temperature:
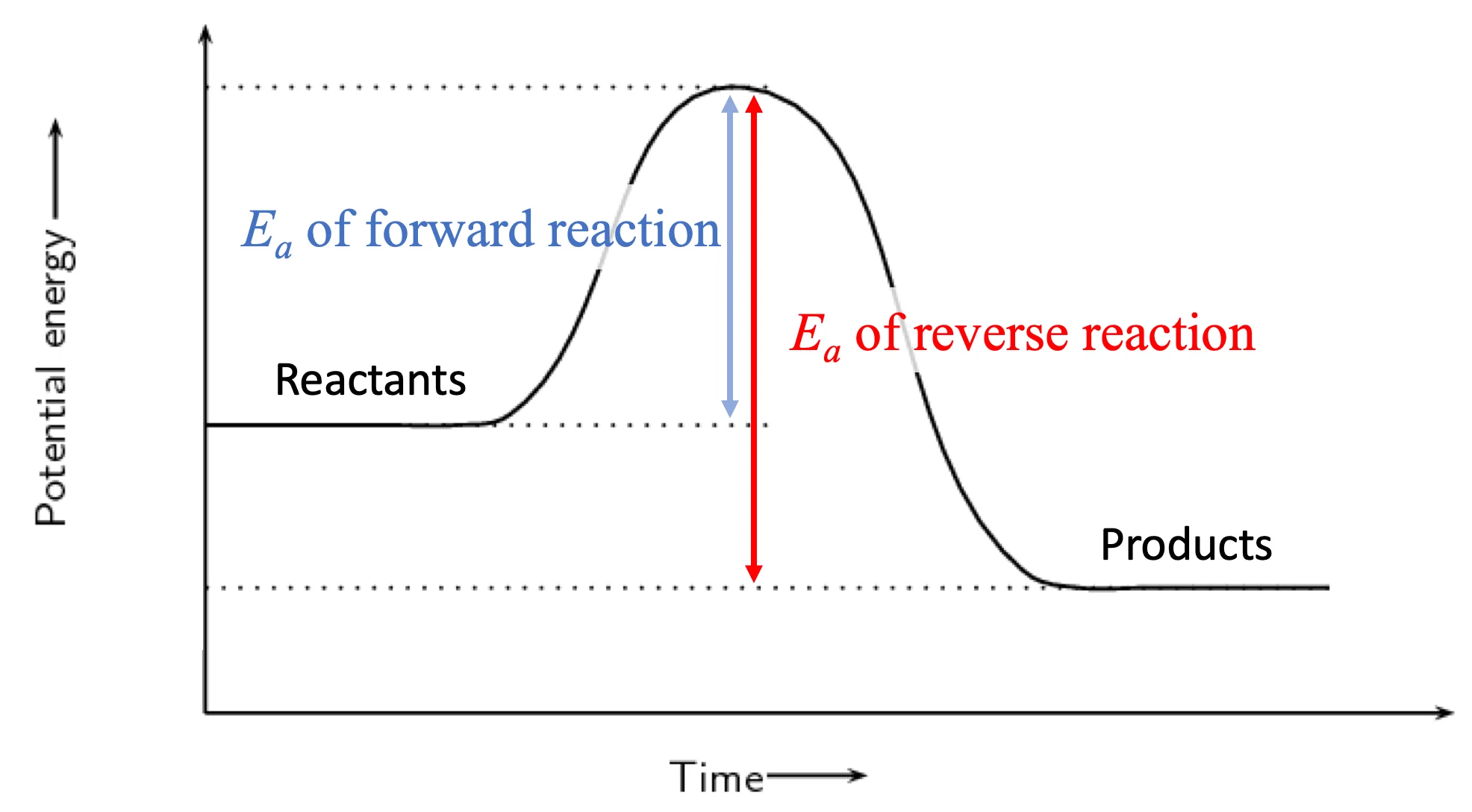
when heated, all reaction rate will increase, but proportionally more in reverse reaction (endothermic reaction). this is because the higher temperature will increase the number of particles with sufficient energy by a greater proportion for the direction with the highest activation energy
decreasing will favour the direction with the lower activation energy (exothermic reaction). this is because the lower temperature will decrease the number of particles with sufficient energy by a greater proportion for the direction with the highest activation energy
 concentration: increase in concentration of reactants shifts equilibrium to favour products/foward reaction increases compared to the reverse. percentage of successful collisions remains constant!!!! total number of collision increases. decreasing concentration reactants = products are more likely to collide, therefore reverse reaction is favoured until new equilibrium is found
concentration: increase in concentration of reactants shifts equilibrium to favour products/foward reaction increases compared to the reverse. percentage of successful collisions remains constant!!!! total number of collision increases. decreasing concentration reactants = products are more likely to collide, therefore reverse reaction is favoured until new equilibrium is found
pressure:
if increased due to temperature, use previous explanation
for gases,concentration is considered in terms of partial pressure. partial pressure determined by imagining one gas is only gas in container ie. how many times will it collide with walls. increasing number of particles of any of the reactant/product gases will increase partial pressure/concentration and collision theory can be applied. converse applies for removing.
adding inert (non-reacting) gas will not alter
volume:
if gas only on one side of reaction: decreasing volume will increase concentration of products/reactants and equilibrium will shift accordingly
if gas on both sides: changing volume will change concentration of products and reactants by same factor. the side with greater stoichiometric change will have more frequent succesful collisions and the equilibrium will shift away
eg. N2O4 (g) → 2NO2
2 mol 4 mol
if you reduce volume of both by half, goes up by factor of 2. therefore 8 mol NO2 and 4 mol N2O4. as NO2 gone up by 4, equilibrium shifts left/reverse
catalyst: lower activiation energy. equilibrium will be reached more quickly, but will not change position
equilibrium graphs
0 gradient indicates equilibrium has been established
vertical line indicates stress has ultimately altered concentration, directly (adding/removing) or indirectly (altering pressure or volume)
smooth curved sections = le chatlier change
temperature = smooth curved response
5.3 calculating the equilibrium constant
equilibrium expressions: shows relationship between concentration of reactants and products
aA + bB → cC+ dD
Keq = [C]c[D]d/[A]a[B]b
aqueous must be in mol/L
treat as unitless
when referring to reverse reaction, inverse
pure solid/liquid present has concentrtion of 1 and disappears from equilibrium expression
big Keq (>10^3) favours products, small (<10^-3) favours reactants
RICE boxes
to determine Keq with incomplete data
Reaction: A → 2B
Initial conc: 3M 0
Change in conc: -x +2x
equilibrium: 3-x 2M
determine using quotient Q
aA + bB → cC+ dD
Q = [C]c[D]d/[A]a[B]b
compare Q and Keq
Q = K at equilibrium
Q < K forward reaction favoured
Q > K reverse reaction favoured
ΔG = 0 then Q = K
ΔG>0 then Q>K
ΔG<0 then Q<K
impact of temperature of Keq
more products = higher Keq
therefore impact of temperature depends on whether system is exothermic or endothermic, and therefore which way heating will shift
different uses of Keq
for ionic: dissociation of ionic compounds can be explored through Keq (Ksp)
for acid/bases: see module 6
solution equilibria
dissolution of ionic compounds in water
water molecules are polar (oxygen partially negative, hydrogen partially positive)
when ionic substances added to water, hydrogen atom is attracted to negatively charged ions, oxygen to positively charged = ion-dipole attraction
when these ion-dipole attractions are strong enough, ions are removed from the ionic lattice and enter solution
when an ion is surrounded by water = hydrated
dissociation: solid ionic compound separates into charged ions
saturated solutions: unable to dissolve more solute at a particular temp
aboriginal people
detoxifying food works due to constant concentration gradient/le chatliers
cycads and other plants eg. yellow walnut: seeds contain the toxin cycasin, solubility of 56.6 g/L. toxins can be leached in multiple ways: roasted (breakdown chemical strucutre) and then leached in still or running water for at least 1-3 days
bitter yams (toxic oxalates) and saltbush also use ;leaching via soaking/placing in running water
conduct a practical to analyse composition of substances are mixed
potassium chloride and silver nitrte
potassium iodide and lead nitrate
sodium sulfate and barium nitrate
ksp
solubility product of dissociation
low ksp = not much solute can be dissolved before solution is saturated
can be used to calculate if a precipitate will form eg. by calculating Qsp, if greater than Ksp precipitate will form
common ion effect: greatly decreases the solubility of ionic compounds due to Le Chateliers
eg. AgCl added to NaCl, there are already Cl ions, so will shift away from products
quantitative analysis of an equilibrium reaction
(Fe3+HNO3)(aq) + KSCN-(aq) ⇌ [FeSCN]2+ (aq)
Keq = [[FeSCN]2+]/[Fe3+][SCN-]
turns red →
exothermic reaction in forward direction! therefore forward direction favours lower temp.
intro:
can be challenging, most methods of detecting inadvertantly involve disturbing equilibrium eg. if you titrate, will react and remove chemical species of interest and equilibrium will shift and mol will be too high
therefore UV spectrophotometry in situ used. involves using absorbance of photons by speciees of interest (in this case, iron thiocynate) to measure concentration
prepare a series of standard solutions with excess Fe(NO3)3, so KSCN is limiting reagent. forced into completion, therefore in static equilibrium
for the second part, variety of volumes were combined and allowed to reach dynamic equilibrium (not limiting reagent). recorded against curve
risk assessment:
uv spectrometer: light can cause eye damage. keep lid closed whilst in operation.
iron thiocynate: corrosive to skin, eye damage, inhalation
nitric acid: corrosive (can severely irritate and burn the skin and eyes with possible eye damage), can irritate nose and throate, can irritate lungs (cause build up of fluid), nausea/vomiting/abdominal pain. not combustible but strong oxidiser that enhances combustion of other substances. explosion risk, releases dangerous gases in fire
potassium thiocynate: irritate respiratory tract (long term exposure may result in dizziness/general weakness), may irritate eyes, skin, cause nausea. non-combustible
constructing a standard curve:
100x more FeNO3 than KSCN, to ensure all KSCN is consumed in the reaction. forces reaction RIGHT, only product and excess iron remain
HNO3 solution is present to stabilise the Fe ions in the 3+ oxidation state. added in different volumes to ensure each solution ends up with the same total volume
questions:
experiment run at 447nm = blue region colour. as solution is orange-red, absorbs this complimentary colour best
NOTE: equilibrium position different to equilibrium constant.
Equilibrium constant is the number that gives the relationship between amounts of products and reactant of a reaction mixture at its equilibrium. Equilibrium position is the moment at which the forward reaction of the equilibrium is equal to the backward reaction
concentration and absorbance directly related
Fe concentration much higher than KSCN to drive to completion
reaction is exothermic forward ie. heat is a product. therefore warmer day will drive left and produce less FeSCN