Introduction to Organic Chemistry and Alkanes
Hyrdocarbon: Compound of hydrogen and carbon only
STRUCTURAL ISOMERISM
Compounds with the same molecular formula but different structural arrangement of atoms.
Chain isomerism:
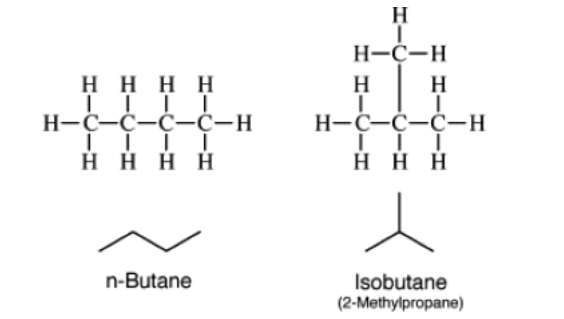 Position isomerism:
Position isomerism:
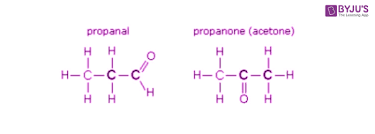
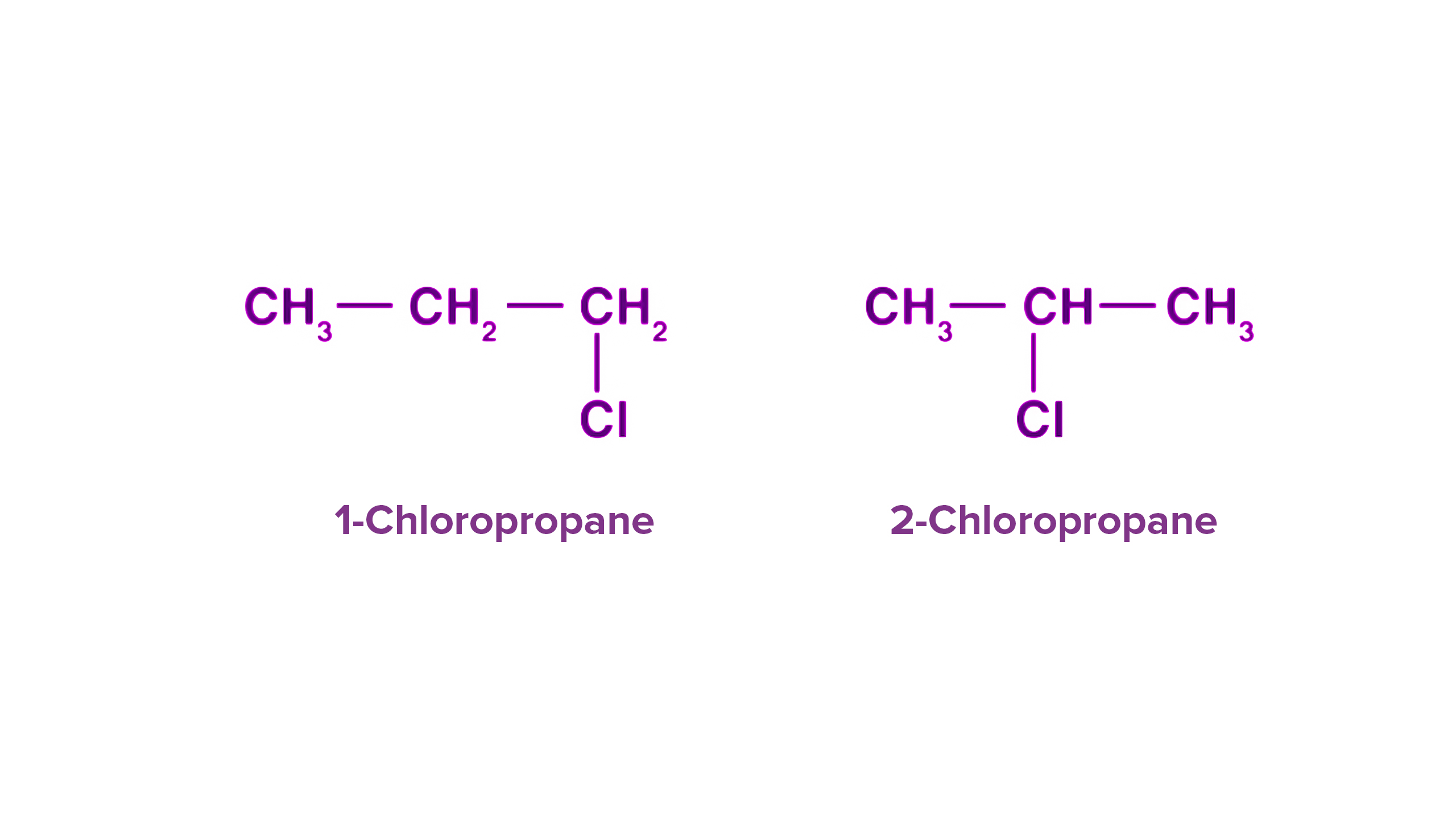 Functional group isomerism:
Functional group isomerism:
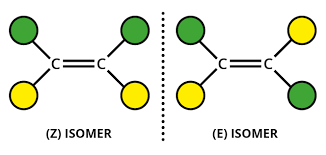
 STEREOISOMERISM:
STEREOISOMERISM:
Compounds with the same structural formula but with a different arrangement of atoms in space.
E isomerism is when the groups attached with the highest electron number are attached to different sides of the carbons.
Z isomerism is when the groups are attached to the same side.
Cis/Trans is when the groups are the same.
ALKANES
General formula: CnH2n+2
They are saturated hydrocarbons.
FRACTIONAL DISTILLATION
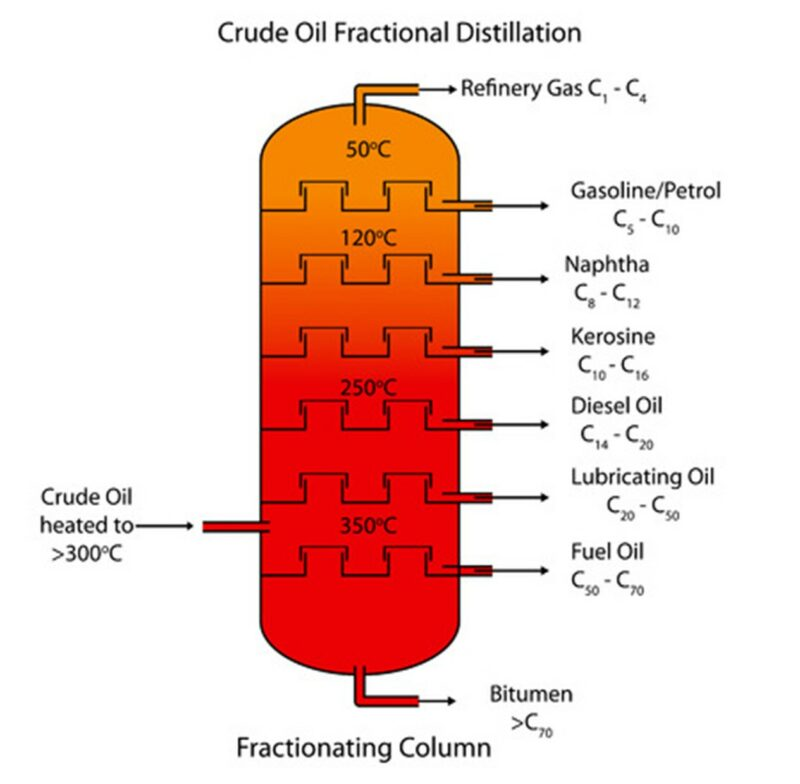 A furnace heats the crude oil to 400oC. The column is hotter at the bottom and cooler at the top. Rising vapour condenses when it reaches the tray with liquid at a temperature just below its boiling temperature. This releases energy which heats the liquid on the tray and evaporates the more volatile compounds.
A furnace heats the crude oil to 400oC. The column is hotter at the bottom and cooler at the top. Rising vapour condenses when it reaches the tray with liquid at a temperature just below its boiling temperature. This releases energy which heats the liquid on the tray and evaporates the more volatile compounds.
To ensure smooth combustions, companies produces fuel with a high octane number by increasing the proportions of branched alkanes and arenes, or blending-in oxygen compounds. The 3 mains methods used to increase the octane number are:
Cracking - which makes smaller molecules and converts straight chain hydrocabons to branched and cyclic hydrocarbons. Smaller chains are in lower supply but in more demand. When conducted in high temperatures and in the presence of steam, a higher proportion of alkenes is produced. When in the presence of a catalyst (zeolite) at 500oC, higher yields of branched and cyclic alkanes are produced.
Reforming - which turn straight chain alkanes into branched chain or cyclic alkanes or arenes such as benzene and methylbenzene and turns cyclic alkanes into arenes. Operates at 500C and catalyst is platiunum or rhodium supported on an intert material such as aluminium oxide.
Adding ethanol and ethers - such as ETBE
CARBON MONOXIDE:
Binds with the haemoglobin in blood cells to from carboxyhaemoglobin. This reduces the volume of oxygen that can be transported in the blood and can result in memory loss, nausea and headaches. High levels can lead to suffocation. Carbon monoxide is often made from malfunctioning boilers.
NITROUS OXIDES AND SULFUR DIOXIDE
Sulfur dioxide is formed from sulfurous impurities when burning fossil fuels. Nitrous oxides are fromed when temperatures rides aboue 2800 degrees. When the temperature is high enough, the nitrogen is able to react with the oxygen in the air. When these particles react with the H+ ions in water droplets in the atmosphere, they form acids. Sulfur dioxide forms sulfuric acid and nitrous oxides form nitric acid. This causes acid rain which lands on fresh water ecosystems and causes soil acidification. It also damages buildings made of limestone and marble.
Incomplete combusion forms carbon monoxide and water and occurs when alkanes are burnt in a limited supply of oxygen. This often happens in car engines. When this occurs, carbon will be produced in the form of soot.
CATALYTIC CONVERTERS
They improve air quality by removing pollutants that would be released from car exhausts.
The converter is a honeycomb of ceramic material coated in a thin layer of metals which act as the catalysts
It is a honeycomb as the large surface area increases the rate of reaction
Catalytic converters mean that 90% of pollutant gases are removed in less than a second
They work in the presence of a catalyst
ADVANTAGES OF USING BIOFUELS
Reduction of use of fossil fuels which are finite resources
Biofuels are renewable
Use of biodiesel is more carbon-nuetral
Allows fossil fuels to be used as a feedstock for organic compounds
No risk of large-scale pollution from exploitation of fossil fuels
DISADVANTAGES OF USING BIOFUELS
Less food crops may be grown
Land not used to grow food crops
Rainforests must be cut down to provide land
Shortage of fertile soils causing low yield costs in growing, processing and transport.
Renewable - a resource of which there is an endless supply because is can be replenished.
Non - renewable - a finite substance that is used up more quickly that it can replace itself, which means it cannot easily be replenished.
Biofuel - any fuel that is derived from biomass, that is, plant or algaw material or animal waste.
FREE RADICALS
Formed by homolytic fission