3.2.5 Transition metals
The D block
Most elements are transition metals - focus on period 4 (Ti - Cu)
What is a transition metal?
- Metal which can form one or more stable ions with an incomplete d sub level.
- d sub level contains up to 10 electrons
- So forms ions that has 1-9 electrons in d sub level
Rules in filling the sub levels
Electrons first fill up from lowest energy level
Electrons fill orbitals singly before pairing
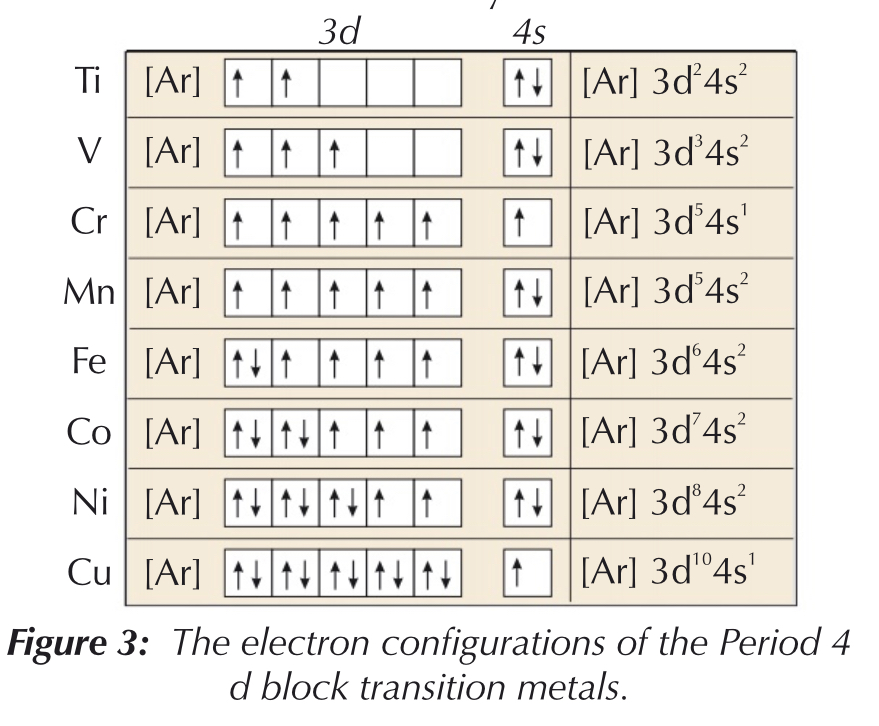
Transition metals follow the same rule - however…
- Cr - prefers to have one electron in each 3d orbital, with 1 electron on 4s orbital
- Cu - prefers to have full 3d orbital, with 1 electron on 4s orbital
Forming a positive transition metal ion
- electrons on ==4s are removed first==, then the 3d
Why are scandium and zinc not transition metals?
- their stable ions don’t have incomplete d sub levels
- Both only form one ion, these ions both don’t have incomplete sub levels, Sc ion has no d sub level, and Zn ion has full d sub level
What are the physical and chemical properties of transition meals?
- ==Physical:==
- High density
- High melting and boiling points
- ==Chemical:==
- Form complex ions
- Form coloured ions
- Good catalysts
- Exist in variable oxidation states
Incomplete d sub level causes these properties
Why do elements show variable oxidation states?
Energies of the 4s and 3d sub levels are similar - different number of electrons can be gained or lost using similar amounts of energy.
What is a coordination number? What is this dependent on?
- Number of lone pairs a cation can accept
- Depends on size and electronic configuration
- Depends on size and charge of the ligand
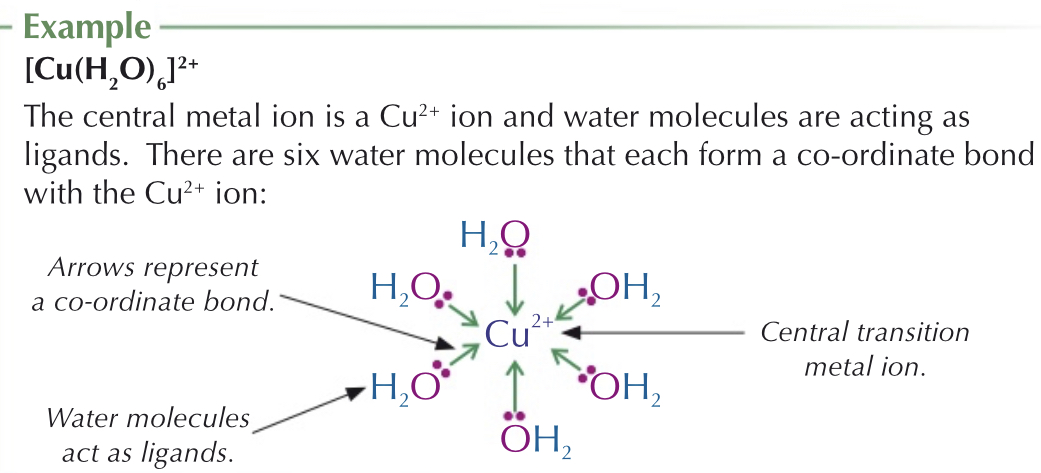
What are complex ions?
Surrounded by ligands - bonded by coordinate bonds
Coordinate bond - one atom provides both electrons in bond
The ligand provides the pair of electrons.
Lewis Acid and Base
- Metal ion acts as a Lewis acid (accepts electrons)
- Ligand acts as a Lewis base (donates electrons)
What is a ligand? Are there different types?
Must have at least one lone pair
Different ligands have different number of lone pairs - so different number of dative bonds
Monodentate: Ligands which only form one dative bond
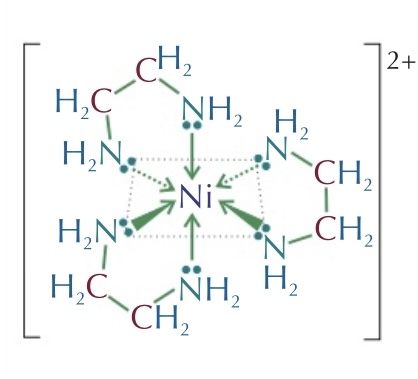
Bidentate: form 2 dative bonds
Multidentate: form multiple dative bonds (extremely stable, reaction results in an increase in entropy
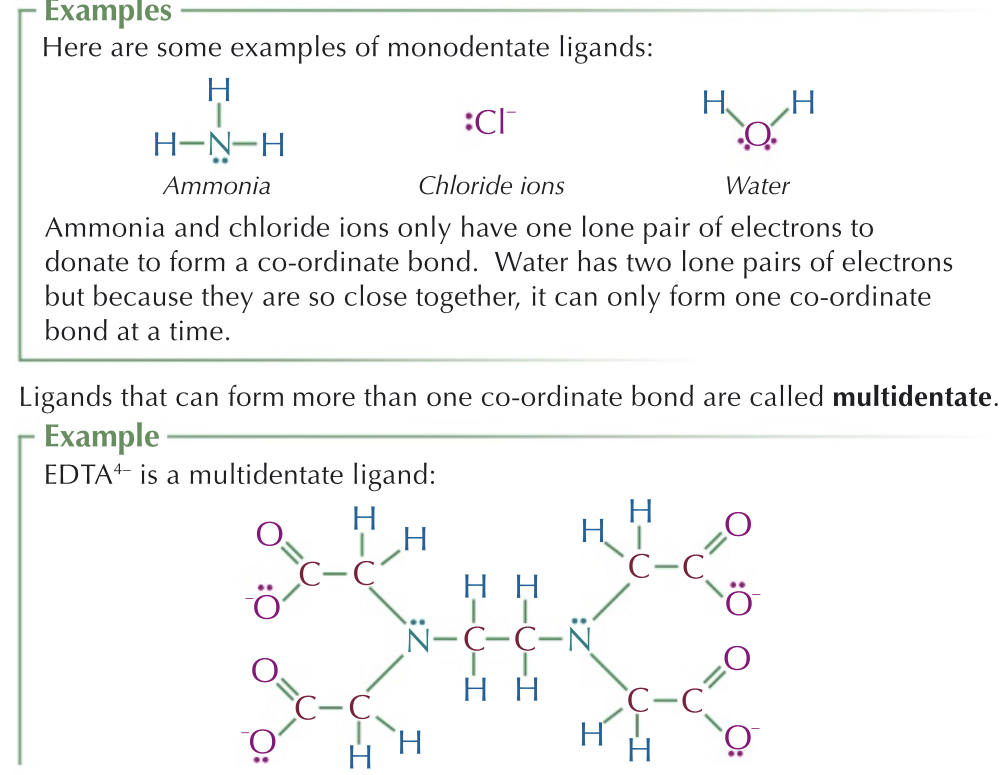
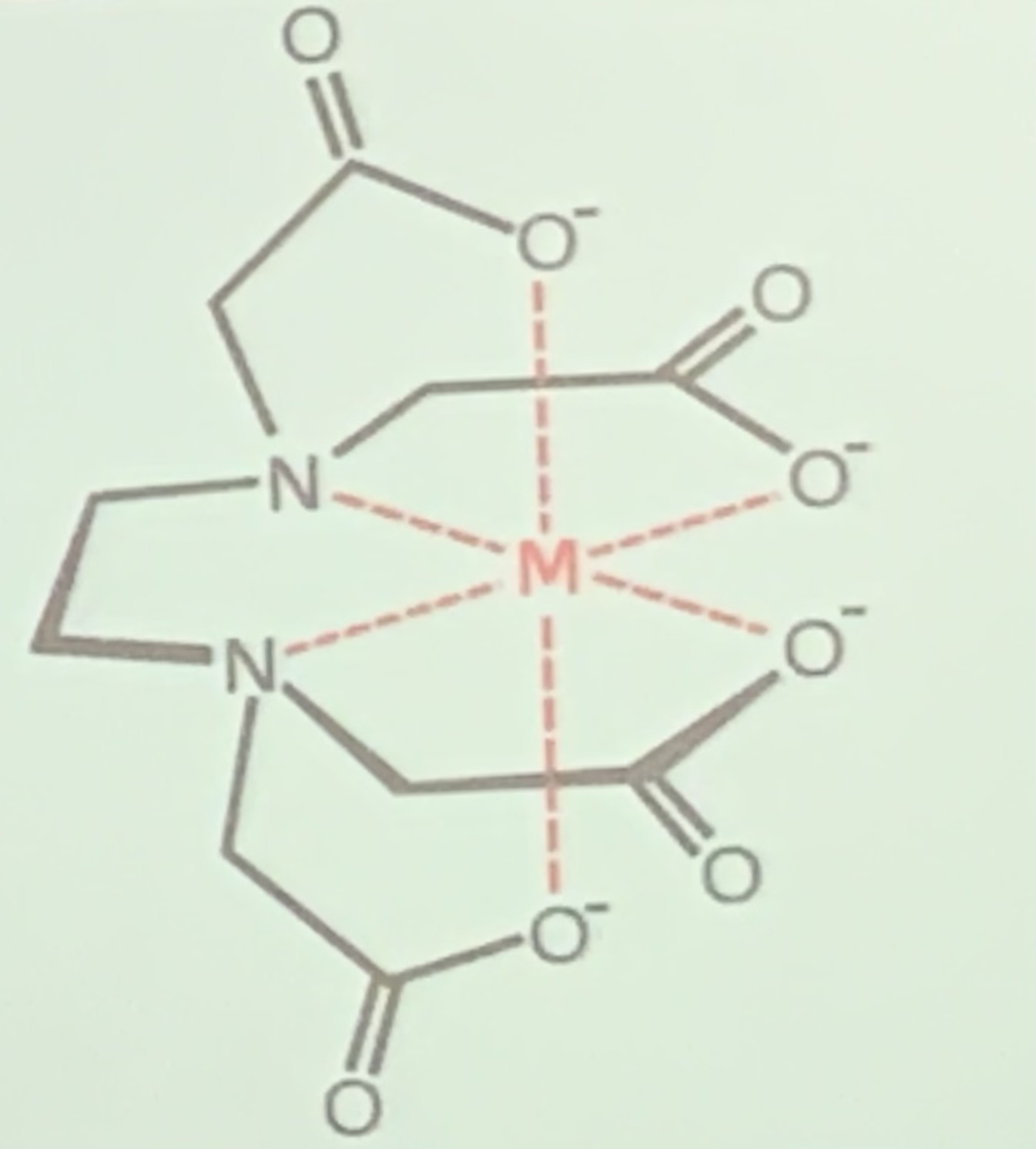
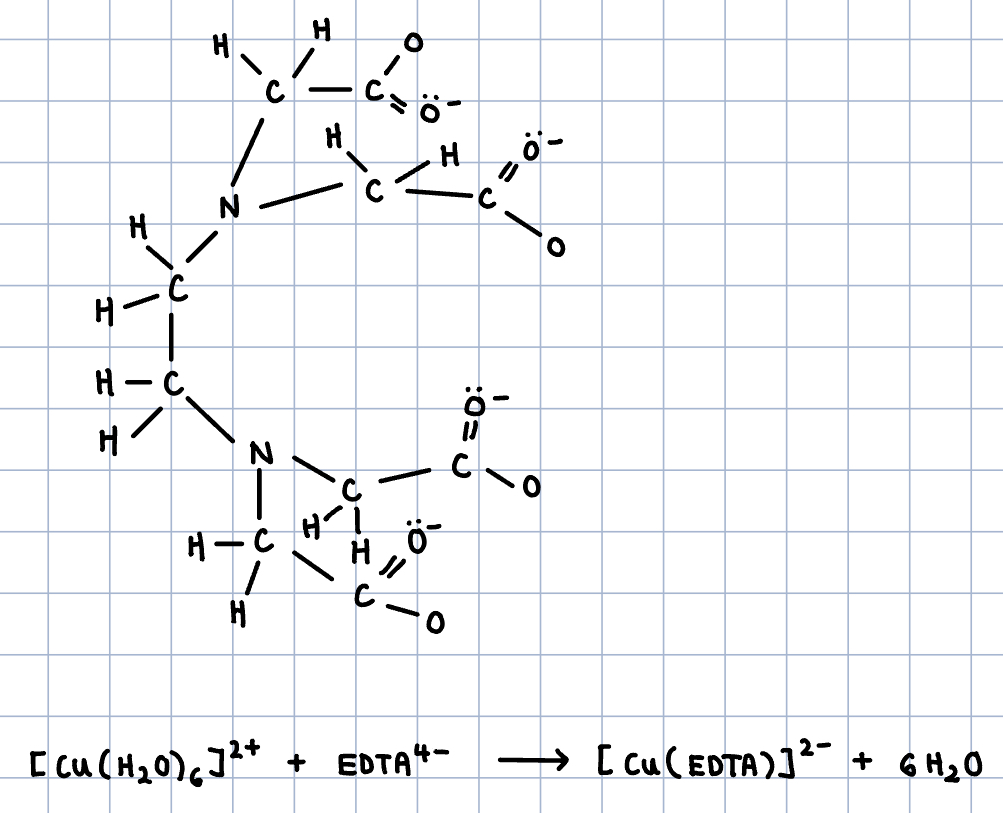
EDTA4- has six lone pairs (2 on nitrogen atoms, 4 on oxygen atoms) - form 6 coordinate bonds with metal ion.
Complex ions with multidentate ligands are called chelates
Looking at the reaction equation:
- This can be used to remove metal ions from solution
- Six water ligands are replaced by one EDTA ligand
==LIGAND: Ethane -1,2 - diamine==
Oxidation states of metals in complex ions - equation

Charge on the ligand and the oxidation state of metal determines overall charge of the complex.
- ==Neutral ligands:== results in positively charged complexes (overall charge of ligand = oxidation state of ligand)
- ==Negatively charged ligands:== result in negatively charged complex
How are shapes of complex ions determined?
Depends on coordination number - number of coordinate bonds
Depends on size of ligand…
Usually small molecules like water can fit 6, bigger molecules like chlorine can only fit 4.
Water ligands form aqua complexes
Ions which has 6 ligands bonded - octahedral shape
4 ligands bonded - tetrahedral shape / square planar
2 ligands bonded - linear
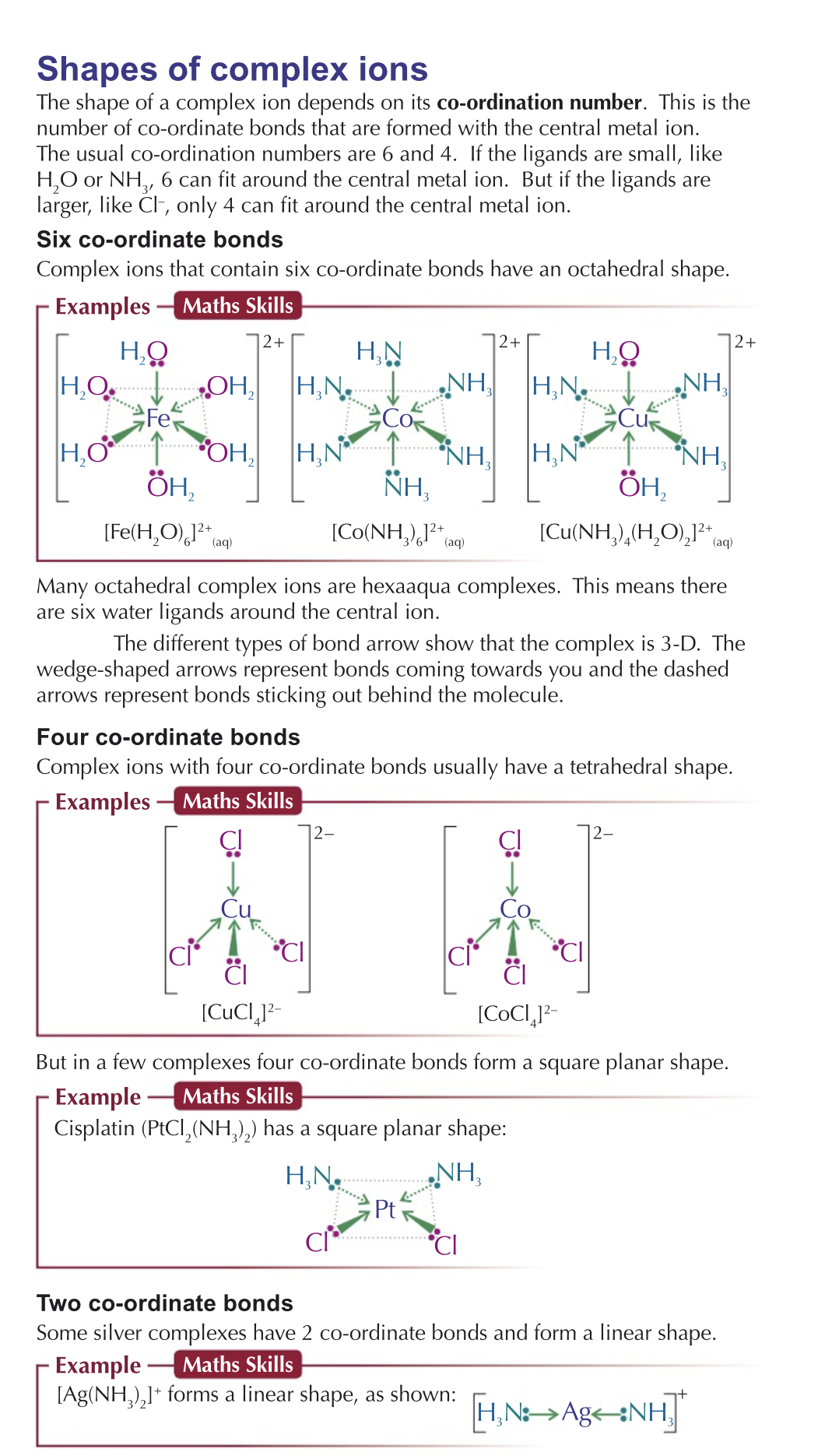
Notation
Charge is written outside square brackets, ligands are written with brackets around it.
How do we name compounds?
| Ligand | Name in complex | Formula | Charge |
|---|---|---|---|
| Water | Aqua | H2O | 0 |
| Ammonia | Amine | NH3 | 0 |
| Cyanide | Cyano | CN- | -1 |
| Hydroxide | Hydroxo | OH- | -1 |
| Chloride | Chloro | Cl- | -1 |
| Number of ligands | Prefix |
|---|---|
| 2 | Di |
| 4 | Tetra |
| 6 | Hexa |
When complex is an ionic, metal ends in “ate”
| Metal | Name of anion |
|---|---|
| Cobalt | Cobaltate |
| Chromium | Chromate |
| Vanadium | Vanadate |
| Copper | Cuperate |
| Iron | Ferrate |
| Silver | Argentate |
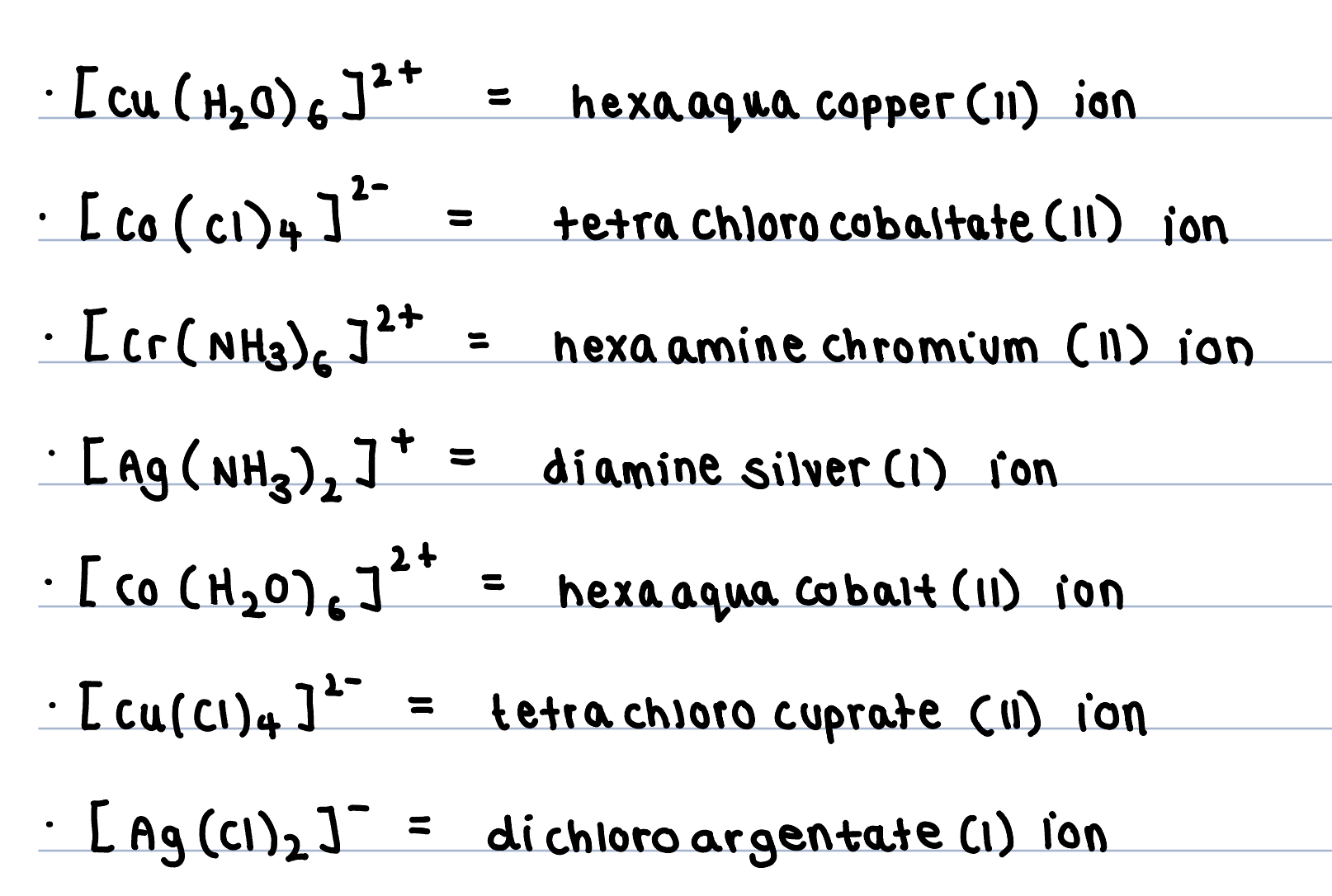
Which complex ions show optical isomerism?
Octahedral complex ions - when three bidentate ligands attach to central metal ion.
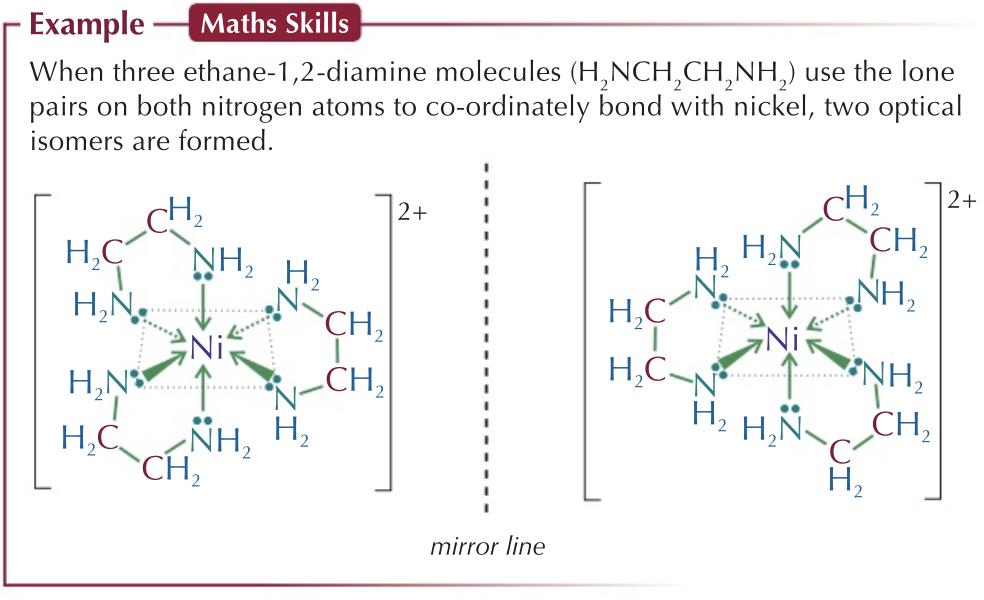
Which complex ions show cis-trans isomerism?
Square planar complex ions - 2 pairs of ligands
Two paired ligands are directly opposite - trans
Same side - cis
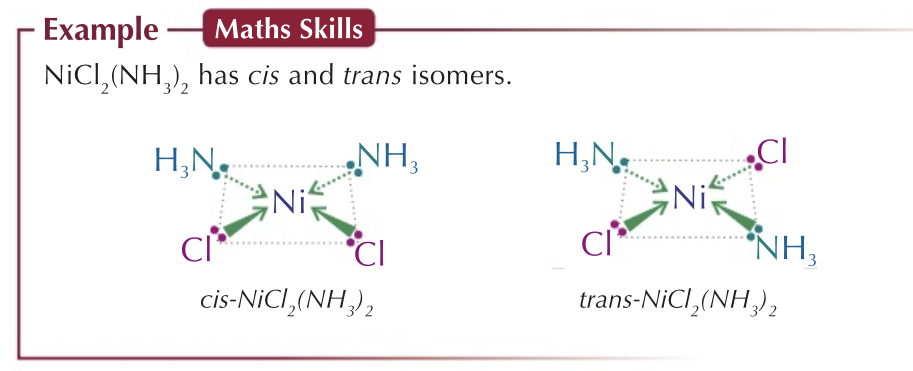
Octahedral complexes can also show cis-trans isomerism:
Where there is ==four ligands== of one type and ^^two ligands^^ of another type.
==Coloured ions:==
How are the 3d orbitals split by the binding of the ligand?
3d orbitals of the transition metal ions have the same energy at first.
When ligand binds to the metal ion - some orbitals are given more energy than others.
This splits the 3d orbital into 2 different energy levels
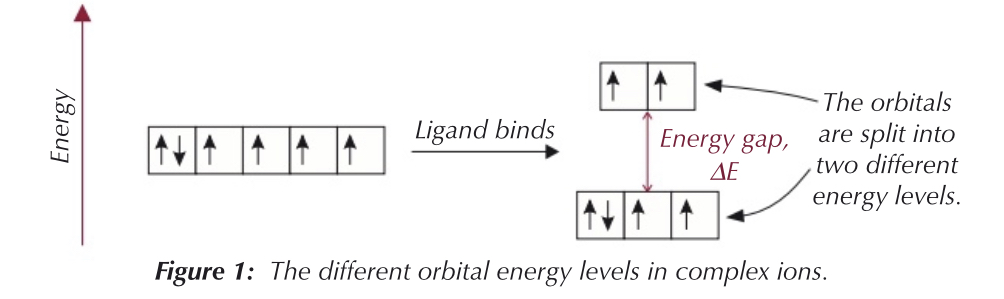
How do we calculate the Energy gap?
Electrons will tend to occupy the ground state (lower orbitals), when light is shined on it, the ground state electrons jump to the excited state - the energy needed for this is equal to the energy gap between the ground state and excited state. They get this energy from visible light.
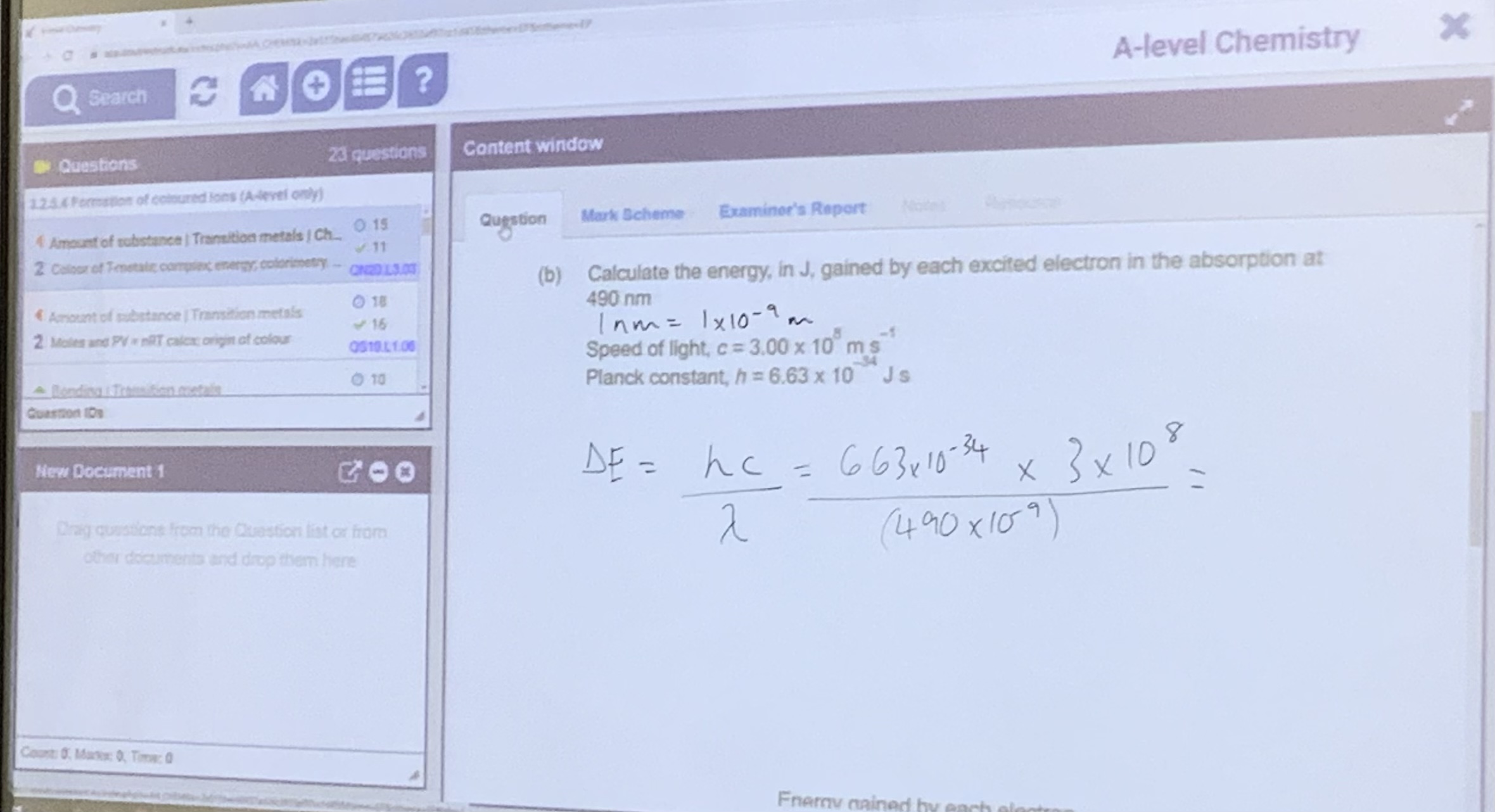
Energy needed for electrons to jump can be worked out using the equation:

Calculation examples:
What affects the size of the energy gap? What does this mean about the colour of the complex?
- Central metal ion
- It’s oxidation state
- Ligands attached
- Coordination number
Changes to the size of the energy gap will also change the colour of a complex
How do we see the colour of the transition metals when in solution?
- Light is emitted on transition metal ion, some wavelengths are absorbed, causes electrons to jump to higher orbitals.
- The wavelengths absorbed depends on size of energy gap.
- White light contain all wavelengths of light, the colours which are not absorbed are seen, colours which are absorbed by the solution are not.
Why do only transition metals with partially filled d orbitals display colour?
- Incomplete sub levels mean that electrons are able to jump to a higher energy level - absorb light
- Those with complete sub levels mean there are no electrons to jump - no light absorbed, all light reflected.
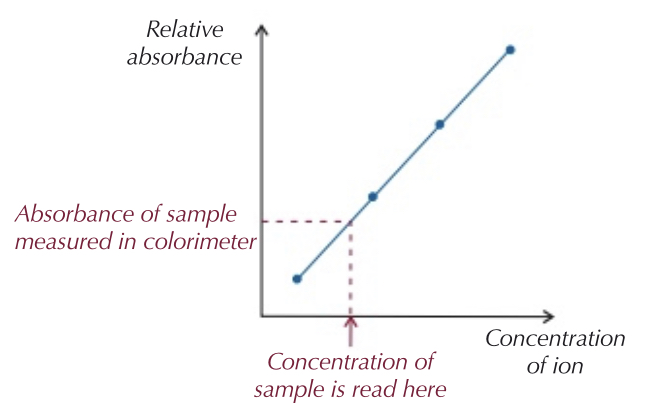
How can spectroscopy measure the absorbance of a solution?
White light is shone through filter
Filter is chosen to only let colour of light that is absorbed by sample
Light passes through sample into colorimeter
This measures absorbance of light
More concentrated a solution, the more absorbance of light
Your results can be plotted on a graph, able to work out concentration using absorbance value.
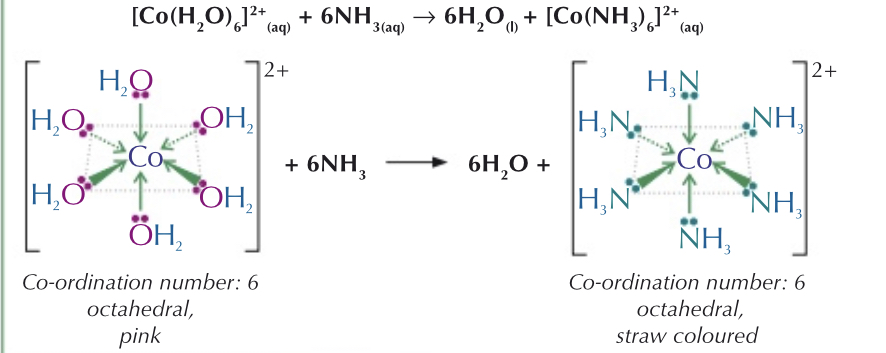
Ligand substitution reactions:
What is ligand substitution?
- One ligand is substituted for another
- Always causes a colour change
What occurs when a similar sized ligand is substituted?
Coordination number of complex would not change, this means that the shape of the complex wont change either. However, there will still be a colour change as a different ligand is present.
What occurs when a different sized ligand is substituted?
There will be a change in coordination number, and therefore shape. There is also a colour change.
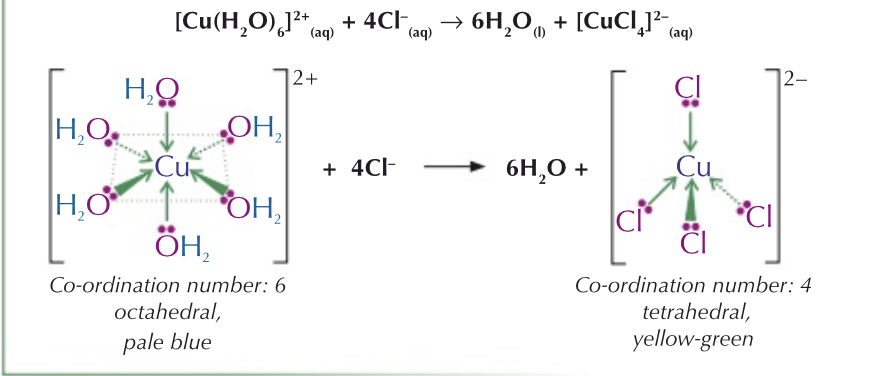
What is partial substitution of ligands?
When not all of the ligands are substituted. For example only 4 out of the 6 water ligands may be substituted by NH3 ligand.

What is the structure of haemoglobin?
- Has 4 haem groups
- In the haem groups, there are Fe2+ ions
- The Fe2+ ion is coordinately bonded to 6 ligands.
- 1 ligand is multidenate, called porphyrin. Contain 4 nitrogen atoms which form circle around Fe2+. 4 coordinate bonds
- The other 2 coordinate bonds are formed from either water or oxygen ligand and globin protein.
How does ligand substitution takes place in haemoglobin as it moves around the body?
- Both water and oxygen bind as ligands
- When there is a high concentration of oxygen at the lungs, the water ligand is substituted by an oxygen ligand (form oxyhaemoglobin)
- At respiring cells, when oxygen is delivered, water ligand is substituted back. (Deoxyhaemoglobin)
How does carbon monoxide affect the substitution of water and oxygen ligands to Fe2+?
- Haemoglobin can substitute its water ligand with carbon monoxide ligands - form carboxyhaemoglobin.
- Bad - form very strong bond with Fe2+ - doesn’t readily exchange with water or oxygen. So cant transport oxygen
- Cause headaches, dizziness, unconsciousness and death.
How does the stability of a complex ion affect its ability to reverse to the old version?
- ligand exchange reactions are easily reversed, unless the new complex ion is much more stable compared to the old version.
- This means stronger bond formation = harder reversal.
- Monodentate ligands are easy to reverse, multidente and bidentate ligands are harder to reverse.
Why is the enthalpy change for a ligand substitution very small?
Strength of bonds broken is very similar to strength of new bonds being made. Difference/change between bonds formed and broken is small.
How does entropy change affect the stability of complexes?
When Monodentate ligands are substituted with bidentate or multidentate ligands, the number of particles increase
Reactions that result in an increase of entropy are more likely to occur
Known as Chelate effect.


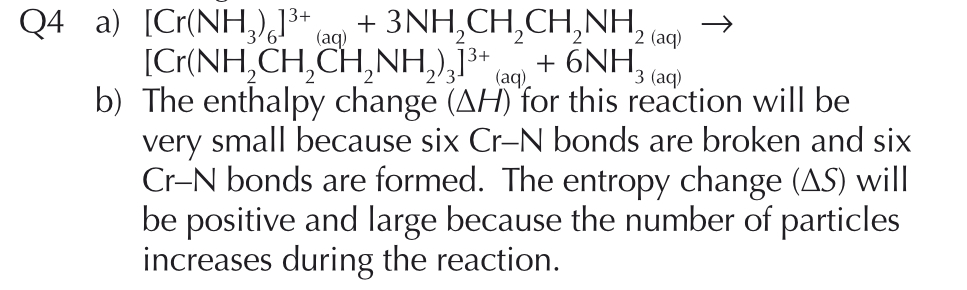
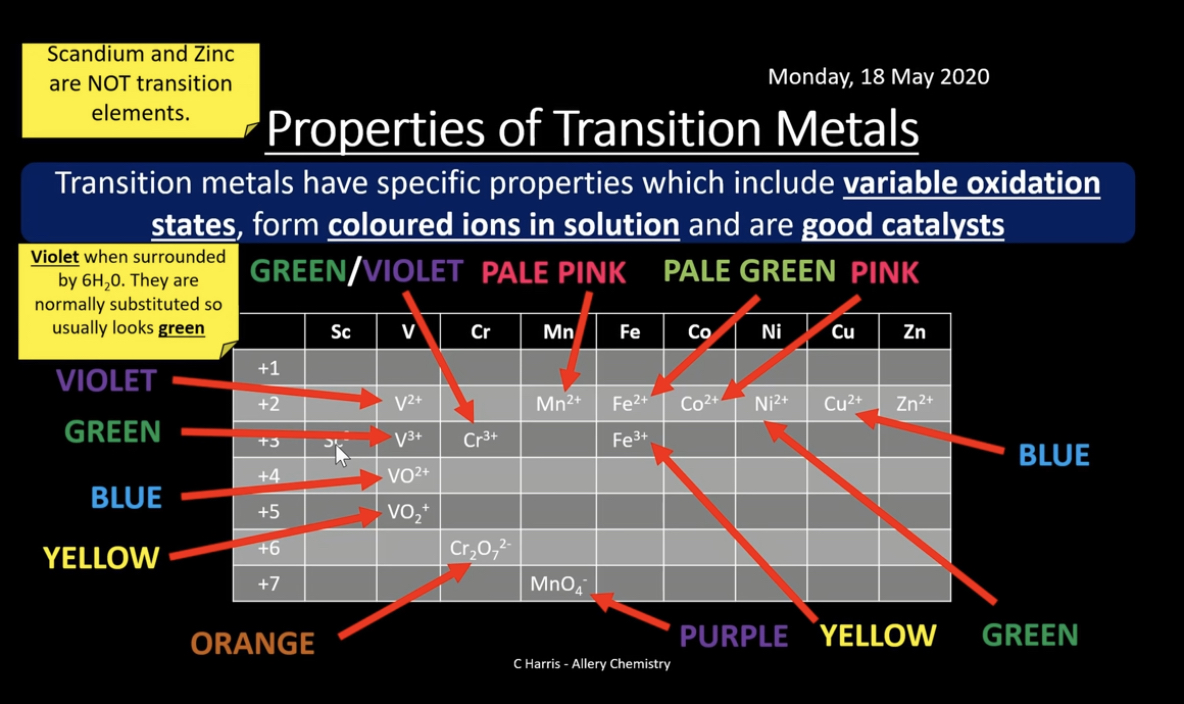
Variable Oxidation States:
What are the colours of the different oxidation states of vanadium? How can we achieve the different oxidation states of vanadium?
- You - yellow (5+)
- Better - blue (4+)
- Get - green (3+)
- Vanadium - violet (2+)
Vanadium (V) can be reduced by using an acidic solution…

What are redox potentials?
- Tells you how easily its reduced to a lower oxidation state (similar to electrode potentials)
- More positive redox potential - less stable so more likely to be reduced
Redox potentials are standard electrode potentials - so measured under standard conditions (1 moldm-3) as it is measured under standard hydrogen electrode. This means redox potential of ion won’t be the same as standard electrode potential, varies depending on environment of ion.
How do ligands affect redox potential?
- standard electrode potentials are measured in aqueous solutions, so any ions will be surrounded by water ligands
- Different ligands will affect redox potentials differently
How does pH affect redox potential?
- Some ions need H+ ions to be reduced
- Some release OH- ions into solution when reduced
- Redox potential is more positive in acidic solutions - as ion is more easily reduced.
How is Tollen’s reagent made and used?
Tollen’s reagent is made by adding ammonia solution to silver nitrate solution - form colourless solution —> [Ag(NH3)2]+
Reduction of [Ag(NH3)2]+ give silver mirror in aldehydes
Aldehyde is oxidised to carboxylate anion, Ag+ ions are reduced to silver metal (Ag)
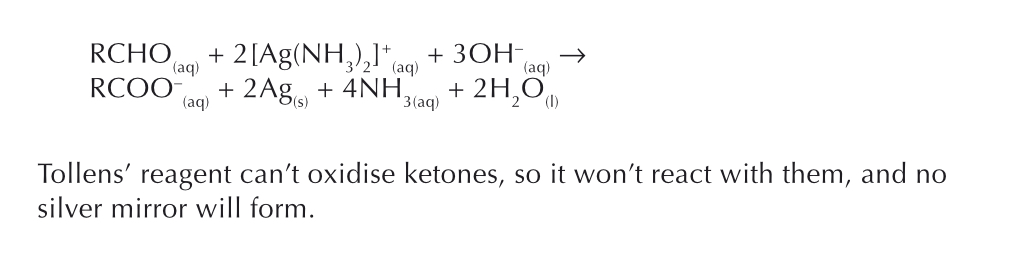
Titration redox calculations:
How do you perform titration with reducing and oxidising agents?
- Transition metals have variable oxidation states - so present in either oxidising or reducing agent.
- Colour change of reaction makes it easy to spot end point of titration.
- Measure reducing agent (e.g. Fe2+, C2O4 2-), add this in conical flask with excess dilute sulphuric acid. (H+ ions allow oxidising agent to be reduced)
- Add oxidising agents (e.g. MnO4 -) using burette, swirling the conical flask
- Reaction will continue until all reducing agent is used up
- Now one drop of oxidising agent after this will produce a colour change.
- Repeat, first titration is rough.
Which 2 titration equations must you know? How can you explain the colour change of these titrations?

When potassium manganate (VII) is used as the oxidising agent, it produces Mn2+ ions, this has a very pale pink colour, it is practically colourless, the real colour change is seen from the dark purple potassium manganate.
How can we combine the redox equations of potassium manganate (VII) and iron (II)?
- Remember that the iron (II) is the reducing agent, and potassium manganate (VII) is the oxidising agent.
Write the oxidation equation and reduction equations - balance electrons after adding water and H+
Now add electrons to balance out charges
Equal the electrons, combine the 2 equations.

Examples of calculation exam questions that may come up:


Transition metal catalysts
Why do transition metals make good catalysts?
- Can change oxidation states by gaining or losing electrons in their d orbital. So can transfer electrons to speed up reactions.
- Variable oxidation states, incomplete d orbital
What is the contact process?
- Industrially make sulphuric acid
- Vanadium(V) oxide (V2O5) is the catalyst
- Vanadium oxide is reduced to vanadium(IV) oxide (V2O4), and it oxidises SO2 to SO3
- Vanadium(IV) oxide is oxidised back using oxygen
If vanadium didn’t have variable oxidation states, it wouldn’t be able to transfer electrons to SO2 to oxidise it, and it wouldn’t be able to be oxidised back into its original state (catalyst).
What is a heterogeneous catalyst?
Different phase from its reactants
What is the Harbour process? What transition metal catalyst is used?
Making ammonia industrially
Iron (s) is used as the catalyst - heterogeneous catalyst
Reactants are all gas state
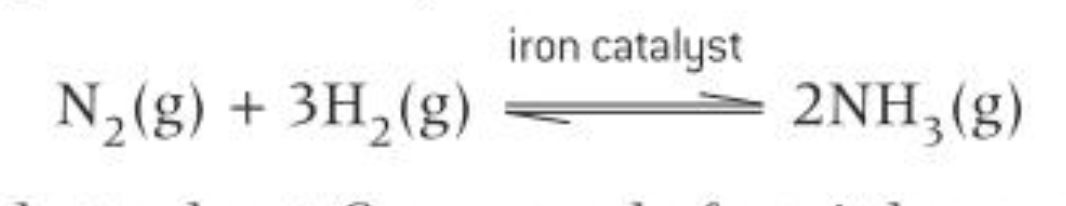
What heterogeneous catalyst is used for the contact process?
- Vanadium(V) oxide - solid
- Reactants are all gas state
How are heterogenous catalysts made to be more efficient and less expensive?
Increase surface area
Spread catalyst on inert support medium - increase surface area to mass ratio. (Image shows honey comb medium)Metal-Aqua Ions

Why do catalyst not last forever?
- catalyst may be covered in impurities overtime - “poisoning”
- Catalysts can become poisoned by impurities that block active sites, block reactants from being absorbed - reduce efficiency, must be replaced. This process is called catalyst poisoning.
- Finely divided catalyst may gradually be lost from support medium
- Reduces efficiency by reducing surface area, slowing down reaction.
- Less product is bring made at one time.
Examples which are quite interesting:
Catalytic converters in car engines:
- Cars which have a catalytic converter can’t use lead fuels as the lead coats the surface of the catalytic converter
- This means that unleaded fuel must be used
Iron catalyst in the Harber process:
- Hydrogen used is produced from methane.
- Methane is a natural gas - contains impurities
- Impurities include sulphur compounds - if not removed, it is absorbed into the iron catalyst and forms iron sulfide
- This stops iron from efficiently catalysing the reaction.
How can we reduce catalyst poisoning?
- Purifying reactants
- Removes impurities that may be absorbed and poison the catalyst.
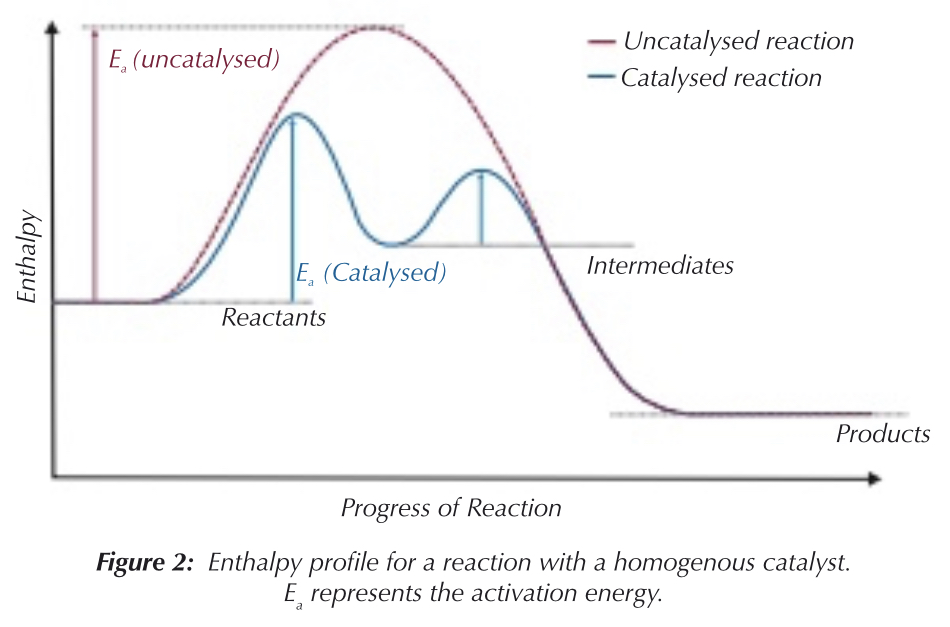
What is a homogenous catalyst? How does it work?
Catalyst in the same state as the reactants
Catalyst combines with reactant to form intermediate species
Intermediate species react to form products, and reform catalyst.
Explain the reaction between iodide ions and peroxodisulfate ions.
Slow reaction as both ions are negative
Fe2+ catalyst is added to speed up the reaction
Fe2+ is oxidised by S2O8 2-
Fe3+ then oxidises 2I-
This produces I2 and 2SO4 2-

Explain the reaction between MnO4 - and C2O4 2- ions
Mn 2+ is used as the homogenous catalyst
It’s an autocatalyst, the product is the catalyst
This means as the product increases, the reaction speeds up.
At the beginning of the reaction, there is no Mn2+ ions, so activation energy is very high. This is because negative ions are trying to collide with eachother.
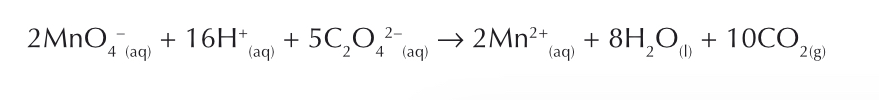
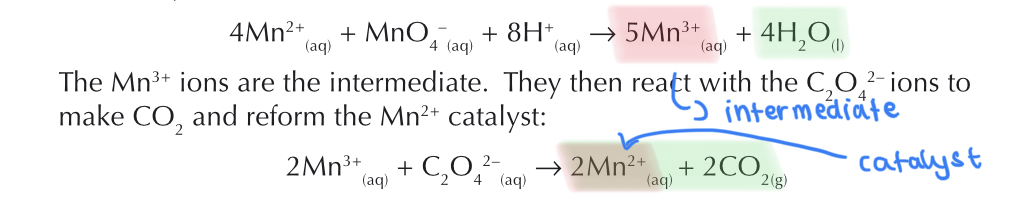
Metal aqua ions
What are metal-aqua ions?
- When metal ions dissolve in water, water molecules form coordinate bonds, forming metal-aqua complex ions.
- Metal aqua ions are acidic! The higher the ==charge==, the more ==acidic== it will be.
- Usually there will be 6 water ligands around the metal ion.
- Water ligand is neutral, this means overall charge will be the charge of the metal ion.
What does the hydrolysis of metal aqua 2+ ions produce?
When water is added to a metal aqua 2+ ion, a hydrolysis reaction occurs. This is where a hydrogen is broken/donated from one of the water ligands, producing a hydroxonium ion with the water. This creates a new complex where there are water molecules as well as one OH group. The OH group is negative, so overall charge of new complex is one less. ==2+ complex ions dissociate weakly, so are not very acidic.==

Why is the hydrolysis of metal 3+ aqua ions more acidic?
- Have higher charge density - larger charge, small size
- Example: if we have two ions with the same size - 2 Fe ions, and one has a 2+ charge, the other has a 3+ charge, the one with the higher will have a higher charge density.
- Higher charge density has an impact on how acidic they are….
- 3+ ions are more ==polarising== than 2+ ions, this means they are able to ==disrupt the O-H bond== (in one of the H2O), so they attract the electrons towards the metal ion - weakening the bond. This means the bond is more likely going to lose a hydrogen (proton) - so more acidic.
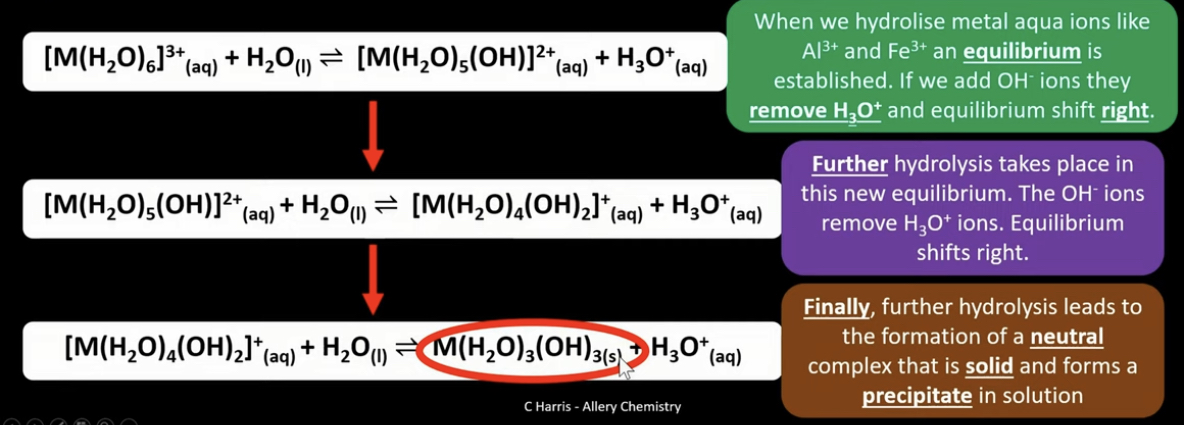
How do we produce an insoluble hydroxide through further hydrolysis?
Use OH- ions to produce insoluble metal hydroxide (adding water)
Equilibrium forms, adding OH- ions will shift equilibrium (set up a new equilibrium) to the right - leading to further dissociation. The products can then be used for further hydrolysis. Adding more OH- ions….
Adding water to aqua complexes will produce hydroxonium ions and another complex ion which has a different charge due to the formation of OH-. At some point, the complex will be neutral - solid is formed.
There are 3 steps to produce a neutral complex
Other charges like 2+ will have 2 steps
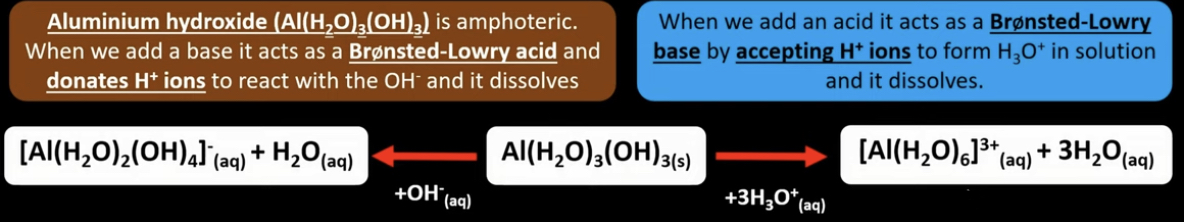
What are amphoteric metal hydroxides?
- Can act as an acid or a base
How do metal hydroxides act like a Brønsted-Lowry base or an acid?
- Able to hydrolyse metal aqua ions (acts as acid - donate hydrogen ion) to form insoluble hydroxide by adding a base.
- Dissolve in both acids and base
- A solid metal hydroxide can be ‘reversed’ back into its aqueous form….
- Add acid (H+), this will dissolve the precipitate
- This is because metal hydroxide acts as a Brønsted-Lowry base (accepts a H+ ion).
Examples:
What is the reaction between metal-aqua ions and ammonia?
Hydrolysis reaction using NH3 (weak base, needs water to form OH- ions) (NaOH can also be used for hydrolysis of metal aqua ions).
When ammonia dissolves in water, it acts as a base and accepts protons from the water ligands. This forms NH4+ and OH- ligands.
When excess ammonia is added….
In some cases, when there is an excess of NH3 there will be a partial ligand substitution reaction, where the OH- and H2O ligands will be displaced by NH3. This happens with Cu2+ complexes
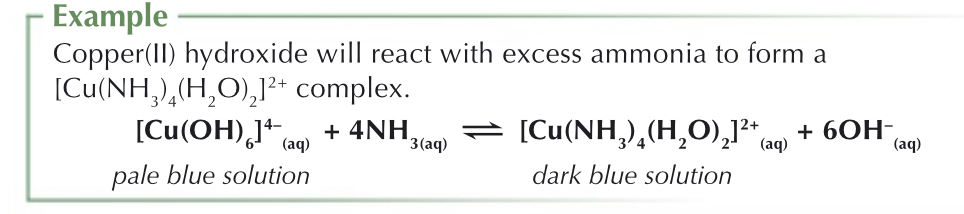
Test tube reactions:
Add sodium hydroxide, ammonia and sodium carbonate to test tubes, drop by drop and excess to see any further changes.
| Iron(II) | Copper(II) | Aluminium (III) | Iron(III) | |
|---|---|---|---|---|
| Sodium hydroxide | Precipitate | precipitate | precipitate (dissolves when excess NaOH is added, as it is amphoteric) | precipitate |
| Ammonia | Precipitate | precipitate (dissolves in excess ammonia, this is due to partial ligand substitution) | precipitate | precipitate |
| Sodium carbonate | Precipitate Readily oxidised in air to form Fe3+, so needs to be made from fresh. | precipitate | precipitate Carbon dioxide gas, so bubbles can be observed. | precipitate Carbon dioxide gas, so bubbles can be observed. |
| Cu2+ | Fe2+ | Fe3+ | Al3+ | |
|---|---|---|---|---|
| Aqueous solution (water) | Blue | Pale green Oxidises readily | Yellow | Colourless |
| Adding hydro dioxide ions or ammonia | Pale blue precipitate | Dirty green precipitate | Orange precipitate | White precipitate |
| Excess OH- | No change (insoluble in excess NaOH) | No change (Insoluble in excess NaOH) | No change (Insoluble in excess NaOH) | Colourless solution**(Dissolves in excess OH-)** |
| Excess NH3 | Dark blue solution (partial ligand substitution) | No change (Insoluble in excess NH3) | No change (Insoluble in excess NH3) | No change |
| Adding Na2CO3 | Green-blue precipitate | Green precipitate | Brown precipitate | White precipitate |