CHEMISTRY
Chemical changes
Acids and alkalis
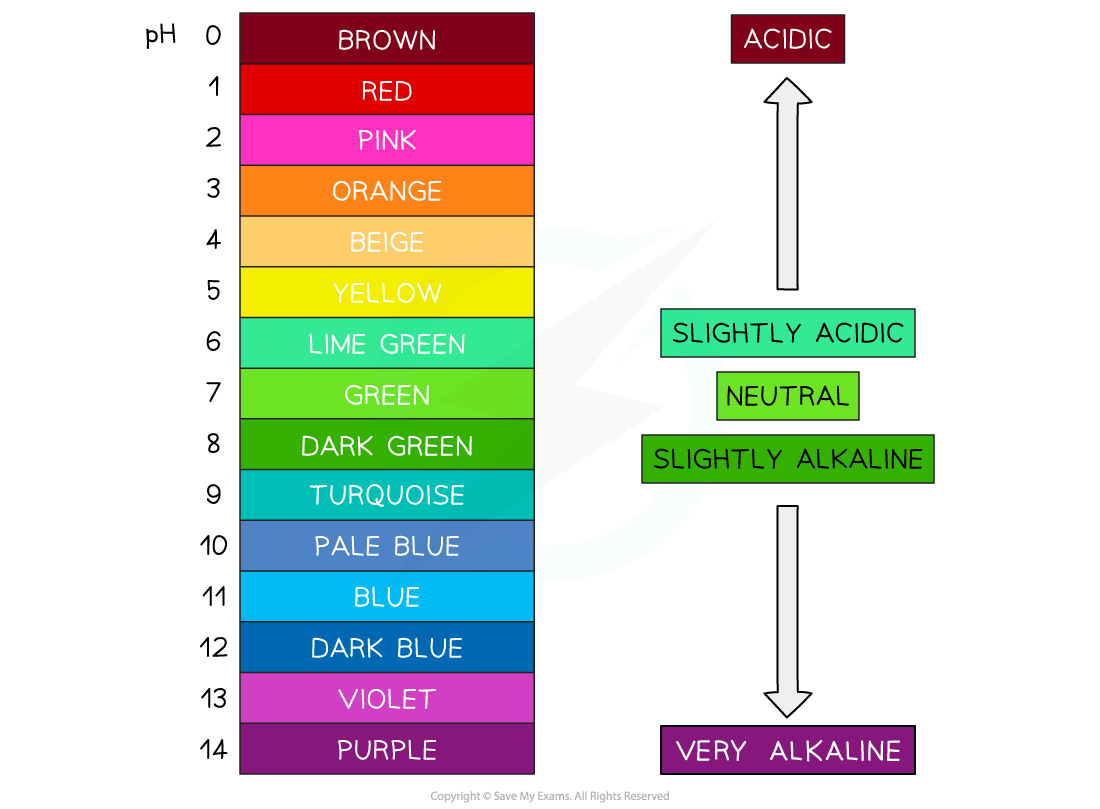
For example, stomach acid is around pH 2, and bleach is around pH 12
Indicators are chemical dyes that change to laine with the pH colour
Universal indicator is the most common, and changes to the same colours as above 🙂
A pH probe and meter give a number reading, and are more accurate and precise
Acids are any substance that forms an aqueous solution with a pH less than 7
A base is any substance with a pH greater than 7
Alkalis are any substances that dissolves in water to form a solution with a pH more than 7
Neutralisation reactions are when you react an acid and a base to form a solution CLOSER to pH 7 (pure water)
They form a salt and water
HCl + NaOH NaCl + H2O
Acids I need to know
Hydrochloric acid is HCl
Sulfuric acid is H2SO4
Nitric acid is HNO3
Alkalis I need to know
Sodium hydroxide is NaOH
Calcium carbonate is CaCO3
Titration practical
A titration is an experimental technique used to find an unknown concentration of an acid or alkali
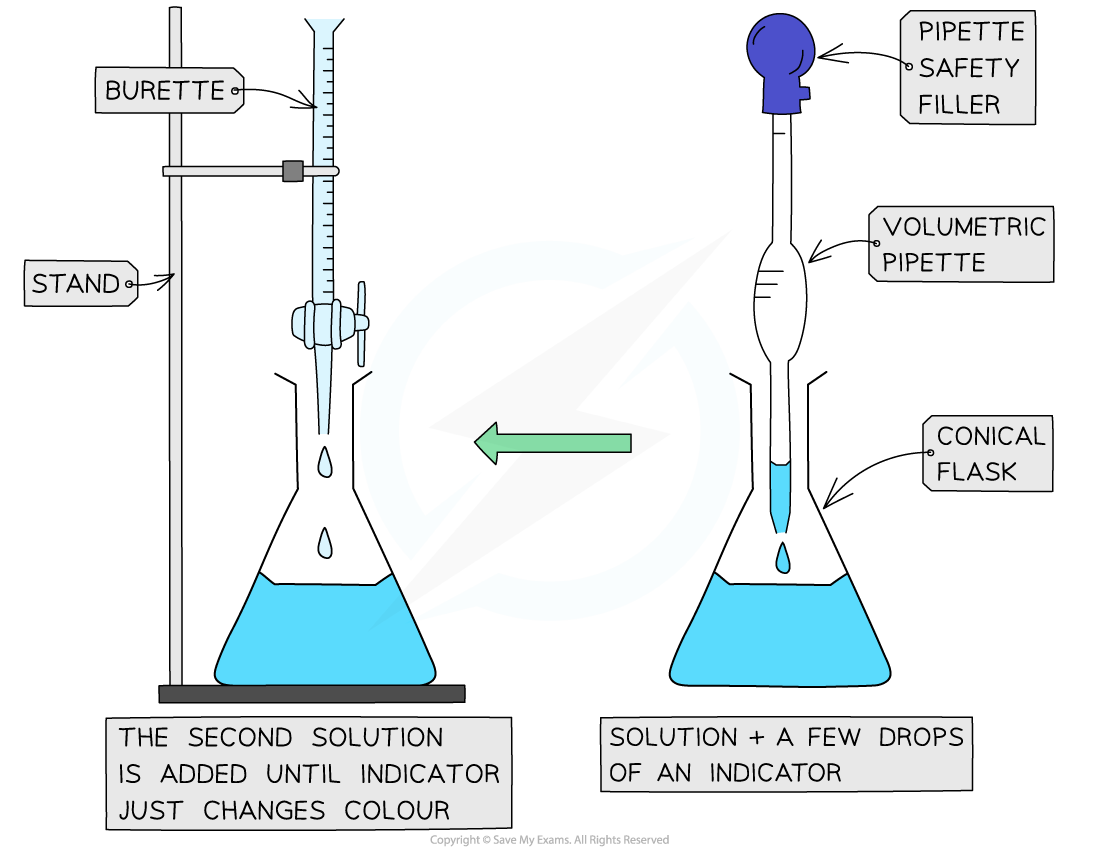
Neutralising an alkali with acid
Use a pipette to add 25cm3 of alkali to a conical flask
Add a few drops of an indicator (can’t be universal indicator) and place on a white tile
Fill a burette with acid and note the starting volume
Slowly add acid from the burette to the conical flask, swirling slowly, so that it is evenly distributed
Stop when the end point is reached - the acid has neutralised the alkali and the indicator has changed colour
Note the final volume reading and calculate the total acid added
Repeat until you get concordant results - within 0.10 cm3 of each other
Calculate a mean
Indicators that can be used:
Litmus is RED in acids, and BLUE in alkaline solutions
Phenolphthalein is COLOURLESS in acids, and PINK in alkaline solutions
Methyl orange is RED in acid, and YELLOW in alkaline solutions
Universal indicator can’t be used as it gradually changes, and you can’t clearly see where it has reached the end point
Strong and weak acids
Acids are ionised in aqueous solutions to release H+ ions
For example, HCl exists as H+ and Cl- ions
Strong acids ionise completely - all particles will dissociate
Like HCl or H2SO4
Concentration is the amount of acid per amount of volume
The amount of H ions per unit of volume
Weak acids don’t fully ionise - their reactions are reversible and stop reacting when they reach an equilibrium (net reactions)
Like CH3COOH - ethanoic acid
pH is a measure of concentration of H+ ions in a solution
The higher the concentration, the lower the pH
HCl is a strong acid and could easily be pH0 at most concentrations
If it was a weak acid, to be pH0, it would have to be very very concentrated
Neutralisation reactions
Metal oxide + acid salt + water
Metal oxides end in O
Metal hydroxides + acid salt + water
Metal hydroxides end in OH
Metal carbonates + acid salt + water + carbon dioxide
To make a soluble salt, we have to react an insoluble base with an acid
We gently heat an acid, and slowly and a base a bit at a time until it stopes dissolving
This shows the acid is neutralised and that the base is in excess
Then filter the excess base, and the product is dissolved soluble salt
To form crystals from this solution, we can gently heat it to evaporate any water (at less than 60 degrees)
When crystals begin to form, stop heating and leave to cool
This will cause more crystals to form, which can be filtered and dabbed dry
The reactivity series and displacement
Potassium | please |
Sodium | send |
Lithium | little |
Calcium | Charlie’s |
Magnesium | monkeys |
Aluminium | and |
CARBON | cheeky |
Zinc | zebras |
Iron | in |
Tin | thick |
Lead | large |
HYDROGEN | heavy |
Copper | cages |
Mercury | most |
Silver | securely |
Gold | guarded |
Elements below carbon can be reduced with carbon (in metal oxides)
Reactivity is how easily something forms a positive ion
Metals + acid Salt + hydrogen
Reaction gets less violent down the reactivity series
Causes bubbles, explosions and temperature changes
Metals + water Metal hydroxide + hydrogen
Only in the most reactive metals
Displacement example:
Mg + FeSO4 MgSO4 + Fe
Magnesium displaces iron, as it is more reactive
Separating metals from metal oxides
Oxidisation is the process of losing electrons (gaining oxygen)
2Mg + O2 2MgO
Reduction is the process of gaining electrons (losing oxygen0
2MgO 2Mg + O2
OILRIG - Oxidisation is loss, reduction is gain
Most metals are fairly reactive
They reactive with oxygen (from the air) to form metal oxides
If they are really unreactive, they don’t react, like gold
To get pure metals, we want to reduce them to remove the oxygen
This is done by reacting metal oxides with carbon (displacement)
Oxygen will react with carbon and leave behind the pure metal
This only works with metals less reactive than carbon
It is cheap and quick
For example, 2Fe2O3 + 3C 4Fe + 3CO2, when mining for iron
More reactive metals are extracted via electrolysis, which uses lots of energy and is expensive
The process of electrolysis
Electrolysis involves using to break down electrolytes to form elements
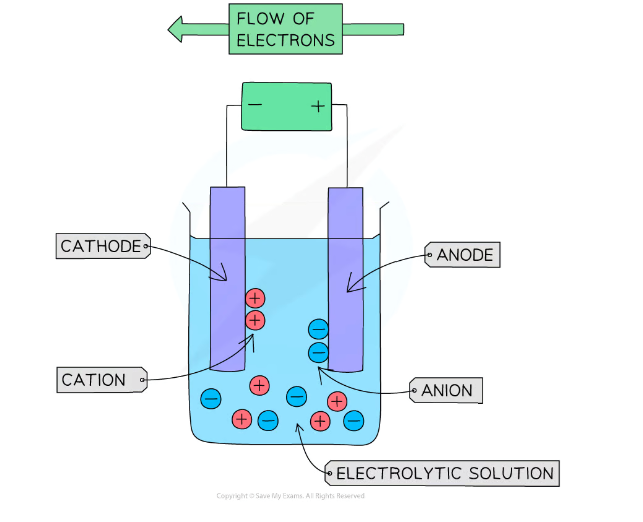
- ions are attracted to the anode (+ electrode)
They are then discharged, and become atoms
+ ions are attracted to the cathode (- electrode)
They are then discharged to become atoms, and produce either a solid or a gas
At the cathode, Pb2+ + 2e- Pb, reduction which forms solid lead
At the anode, 2Br- Br2 + 2e-, oxidisation which pairs up and forms gas (covalent bonds)
Electrons at the anode that are loss are passed through the wire to the cathode, where they are given off to the positive ions to form atoms
Electrolysis of aqueous solutions
Electrolysis is used to split compounds into their elements
Soluble compounds can be dissolved in water to become aqueous, so charged particles are free to flow
Because the compounds are dissolved in water, both the ions of the compound, and H and HO ions will be present
For example:
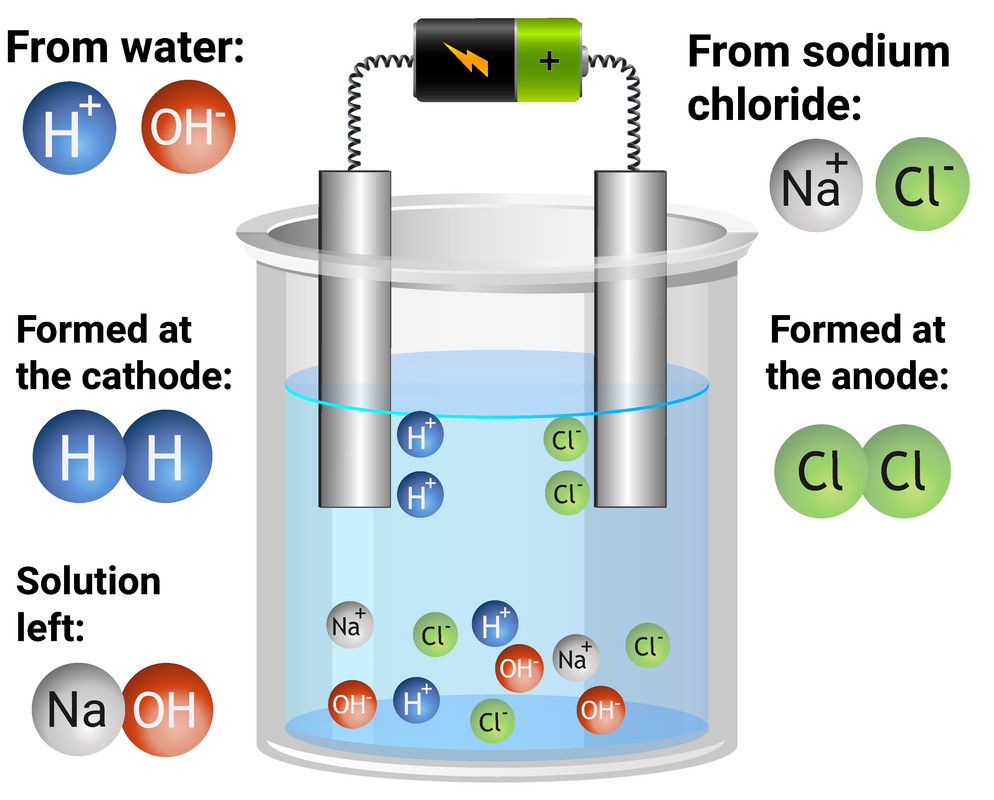
The cathode attracts positive ions - Na+ and H+
Only the less reactive will be discharged at the cathode
As hydrogen is less reactive than chlorine, hydrogen atoms will be formed, and given off as hydrogen gas
The anode attracts negative ions - OH- and Cl-
If a halide is present, it will be discharged. If not, OH- will be discharged
Halides are any of group 7 - F, Cl, Br, I and At
Chloride is a halide, so chloride is discharged, and forms chlorine gas at the anode
This leaves NaOH in the beaker as a solution
Oxidation and reduction in terms of electrons
Oxidation is loss, reduction is gain
Reduction and oxidisation reactions both take at the same time - redox reactions
This is because the electrons that are loss also need to be gained
Ionic equations are used to show displacement
They only show the particles that take part in the reaction and change
Ca + Fe2+SO42- Ca2+SO42- + Fe
Ca + Fe2+ Ca2+ + Fe
SO42- is left out as it doesn’t change
Half equations show the gain and loss of electrons for each element involved
Ca Ca2+ + 2e-
Fe2+ + 2e- Fe
The charged particles are always placed on the same side
Reactions of acids with metals
The products of reactions of acids with metals are always a salt and hydrogen
Acid + metal salt + hydrogen
They are redox reactions
2H+ + Mg Mg2+ + H2
Magnesium loses electrons - is oxidised
Hydrogen gains electrons - is reduced
Soluble salts
Soluble salts can be prepared by reacting an acid with a suitable insoluble reactant
The reactant can be a metal, metal oxide, carbonate or a metal hydroxide
The reactant depends on the salt required
For example, copper doesn’t react with dilute acids, so can’t be used, and sodium is too reactive to use
The reaction between an acid and a metal produces hydrogen
Hydrogen is flammable, so we usually use metal oxides or carbonates and an acid instead
Making a salt
Add the powdered insoluble reactant to an acid in a beaker, 1 spatula at a time
The acid can be gently heated by a Bunsen burner to increase the reaction speed (particles have more energy)
Stir between each addition and continue until the powder is in excess and no longer reacts
Filter the mixture in the beaker with filter paper and a funnel to remove excess solid - this means the filtrate now only contains the salt and water
Heat the solution in a evaporating dish over a water bath
Stop when small crystals begin to appear - the solution is now saturated and most of the water has evaporated
Leave the solution for 1-2 days at room temperature to allow the rest of the water to evaporate and leave large crystals
Dry by gently dabbing with filter paper
Electrolysis of molten ionic compounds
This is the process of separating elements in insoluble ionic compounds (splitting with electricity)
In electrolysis, the electrolyte has to be a liquid or aqueous solution that contains an ionic compound - so the ions can move freely
CuSO4 is soluble, and can be dissolve in water to create this
However, lead bromide is insoluble and has to be melted and made molten to allow ions to move freely

PbBr2 is heated to become molten
It splits into it’s two ions - Br- and Pb2+
Br- is attracted to the positive anode and is discharged as bromine gas
Br- Br + 2e-
Two bromines react covalently to form 2Br as a gas
Pb2+ is attracted to the negative anode and is discharged as solid lead
Pb2+ + 2e- Pb
This creates a layer of molten lead in the beaker
Ions are being oxidised and reduced at the electrodes
Cathode = reduction
Anode = oxidisation
Electrons that are lost when Br- become Br are transported via the wire with current to the anode, where Pb2+ gains two electrons
Extracting metals with electrolysis
Reactive metals are extracted from metal oxides by melting and making them molten compounds
The cheapest way to reduced a metal from an oxide is with carbon, but it only works with metals that are less reactive than carbon
So we use electrolysis instead, which is expensive and needs lots of energy
Electrolysis only works in ions can freely move through the solution
For example, Al2O3 Al + O2
Aluminium is more reactive than carbon, so electrolysis is used
Aluminium oxide is found as a solid, and found mixed with bauxite when mined
This needs to become molten 🔥
First, we need to purify Al2O3 from bauxite
Then, we mix aluminium oxide with cryolite as it lowers the melting point
Aluminium oxide has a very high melting point (it was ~ 2000 degrees C), but is lowered by cryolite
We then melt this mixture to become molten - lots of energy is required as the melting point is still high
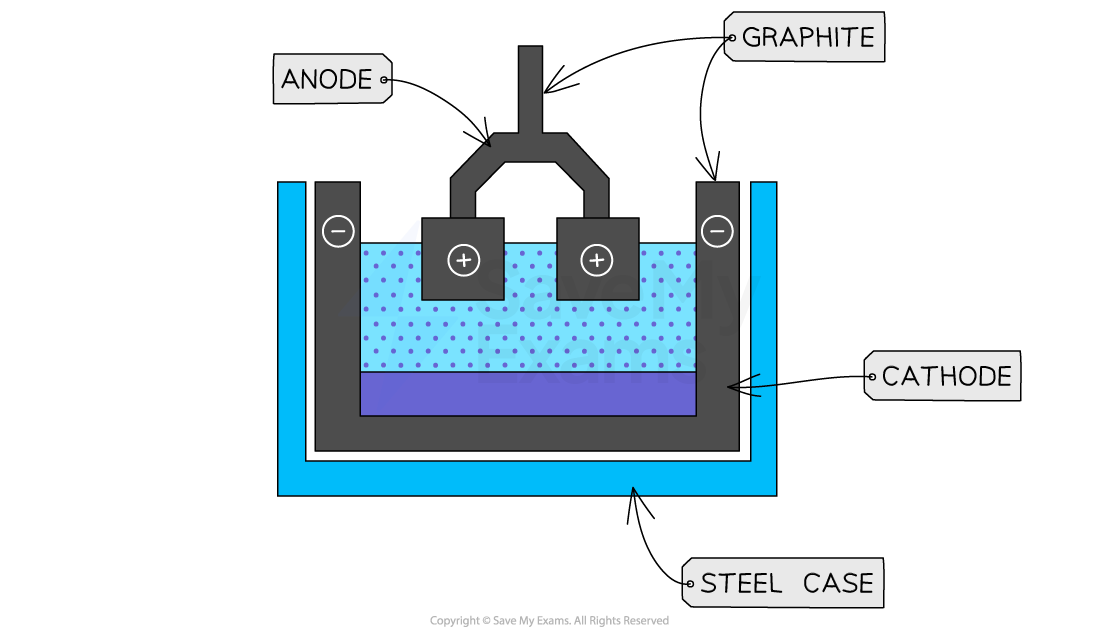
The electrodes are made of graphite, and the cathode is found around the outside of the steel case
O2- is attracted to the anode in the centre
At the anode, it is discharge and becomes oxygen gas (makes a pair and covalently bonds once atoms0
2 electrons are lost per oxygen, and they are transferred through the wire to the cathode for reduction
2O2- O2 + 4e-
Al3+ is attracted to the cathode around the edge
It is discharged and becomes Al
3 electrons are gained from the electrons lost at the anode
The aluminium formed pools at the bottom, and leaves via a channel at the bottom to be collected
Al3+ + 3e- Al
2Al2O3 (l) 4Al (l) + 3O2 (g)
Atomic structure and the periodic table
Atoms, elements and compounds
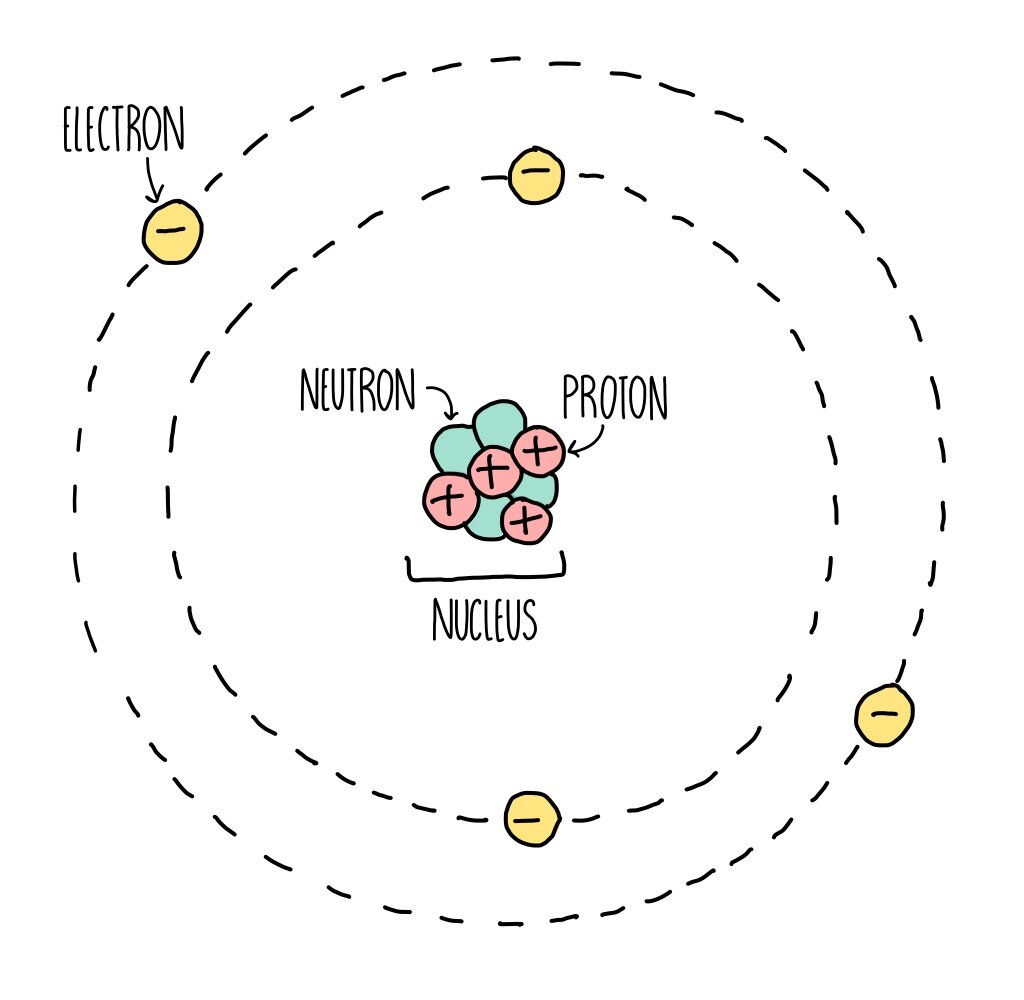
Atoms make up everything and have a radius of around 0.1 nanometres (1 × 10-1m)
Neutrons have a relative mass of 1, and no charge
Protons have a relative mass of 1, and a positive 1 charge
Electrons have a very small mass, and a charge of negative 1
Elements are different types of atoms, and are shown on the periodic table

An isotope is an element with the same number of protons, but different numbers of neutrons, so therefore different mass numbers
Relative atomic mass of an element’s isotopes = the sum of(isotope abundance x mass) / sum of percentage of all isotopes
Should be divided by 100 if all isotopes are present
Compounds are two or more different elements that are chemically bonded
H2O or CO2, but not O2 OR Cl2
Elements in compounds are always found in the same proportions
Molecules
Molecules are two or more atoms chemically bonded
For example, O2 and CO2 are both molecules
Electronic structure
The first shell of an atom has 2 electrons, then 8 electrons and then 8 electrons (2,8,8)
The group number tells you the number of electrons an element has in it’s outer shell
The number of shells an element has is the same as the number of shells it has
The size and mass of atoms
Atoms have a radius of around 1 × 10-10 m, or 0.1 nanometres
Their mass is made of the total number of neutrons and protons
Development of the model of the atom
Democritus - Atomic theory
In 500 B.C
Everything is made up of tiny indestructible particles, with empty space between
John Dalton - In the 1800’s
Everything is made up of ‘solid spheres’
Different types of spheres are different elements
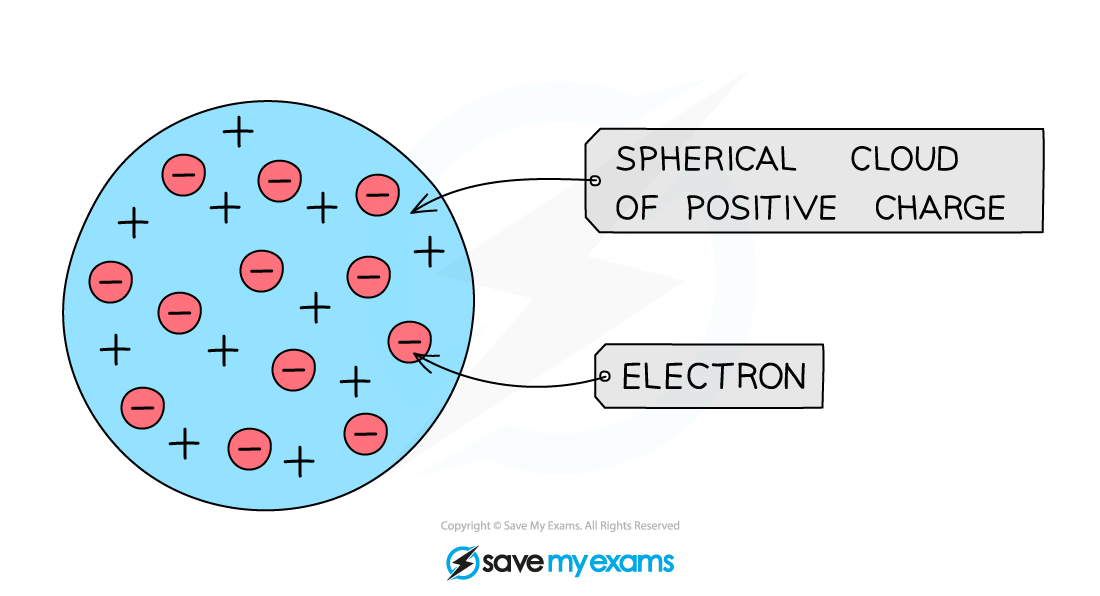
JJ Thompson - The plum pudding model
In 1897
He found that atoms couldn’t be solid spheres and that most contained charged particles
He thought the atom was a ball of positive charge with discrete negative particles embedded in it
Ernest Rutherford - Alpha scattering experiment
In 1909
He fired positive alpha particles at a thin sheet of gold
If the positive mass was spread out, like in the plum pudding model, all the particles should pass through as the positive charge is not dense enough to repel
However, not all of the particles did and some were deflected
This showed that there was a concentrated positive nucleus - the nuclear model
Niels Bohr - In 1913
He suggested that electrons orbit the nucleus in shells, which prevents the atom from collapsing like in Rutherford’s model (1909)

Rutherford - In 1913
He suggested that the nucleus in made up of small discrete particles, called protons, and wasn’t just a cloud of charge
James Chadwick - In 1932
He found that the nucleus contained neutrons with mass
The periodic table
The periodic table was created by Dimitri Mendeleev in the mid 19th century
He left spaces to predict new, undiscovered elements
Columns are called group and have similar chemical properties and have the same number of electrons in the outer shell
Rows are called periods and shows the number of shells an element has
The development of the periodic table
Before Dimitri Mendeleev, neutron and proton scientists tried to classify their discoveries in order of atomic weights
They were incomplete, and the groups were often inaccurate
Dimitri Mendeleev made the periodic table in 1869, and he predicted new elements and left spaces in his table
He included elements mass number, element symbol and atomic number in his table
He made groups with similar chemical properties, with the same number of electrons in the outer shell
Metals and non-metals
Metals are found on the left side of the periodic table
They form positive ions - they lose electrons
The fewer electrons they have in their outer shells, the easier they are lost
They get more reactive the further down the table you go
This is because they have more shells, so the outer shell electrons have little attraction to the nucleus and are lost easily
Metals metallically bond so are very strong
They are malleable, conductors of electricity and heat
They have high melting and boiling points, and they are shiny and sonorous
Non-metals are found on the right side of the periodic table
They form negative ions, or none at all
Non-metals have lower densities than metals, are brittle and dull in colour
They don’t conduct and have low melting and boiling points
The transition metals have typical metal properties
They can form more than one ion
For example, iron (II) and iron (III)
They also form good catalysts
Group 1 - The alkali metals
The group 1 elements are lithium, sodium, potassium, rubidium, caesium and francium
Their properties include:
Soft, low densities and low melting points, which are different to other metals
They get more reactive down the group
Their melting and boiling points decrease down the group
They have one electron in their outer shells, which can be lost mor easily the further down the group as the are more shell
They are further from the nucleus, so less attraction as there is a further distance between
They are almost always found as ionic compounds
With electrostatic forces between non-metal and alkali metals
Alkali metals and water react vigorously to form metal hydroxide and hydrogen gas
Alkali metals and chlorine gas react to form metal chloride salt
Na + Cl NaCl (salt)
Alkali metals and oxygen react to form metal oxides
Lithium and oxygen react to form lithium oxide
Sodium and oxygen react to for sodium oxide OR sodium peroxide
Potassium and oxygen react to form potassium peroxide OR potassium superoxide
Group 7 - The halogens
All of the halogens are a bit dangerous ☹
They all exist as diatomic molecules
Make covalent bonds to gain a full outer shell, like chlorine
They can form covalent bonds with other non-metals as well
Properties and trends of the halogens:
Their melting and boiling points increase down the group
Their reactivity decreases down the group as the more shells there are, the weaker forces of attraction there is from the nucleus to gain more electrons
They form negative 1 ions with metals
Their name changes to end in -ide, like chloride or fluoride
In displacement reactions, a more reactive element can displace another element that is less reactive
Cl2 + 2KBr Br2 + 2KCl - bromide is displaced by chlorine
The more reactive element will always displace the less reactive one
Group 0 - The noble gases
The noble gases exist as colourless gases
They are all unreactive and have a full outer shell
They exist as singular atoms
They are non-flammable
Their boiling points increase down the group
The transition metals
The transition metals are found in the middle of the periodic table (between groups 2 and 3)
Their properties in comparison to group 1
They both conduct electricity
They are both shiny
They have higher melting and boiling points
They have higher densities and are stronger and harder than the alkali metals
They have slow or non-existent reactions with oxygen, water and the halogens
They react vigorously with group 1
They make good catalysts
Energy changes
Energy transfers during exothermic and endothermic reactions
Different chemicals store different amounts of energy in their bonds
CH4 + 2O2 CO2 + 2H20
Before this reaction, the bonds have more energy, and after energy has been released to the surroundings, through heat
Exothermic reactions release energy to the surroundings
Energy is EXiting the reaction
The surroundings get hotter
Transfers energy to surroundings
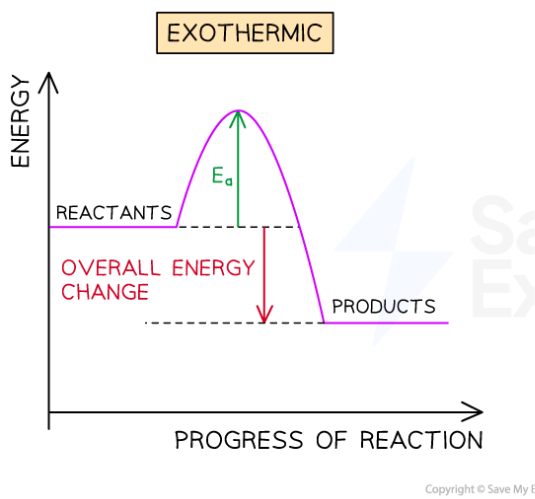
Endothermic reactions take in energy from the surroundings
Energy is ENtering the reaction
The surroundings get cooler
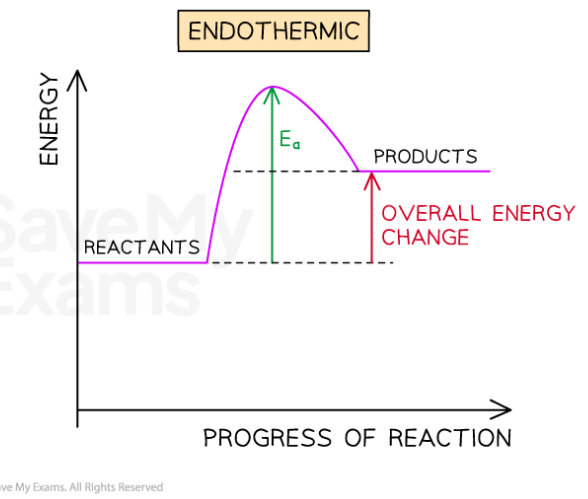
Activation energy is the minimum energy the reactant particles need in order to collide with each other enough to cause a reaction
Energy changes of reactions
Bond energy is the amount of energy required to break one mole of a particular bond
For example, a H-Cl bond requires 431 kJ/mol (energy needed to break one mole - 6.02 × 1023 - of these bonds)
Bond breaking is exothermic, and making bonds is endothermic
To calculate:
Work out how many bonds are breaking in the reactants, and calculate the total energy required by multiplying the number of specific bonds by the kJ required
Work out the same for the products
Find the difference in the total energy required for the reactants and the products 🙂
Cells and batteries
Electrochemical cells use chemical reactions to produce electricity
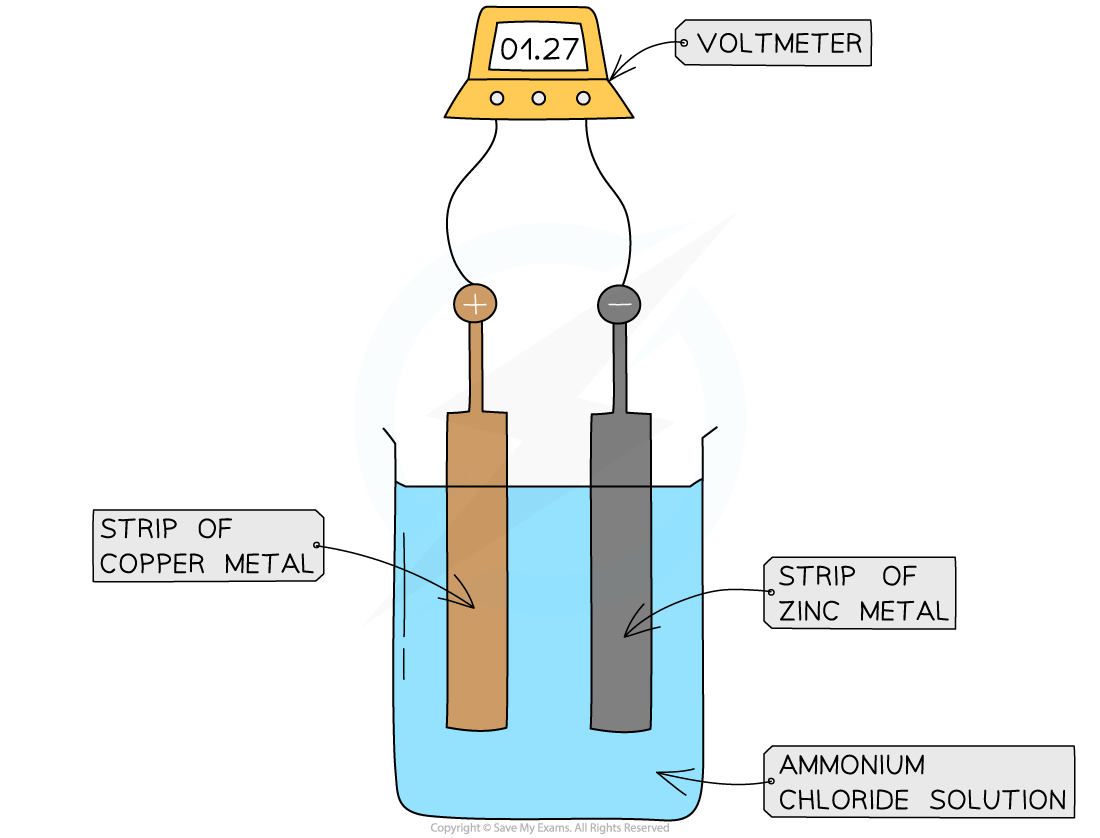
The electrodes have to be made out of two different types of metal, and they conduct
A cell can be made by connecting two different metal electrodes with wire and placing them in contact with an electrolyte solution
An electrolyte is a liquid through which charged particles can flow - creates a flow of charge, and therefore a cell
Batteries are similar, but consist of two or more cells connected in series to provide a greater voltage
Factors that affect the voltage of a cell or battery include:
The metals used - the greater the difference in their reactivities, the greater the voltage
The type and concentration of electrolyte
The conditions, such as temperature
Rechargeable batteries work as the chemical reactions inside can be reversed when an external electrical current is supplied
They are used in phones and laptops
Non-rechargeable batteries are where the reactions stop once one of the reactants has been used up
Used in smoke alarms, TV remotes, etc.
Also called Alkaline batteries
Fuel cells
Fuels cells are electrochemical cells that converts energy between chemical and electrical
We can convert the energy of oxygen and a fuel to release electrical energy we can use
The most common type of fuel cell is the hydrogen-oxygen fuel cell
It forms water and creates lots of electrical energy
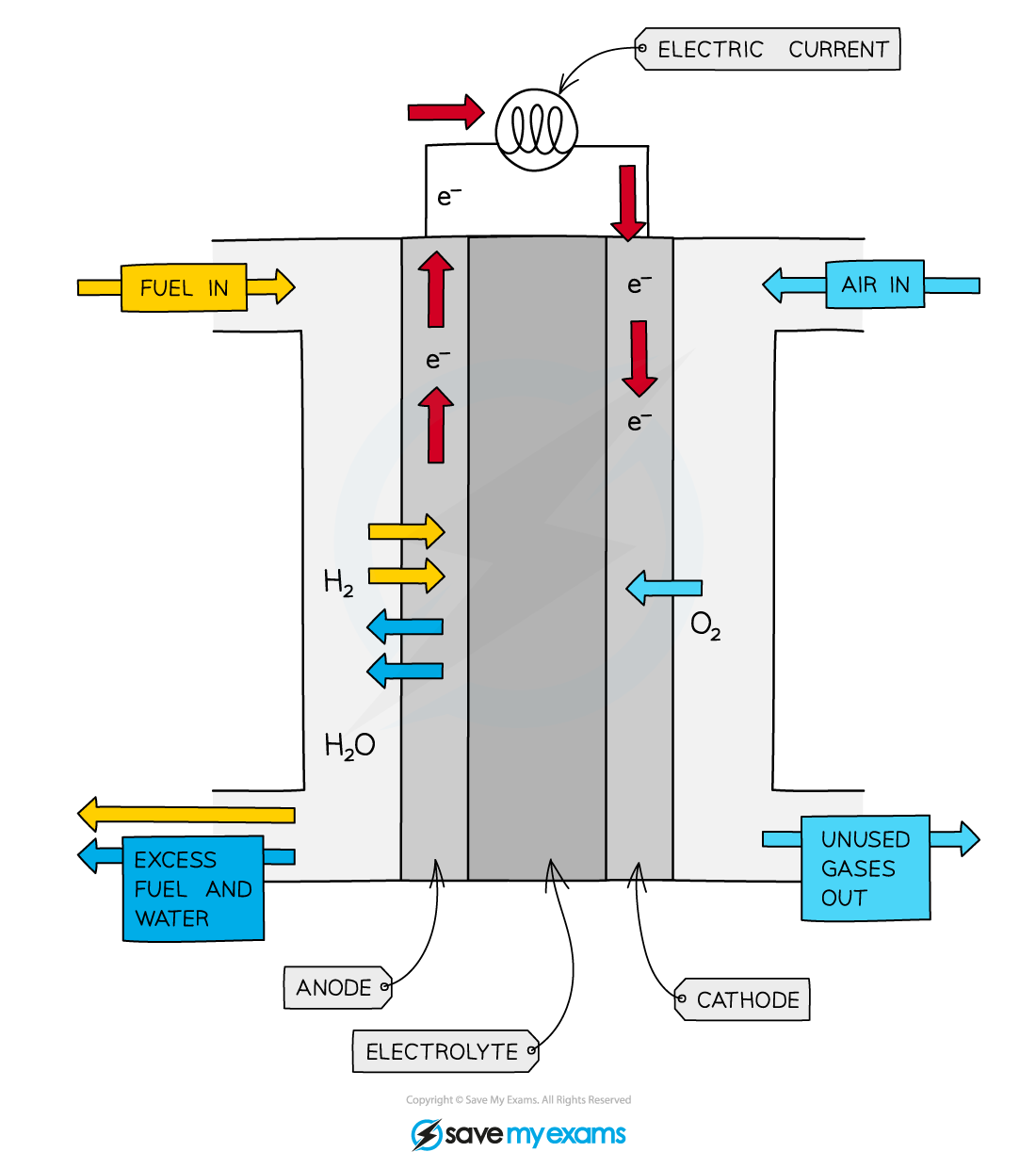
H2 enters through the left of the fuel cell, and is oxidised by the anode (-) to split into two positive hydrogen ions, and two electrons
The oxidisation of hydrogen - H2 2H+ + 2e-
The electrons pass through the wire, creating a current and electrical energy, to the cathode
The hydrogen ions pass through the electrolyte to the cathode (+)
Oxygen enters from the right side
Hydrogen ions and electrons can react with oxygen to produce water
Two H2 and one O2
O2 + 4H+ + 4e- 2H2O
The water leaves the fuel cell via the outlet, as well as heat (non-useful energy)
The overall equation of this process is O2 + 2H2 2H2O
The electrical energy comes from the flow of electrons through the wire
As the fuel enters the cell, it becomes oxidised, creating a potential difference across the cell
PROS - Only requires oxygen and hydrogen
No waste is created
They last longer than batteries
Simple process
CONS - H2 is expensive to store as it takes up lots of space
H2 is explosive in air ☹
To make hydrogen fuel, we need energy, often from fossil fuels
Bonding, structure and the states of matter
Ionic bonding
An ion is a charged particle, which is formed when elements gain or lose electrons to gain a full outer shell
Elements are more likely to form ions if there are not many electrons lost or gained
Takes less energy
Groups 1,2,6 and 7 are most likely
Groups 3,4 and 5 are rarely seen as ions
Happens between a metal and non-metal
For example, NaNa+ + e- and Cl + e- Cl-
Electrons are transferred
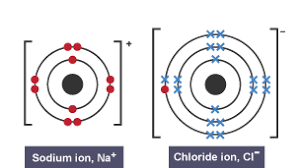
NaCl becomes an ionic compound and are attracted by strong electrostatic forces (have opposite charges)
Ionic compounds
Metals and non-metals form ionic compounds with strong electrostatic forces and ionic bonds
Usually group in large numbers
Regular lattice structures (3D)
Each ion is attracted to all those around it
Properties - very high melting points as lots of energy is required to overcome strong ionic bonds
can conduct electricity when molten or aqueous as charged particles are free to move
NEED TO KNOW - Hydroxide = OH-
Sulphate = SO4-
Nitrate = NO3-
Carbonate = CO3-
Ammonium = NH4+
Covalent bonding
The sharing of electrons in the outer shell between non-metals to gain a full outer shell
Cl - Cl
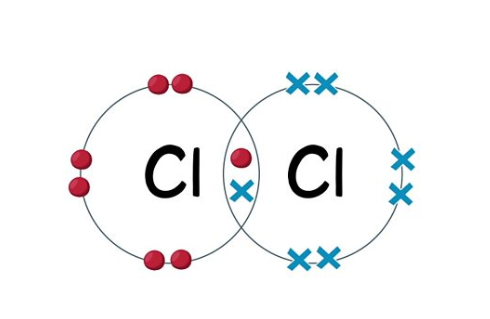
They can become:
Simple molecular substances - Small molecules with strong covalent bonds between atoms and weak intermolecular forces between molecules
Like water, ammonia, chlorine or methane
Polymers - Long chains of repeating units (monomers)
Giant covalent structures - diamond, graphite + silica dioxide
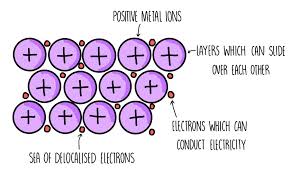
Metallic bonding
Happens between metal atoms
Solid metals are in a giant structure arranged in a regular pattern with delocalised electrons
They give up their outer shell electrons and share them with the other metals
The atoms all become + ions
The lost electrons can freely move so are delocalised
There are strong forces of attraction between the ions and electrons, which hold everything together in a regular structure
Metals are strong, so have high melting and boiling points
They are good conductors of heat and electricity ⚡
Their delocalised electrons can feely move and carry electrical current through the structure
Metals are malleable - their regular structure allows layers to slide
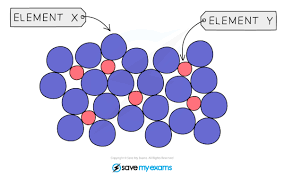
Alloys are when 2 or more different metals or a metal and non-metal form metallic bonds, with different sized atoms
This disrupts the regular structure, so layers can no longer slide
Alloys are stronger than pure metals
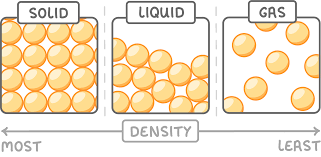
States of matter
Solids - Strong forces of attraction (holds them close together)
Fixed, regular position
Definite shape and volume - can vibrate
Liquids - Weak forces of attraction (particles are free to move and flow)
Can flow
Compact and definite volume, but not shape
Move to fit a container
Gas - Very very weak forces of attraction
No definite shape of volume
Fill a container
Particles are free to move
Constantly moving with random motion - move in a straight line and are deflected when hit
State symbols
Solid - s
Liquid - l
Gas - g
Aqueous - aq
Properties of ionic compounds
High melting and boiling point
Lots of energy is required to overcome strong electrostatic forces of attraction, and there are lots of forces
Can conduct when aqueous or molten
Charged particles are free to flow through the structure
Properties of small molecules
Low melting and boiling points
Weak intermolecular forces between molecules, that need little energy to break
COVALENT BONDS ARE NOT BROKEN
Generally liquids or gases at room temperature
Do not conduct
Properties of metals and alloys
Metals are soft and malleable, shiny, good conductors of heat and electricity and have high melting and boiling points
Alloys are hard (no layers), have high melting and boiling points and are good conductors
Giant covalent structures
Simple molecular substances have low melting points, strong bonds between atoms and don’t conduct
Giant covalent structures have huge numbers of non-metal atoms
Arranged in a regular repeating lattices
Have high melting and boiling points as there are a lot of covalent bonds
Very strong - lots of bonds
Generally don’t conduct (apart from graphite and graphene)
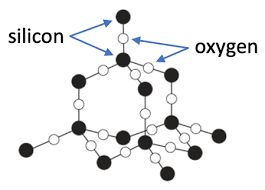
Silica dioxide is made of silicon and oxygen in a ration of 1:2
Makes up sand 🏖
Diamond and graphite
Allotropes of carbon
Diamond is a giant covalent structure💎
Is very strong
Each carbon is bonded to 4 other carbons (max amount)
It doesn’t conduct as there are no delocalised electrons
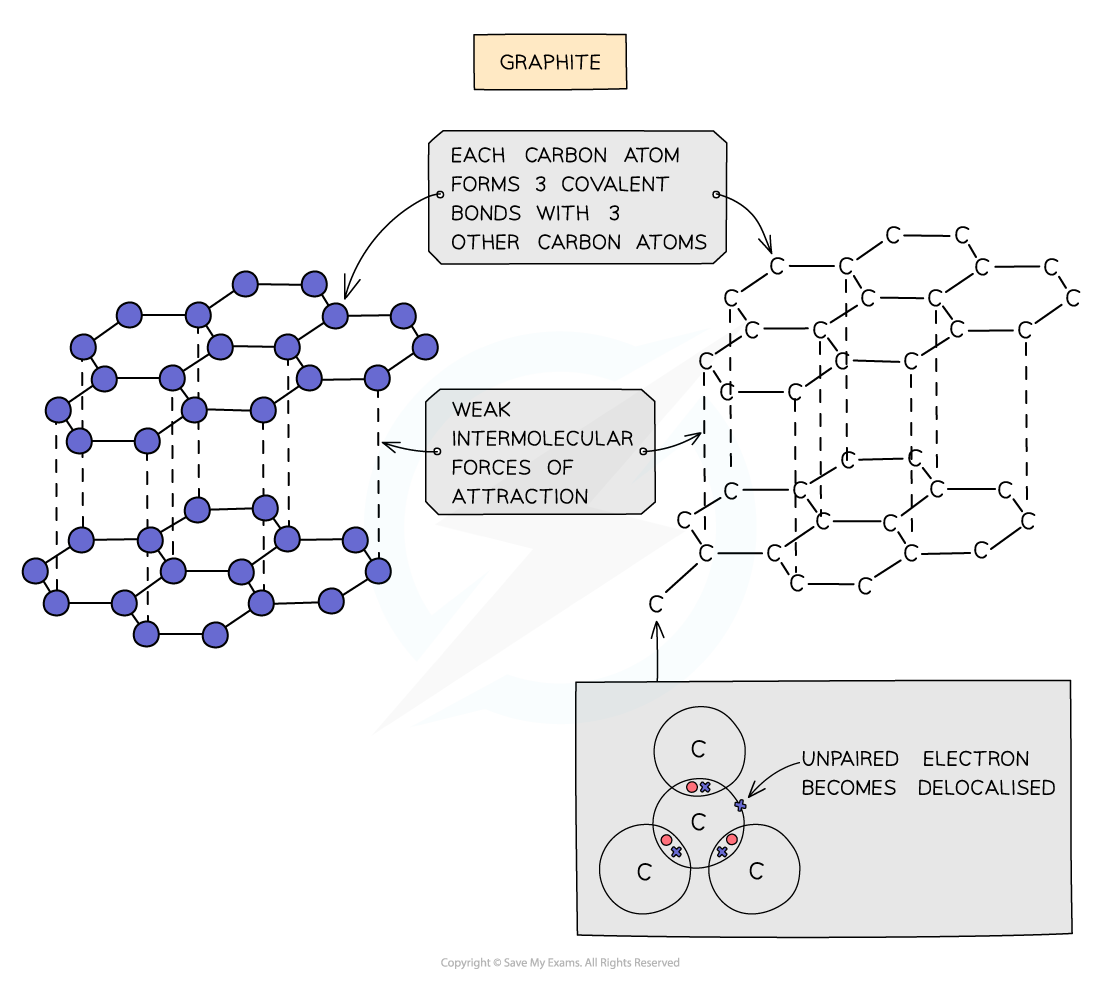
Graphite is a giant covalent structure
Is very strong
Each carbon makes 3 out of 4 covalent bonds possible
Is arranged in layers with weak intermolecular forces between them
This allows the layers to slide over one another and makes it soft
Has a high melting and boiling point
Can conduct electricity - only ¾ bonds are made, so there are left over electrons
Become delocalised (one per carbon atom) and are free to move through the structure and carry charge
Graphene is a single layer of graphite
Graphene and fullerenes
Are allotropes of carbon
Graphene is a single layer of graphite and can conduct electricity as there are delocalise electrons
Useful in electronics (conducts and is small)
Fullerenes are tubes and spheres made out of a single sheet of graphite
Spheres can be used to surround molecules (like drugs) and used to deliver to specific areas of the body
They have a large surface area : volume ratio, so make good industrial catalysts
Tubes can be used in nanotechnology as conductors, to strengthen tennis rackets (adds strength without weight as high length : diameter)
Buckminster fullerene is a hollow sphere that is made of 60 carbon atoms and is used for drug delivery
Using tiny structures is called nanotechnology
Medicine, fashion, batteries and food
Nanoparticles
Nanoparticles are really really really tiny particles - 1nm - 100nm (0.00000001m)
Nanoscience is produces new nanoparticle materials
They have a large surface area : volume ratio
Good for catalysts as surface area increases its efficiency
Nanomedicine uses fullerenes to deliver drugs around the body
Electrical circuits use them to make tiny computer chips as some can conduct
Silver nanoparticles have antibacterial properties so can be infused into wound dressings and masks
Issues with nanoparticles
They are relatively new so we are not aware of all risks (long term)
For example, sun cream with nanoparticles allows for better skin coverage but we are unaware if it can enter our body through the skin and potential damage cells
They are also possibly damaging to the environment
Sizes of particles and their properties
Atoms and small molecules - 0.1 nm
Nanoparticles - 1 to 100nm
Fine particles - 100 to 2500 nm
Coarse particles - 2500 to 10000 nm
The smaller the particle, the higher the surface area : volume ratio, so increased reactivity
Polymers
Polymers have very large molecules and their atoms are joined by strong covalent bonds in long chains
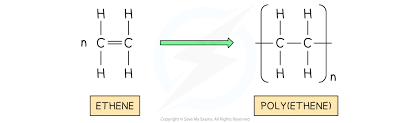
Solid at room temperature
Higher boiling points than strong intermolecular forces and lots of bonds to overcome