Chemistry MYP4 (2).docx
Unit 1- What is matter? How do we use matter?
Unit details
SOI- Forms vary in their orientation, as a consequence of relationships and interactions with conditions.
Related concept- forms, conditions, consequences
Global Context- Orientation in space and time
Key concept- Relationships
Matter: Matter is anything that takes up space
Matter can be defined as the amount of a substance to exist or any object which has a mass and occupies space
There are 2 types of matters
- Pure substances: elements & compounds
- Mixtures: homogenous & heterogeneous
Element: a simple substance which cannot be broken down into smaller parts
Compound: when 2 or more elements are combined together in a definite ratio by mass
Law of conservation of mass-
- The law of conservation of mass states that mass in an isolated system can neither be created or destroyed,
- Energy is only transferred into different forms and the total mass of the reactants in a chemical reaction equals the sum of the mass of the products
- It prevents gas from escaping
- Example: Barium Chloride + Sodium Sulphate → Sodium Chloride + Barium Sulphate.
- As seen above, none of the chemicals are lost and all of them are just displaced or changed positions hence nothing is being destroyed or created
3 Basic forms of matter
Gases
Interparticle spaces- maximum
Intermolecular force of attraction- very weak
Movement of particles, kinetic energy- yes
Low density
Compressible in some cases
No fixed shape
No fixed volume
Diffuses quickly
Liquids
Interparticle spaces- medium
Intermolecular force of attraction- medium
Movement of particles, kinetic energy- yes, but little
Medium density
Medium compression
No fixed shape
Fixed volume
Partial diffusion
Solids
Interparticle spaces- maximum
Intermolecular force of attraction- maximum
Movement of particles, kinetic energy- none
High density
Not compressible
Fixed shape
Fixed volume
Slow diffusion
Bose Einstein Condensate
- 5th state of matter
- The BEC was named after albert Einstein and an Indian mathematician. The classic example of the BEC and its original gas was rubidium atoms.
- It is a state of matter in which separate atoms or subatomic particles are cooled to near absolute zero.
- Subatomic level: the subatomic particles can be considered as small wave packets according to the quantum mechanics. When we lower the temperature to near 0 kelvin, the waves start to expand and have a longer wavelength, this causes them to overlap as the stop being individual particles and become a single quantum entity.
- The BEC is formed by cooling a gas of extremely low density, about one hundred thousandths of normal air density, to extremely-low temperature.
- It has particles called bosons, which interact with one another like atoms. However, due to the temperature they are converted from atoms to bosons.
- The bosons are formed at absolute 0K, which is -273.15 degrees Celsius.
- 2 examples of materials with the BEC are superconductors and superfluids.
- All BEC’s are extremely fragile.
- The BEC is used for research on dark matter and their interactions.
How do we get such low temperatures?
- Find some free atoms of the same element.
- Hit them with lasers from all 3 axes on 6 sides. The laser in this case is to hit the atoms and this gives a recoil to the atoms which slows them down.
- As they slow down, the temperature gets lower and lower.
- Later, a magnetic field is used to keep the atoms intact, and this is used to remove the hotter atoms.
Plasma
- 4th state of matter
- Plasma is the state of matter that consists of a mass of free electrons and positively charged particles called cations.
- It has no fixed shape or fixed volume. It can expand and evenly distribute itself in anything which it is put into.
- Plasma is also known as ionized gases.
- For certain substances, if we continue to apply heat to their gaseous forms, another change of state can occur, when mixed together.
- The substances can go from a gas to a state of matter called Plasma. For this change of state to occur, very strong heat must be applied. When the heat is sufficiently strong, the electrons are stripped from their respective atoms, creating free electrons and positive ions (+).
- The atoms are both negative and positive, but plasma is neutral as their equal amounts of positively charged articles.
- Since free electrons are present, substances in plasma form can conduct electricity. This is the difference between plasma and gases, as gases cannot conduct any form of electricity. The atoms in plasma are loosely packed close to the nucleus, which helps it conduct electricity with heat energy.
- Plasma is found in fluorescent light bulbs, neon signs and some forms of naturally occurring plasma are lightning, the northern lights and electricity. Fire can also be classified as plasma.
- Plasma is the most common state of matter and makes up almost 99% of the entire universe.
Interconversion of states of matter
- When the temperature increases, kinetic energy increases. This causes force of attraction to decrease, hence the state of matter changes.
- Higher temperature= Solidà Liquid à Gas
Lower temperature= Gas à Liquid à Solid - The temperature at which solid exists is 0 degree Celsius, which is 237.15K.
- A ‘kelvin’ (K) is the standard unit for temperature in forms of matter.
When a solid gets warm, it melts. The particles inside are moving, but not away from each other. They keep moving in their place. When energy is supplied, particles move even more. This causes the solid to change into a liquid. Hence, when a solid heats up and begins to melt, the attraction becomes weaker as particles move faster. Melting includes a transfer of energy. Energy is also needed to turn a liquid into a gas. More energy allows the particles to move more, and it leads them to separate, they later evaporate and turn into gas. Gases can be turned into liquids, but only when they are in cold temperature. When a gas changes directly into a solid it is called deposition.
Heating curves and cooling curves
Heating curve
The graph of temperature against time is called a heating curve. ![]()
- The temperature goes up the longer the heating continues.
- There are 2 horizontal flat parts; BC and DE. They show when a change of state happens.
- The plateaus are called phase changes.
- The first change of state (BC) is melting- changing from solid to liquid. The temperature stays the same but the substance melts and the form changes.
- For water, the temperature is 0 degree Celsius, because its melting point is 0°C
- In BC both liquid and solid exist in various ratios, starting at full solid (100%) and changing to full liquid (100%).
- The second change of state (DE) is boiling, changing from liquid to gas.
- The temperature stays the same while the substance boils.
- For water the temperature is 100°C because that is its boiling point.
- In DE both liquid and gas exist in various ratios and the state changes from full liquid to 100% gas.
![]()
Different substances have different melting and boiling points, but the shape of their heating curves is very similar. For example, the heating curve of iron a metal that melts at 1538°C and boils at 2861°C is similar to the one of water and follows a similar pattern.
Cooling curves
Cooling curves show how the temperature changes as a substance is cooled down.
- Cooling curves have horizontal flat parts where the state changes from gas to liquid, or from liquid to solid.
- Lauric acid has a melting point of around 45 degrees and is easily melted in a test tube placed in a beaker of hot water.
- The liquid may be cooled by putting the boiling tube in a beaker of cold water or just leaving it in the air.
- The melting and freezing occur at the same temperature, during freezing the energy is removed and during melting the energy is absorbed.
- The melting and freezing point for water is the same, which is 0 degrees Celsius.
Energy changes
- Since temperature is a measure of Average Kinetic Energy any change in temperature is a change in kinetic energy.
- All of the diagonal line segments on a heating or cooling curve show a temperature change and therefore a change in kinetic energy.
- During these regions, a single state of matter exists, and the sample is either getting hotter or cooler.
- During the horizontal line segments, there is no change in temperature, so kinetic energy remains constant.
- However, all the energy that is absorbed or released is related to changes in potential energy.
The 3 P’s- Plateau, Phase Change and Potential Energy change
The heating curves for pure and impure substances are different.
- If we have a mixture of naphthalene (pure) and wax (impure), their heating curves will vary.
- Mostly, the heating curve for a pure solid stops rising at its melting point.
- The heating curve for wax, which is a mixture of substances shows that the solid wax will melt over a range of temperatures.
- The presence of impurities in the solution increases its boiling point.
Cooling curve for changing a liquid into a solid-
![]()
The Kinetic Molecular Theory of Gas
List of rules and expectations of how we expect gases to behave
- Kinetic energy is a type of energy that everything has if it is moving.
- Example: truck driving down a highway, person walking, picking up a pencil
- The faster something is moving the more kinetic energy it has.
- Doing something very fast has more kinetic energy than something happening slowly.
- When an atom is moving fast, it has more kinetic energy and one moving slow has less.
- Ideal gas- if it follows all the rules set for the KMC.
- Solids- In a solid the atoms/particles are packed very tightly, and they only move a little around the same area, hence they have little kinetic energy.
- Liquid- The particles in a liquid have more kinetic energy since they are freer to move but they are still locked in with the surrounding atoms.
- Gases- they have the most kinetic energy. All the gas particles are in constant motion and are moving around the place. They move so fast that their average speed in room temperature is a thousand miles an hour.
Amount of kinetic energy: Solid<<<Liquid<<<Gas
- Gases consist of very small particles that are far apart relative to their side.
- Gas particles are in constant random motion. The moving particles constantly collide with each other and with the walls of the container.
- Collisions between gas particles and container walls are elastic collisions.
- There are no forces of attraction or repulsion between gas particles.
- The average kinetic energy of gas particles depends on the temperature of the gas. The hotter it is, the faster it moves.
Invisible properties of gas
- We cannot see gases as they are invisible, but we can feel them.
- Gases always move in a straight line.
- Gases are so small; they occupy no volume on their own- if they are not in a group.
- If gas molecules collide, their energy remains constant.
- Gases do not attract one another, but they do collide.
- Gases have energy that is proportional to the temperature. The higher the temperature, the higher the energy the gases have.
- At the same temperature, all gases have same energy. No matter what type of gas it is.
- If gases do bump into one another, the energy is transferred from one particle to another, and the total amount never changes.
- Gases have the characteristic of expandability. Since they can move around, they will spread out to fill any area we give them.
- We can also take a plunger and push it inside a container, all of it will end up squeezing into one. For example, this process occurs during swimming or scuba-diving when you need a tank to store oxygen. Gases are also compressible.
Gas Laws
What creates pressure in a gas?
When gas is in a container and the molecules and particles collide and hit each other, the container gets pressured from all side. The particles in a gas absorb heat, which gives them energy to move. By moving, the pressure is increased.
If there is more heat, the kinetic energy increases which leads to an increase in speed. Increasing temperature causes an increase in the pressure created by the gas.
Gas- P, T, V, amount of gas
P- Pressure
V- volume
n- number of particles in the gas
T- temperature
Boyles Law
- Gas law which tells us about the relationship between the pressure and volume of a gas.
- Less volume= more pressure
- P & V of a gas: at constant temperature and amount of gas
- If you compare a gas with a different amount of pressure, the temperature and amount of gas must remain the same (Controlled variable)
- Pressure is inversely proportional to the volume of the gas.
- Formula- PV=K
- The graph for Boyles law is always between volume and pressure, with V being the y axis and P being the x axis. The graph also includes a curved line going which goes down.
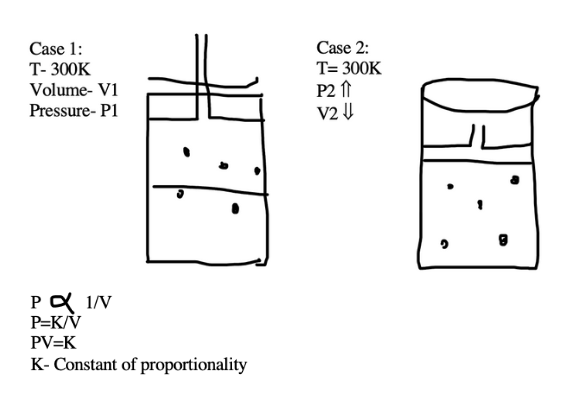
For a particular gas at constant T, n
In case 1:
P1V1= k ----1
In case 2,
P2V2= k ----2
à P1V1= P2V2
Charles Law
- Relationship between temperature and volume.
- V, T at constant n, P
- As temperature increases, the volume of the gas increases.
- As temperature increases, gas increase because the particles move away from each other with that energy.
- Formula- V/T=k
If we go from case 1 to case 2, the volume will increase since there will be more kinetic energy due to the increase in temperature.
Combined gas Law
- Relationship of pressure, volume, and temperature
- Formula: PV/T= constant
- Gay Lussac law
P1V1/T1= P2V2/T2
Ideal Gas equation
- Only applicable for ideal gases
- Formula- PV=nRT
Where n is number of particles and R is the constant.
Elements and Compounds
- Compound- When 2 or more chemicals are combined, a brand-new reaction and substance occurs.
- Elements- substances which cannot be broken down into something simpler.
- When chemicals are combined to make a compound, it is called a chemical formula.
- Chemical formula shows the combination of a compound.
- Water= compound; chemical formula= H2O
- Solutions- homogeneous mixture in which the substances are blended evenly.
- Mixture is heterogenic and a compound is homogeneous.
- Mixtures- a physical combination of 2 or more substances that retain their own properties and can be easily separated.
- Heterogeneous mixture= with visible ingredients, mixed substances are visible solutions.
pH levels
pH levels
0--------------14
0-7 acidic
7-14 basic
7= neutral
Dependent of concentration of hydrogen ions in a substance.
pHà Power of Hydrogen
pH=1/H+
0:H+ More
14; H+ Less
Separation Techniques
For every separation technique, there is a principal to be followed.
- Hand Picking: the components of a solid-solid mixture can be separated by hand picking. It is only used when the particles are large enough to be seen clearly. It is also the most commonly used method of separation.
- Threshing- this is the method that is generally used by the farmers to separate the grains from the stalks after harvesting. The dried stalks are beaten or threshed to separate the grains. However, in large farms threshing is done with threshing machines.
- Winnowing- components of a mixture which are separated by wind or by blowing air. In winnowing, the mixture is allowed to fall from a height and the light components get separated from the heavier ones due to the wind blow.
- Sifting or sieving- it is used to separate a dry mixture which contains substances of different sizes by passing it through a sieve, which is an object with tiny holes.
- Magnetic separation- process of separating substances with magnetism. Different materials are attracted to different magnets that have certain properties; the magnetism of the object used helps separate the substances.
- Sublimation- it is the process where a solid changes phase and turns directly into gas without passing through a liquid phase.
- Sedimentation- when the heavier component in a mixture settles after the water is added to it, the process is called sedimentation.
- Decantation- When the water (along with the dust) is removed, the process is called decantation. This is a quick but rough method of separating a mixture of liquid and undissolved solid.
- Filtration- it is used to separate an insoluble solid from a liquid in a solid-solid mixture. For example, separating sand from a mixture of sand and water.
Evaporation- separated dissolved solids from a liquid or solvent in a solid-solid solution.
Salt can be obtained by evaporating seawater.
Through evaporation, the seawater (solution) is heated until all liquid (solvent) evaporates and leaves the dissolved solids (solutes) as residue.
2 types of evaporation techniques generally used in labs:
- Boiling to dryness- used when solute left behind is not easily decomposed under temperatures higher than 100 degrees Celsius.
- Evaporation using steam bath- used when solute is unstable and decomposes easily at temperatures higher than 100 degrees Celsius.
- Crystallization- separation technique that results in formation of pure solid particles from a solution containing the dissolved substance. As the solvent evaporates, the dissolved substance comes out of the solution and collects as crystals. It is the solvent that evaporates away, and not the solution. It produces highly pure solids.
- Distillation- during the process as heat is applied, the water began to boil off and became water vapor which rose up to the top of the flask. The water vapor entered the condenser where cold water from the inlet caused the water to condense and run off into the receiving of the flask. As the water is removed from the solution, the solution got deeper in color because it becomes more concentrated.
- Fractional distillation- the breaking down of mixture into its component parts. This is done by boiling the mixture and separating the products by their varying boiling points.
- Chromatography- flow the mixture that is to be separated, through a material that retains some components more than others. This causes different components to flow through the material at different speeds, which causes them to separate.
Diffusion- movement of particles from higher concentration to lower concentration
Explanation: Kinetic theory of matter: When particles are in constant motion, it is known as Brownian motion. Particles collide and bounce from one place to reach another and spread.
Factors which affect diffusion
- If highly concentrated, then diffusion will be faster
- Temperature- more kinetic energy when there is more temperature, hence the Brownian motion is more due to the increase in movement of particles.
- Molecular mass- if it is greater, diffusion will be slower, if it is lighter, diffusion will be faster.
In a solution which is a homogeneous mixture of 2 or more substances, there are 2 main components: the solvent and the solute.
Solvent:
- The solvent is the component present in a larger quantity and is responsible for dissolving the other substance, the solute
- It is the substance that is usually in the liquid phase, but not always and it does the dissolving.
- In many cases, water is a common solvent, especially in biological and chemical systems. For instance, in saltwater, water is the solvent that dissolves the salt (solute)
Solute:
- The solute is the substance that is dissolved in the solvent
- It is typically present in a smaller quantity compared to the solvent.
- The solute can be a solid, liquid or gas, and it gets dispersed evenly throughout the solvent.
- For example, in sugar-water solution, sugar is the solute that dissolves in water (solvent).
Classification of matter
- Atoms: smallest part of any element while still retaining the properties of that element. Made up of protons, neutrons and electrons which are the 3 subatomic particles. Examples: hydrogen atom, gold atom
- Element: substance made up of only one type of atom. Example: gold, silver, iron
- Compound: composed of 2 or more different atoms chemically bonded which cannot be separated by physical means. Atoms are fixed in ratios such as H2O2 or H2O.
- Mixture: composed of 2 or more different atoms that are not chemically bonded ad can be separated by physical means. Example salt and water
- Molecule- 2 atoms (same or different) chemically bonded. Can be molecular element or molecular compound.
Chemistry- Criterion B & C
Variable manipulation
Independent variable- changed
Dependent variable- measured
In the title, both IV and DV must be mentioned
Scientific reasoning
- Mention the trend observed
- Provide scientific reasoning
- Add 3 data from the data values received to support observation- one at lower, one at middle, one at higher: include variation of all data received to be able to show the conduction of the investigation. This also gives an overview of the entire process and investigation which took place.
Validity of hypothesis
- If the hypothesis formed is valid/invalid/ partially valid
- If clear scientific reason is mentioned or not
- If the hypothesis talks about IV, DV and CV (with If, then, because format)
Validity of method
- 5 range of IV- 5 different investigated values
- IV and DV chosen are correct to get desired result
- CV mentioned clearly
- List of apparatus and logical steps are given
- 3 trial values and final average is calculated-with logical data collection table
- Safety & precautions taken while conducting the experiment
- For points mentioned above, if yes then it is a strength, if not mentioned it’s a limitation.
Improvements and extensions
Improvements- Add any 2 limitations mentioned for validity of hypothesis and mention how they can be worked upon and improved.
Extensions- Change the CV and IV, application of some other level (increase in number of values calculated), conducting the experiment more times to get accurate results- impact of different things which we can study
For situation/question shown below-
IV- temperature
DV- Time taken for diffusion
Since: in title both IV and DV should be mentioned
Title- impact of temperature on time of diffusion of methylene blue
IV- Temperature | DV- Time of diffusion | Average |
Scientific reasoning- trend: when temperature is high, the particles move faster.
Lab experimentation
Safety:
- Gloves
- Goggles
- Lab coat
Test tubes:
100ml test tube for heating and boiling (big)
50ml for cooling (small)
Use holder (test tube holder) to move around and hold the test tube
Stand to keep test tubes- test tube stand
Flasks:
Beaker Approximately 400ml Used for storage or holding large liquids Used to heat substances | Conical flask Also known as Elsmere flask 200 ml Used for reacting chemicals Since it is closed the chemicals will not splash | Round bottom flask Used for distillation Heating of any large quantity |
Glass tube (pipe) with grounded joints which connects condenser and the flask
Clamp stand will hold the flask
Tripod stand holds the wire gauge below the flask- wire gauge is used to balance the heating
Receiver flask is connected to the flaks by a double volt tube: the vapor will go inside, and the water will be outside in another tube (one tube is inside the other)- the water helps cool the vapor inside
Measuring cylinder & bottle brush
Funnel- used for filtration
Filter paper will be used
Fold the filter paper into a cone and put it through the funnel
Residue remains at the top and filtrate goes through the filter paper
Tripod stand holds the funnel above the beaker
China dish- used for evaporation and crystallization
Made of porcelain
It is open and can withhold a higher temperature
Dropper/teat pipet: Used for adding chemicals to a certain substance
Spatula- spoon with a small circle part
Used to add dry quantity to a mixture or compound
Thermometer till 100o Celsius
Different thermometer for above 100
Thermometer should be at your eye level to measure and be able to read it properly
Litmus papers- red and blue color
pH calculator
indicators for acids and bases
blue-> red = acidic
red->blue = basic
Burette stand - measure volume of liquid
Very accurate and precise
Any volume between 0-50ml
Pipette- used to measure liquids accurately
Between generally 25 ML- only 1 volume
Used for titration
Suction pump must be used to pull water up and through the tube
Separating funnel- to separate immiscible liquids
For oil and water, once the tap is opened only the water goes into the beaker and the oil remains at the top
Unit 2- What determines chemical change?
Physical changes | Chemical changes |
|
|
Types of reactions:
- Synthesis- combining many reactants to produce one product
- Single displacement only one thing is being displaced or removed (the stronger/higher reactant element can displace the weaker one)
- Double displacement- 2 replace each other and exchange places
- Decomposition- one product breaks into simpler products
- Combustion- burning, creation of heat and light
Types of reactions
Exothermic | Endothermic |
|
|
Rate of reaction
- The rate of reaction is how long a substance takes to form a product
- Zn + Hcl à 2nCl2 + H2 (bubbles are visible)
- Amount of bubbles = rate of reaction
- Rate of reaction = volume of H2 gas / time
- Temperature: When the temperature energy increases, the number of molecules which can reach the activation level also increases, due to the increase in kinetic energy. This also creates a larger number of effective collisions between the molecules which leads to a higher rate of reaction.
- Concentration: if we increase the concentration, there would be a difference in ratios. If the concentration is higher, more particles would be able to reach the activation level. As there will be more particles, the rate of effective collisions would be higher hence increasing the rate of reaction
- Surface area: the powdered zinc will have a higher rate of reaction compared to a piece of zinc as the inner particles are exposed to the reaction. Interactions and effective collisions would be more, hence the rate of reaction would be faster.
- Catalyst: chemical that speeds up the reaction. Adding a catalyst decreases the activation energy level hence more particles would be able to reach it quicker, which increases the number of effective collisions therefore then increases the rate of reaction.
Reversible reaction
- Products get converted to reactants
- If a product can be turned back into the thing it was before the reaction, it is a reversible reaction
Chemical equilibrium
- Rate of forward reaction = rate of backward reaction
- Concentration of reactants and product is constant and won’t change
- E.g.: in a fountain, the volume of water goes up and down, but it remains the same throughout
- Le Chateliers principle: if a chemical system at equilibrium experiences a change in its concentration, temperature or total pressure, the equilibrium will change itself to maintain the balance again.
Factors that affect chemical equilibrium-
- Concentration: the equilibrium shifts to the forward direction or product size. Increase in concentration of R to balance. The equilibrium will shift to the backward direction or reactant side, or left side to balance.
- Temperature: forward = exothermic, backwards = endothermic
Increase in temperature | Decrease in temperature |
|
|
- Catalyst: does not impact as only increases rate of reaction and chemical equilibrium remains unaffected.
- Pressure (only impacts gas): increase in pressure shifts the reaction to the side which has the less no. of total molecules or vice versa.
Parts & notations in a chemical reaction
- Reactants: what make the product (A+B)
A + B = C - What is being made (C)
A + B = C - Subscripts: used to indicate the no. of atoms of an element in a molecule
H2= 2 atoms of hydrogen - State symbols: indicate the state of each element
(s)= solid
(l)= liquid
(g)= gas
(aq)= aqueous in water - Reaction arrows
One way à
Reversible ⇆ - Coefficients: used to balance the equation, indicate the amount of reactants and products.
2H2 + O2 à 2H2O
it shows that 2 elements of water are formed with 2 hydrogen atoms and 1 oxygen atom
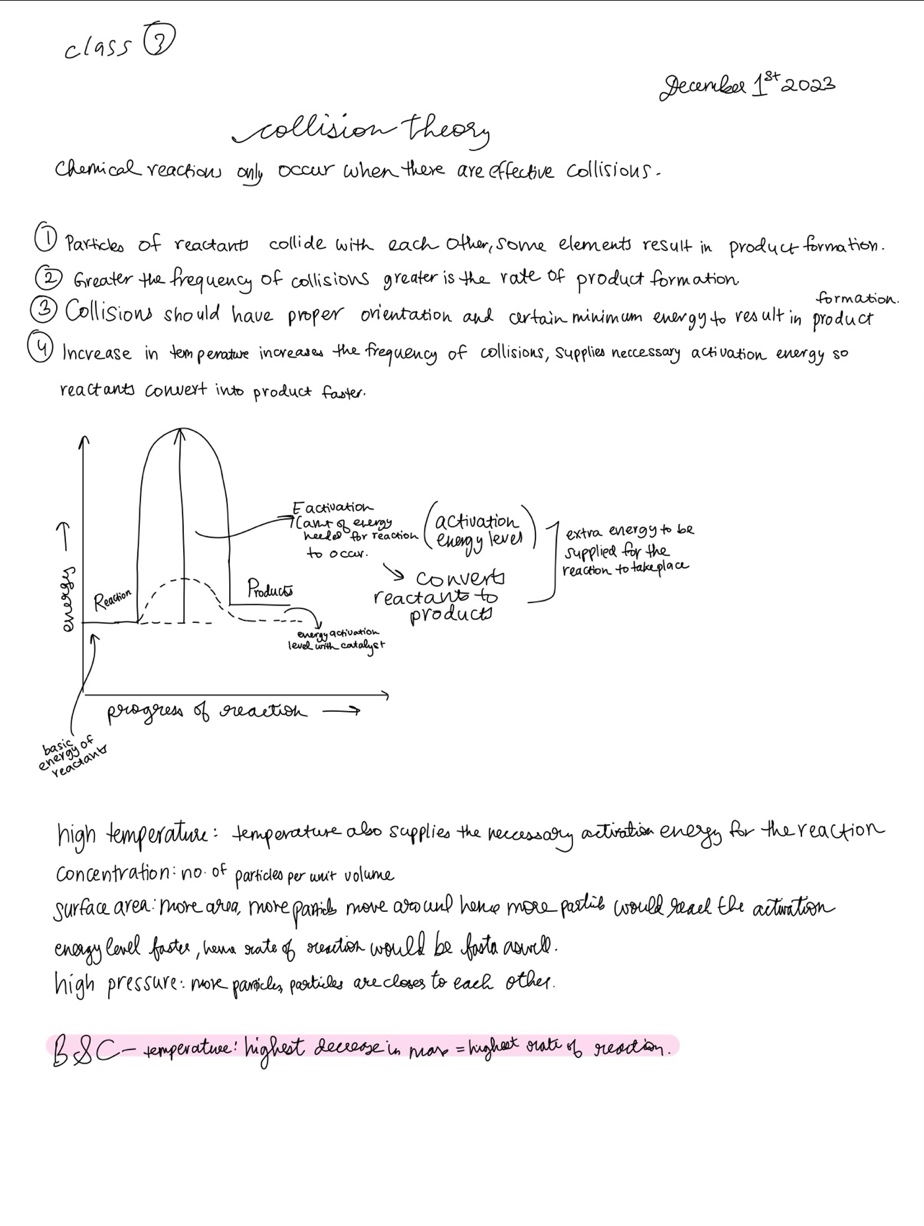
Collision theory
- Particles of reactants collide with each other, some elements result in product formation
- The greater the frequency of collisions, the greater its rate of product formation
- Collisions should have proper orientation and certain minimum energy to result in the formation of a product
- Increase in temperature increases the frequency of collisions, supplies necessary activation energy so that reactants convert into products faster.
Unit 3- Stoichiometry
SOI: evidence obtained during change may help in decision making for the required movement and bring balance.
Molar mass: sum of the total mass in grams of the atoms present to make up a molecule per mole- g/mol
Formula mass: sum of the atomic weight of the atoms in the empirical formula of the compound
Relative atomic mass: the average weight of the masses of an elements isotopes in comparison to the mass of a carbon 12 atom
relative molecular mass: mass of a molecule compared to the mass of a carbon 12 atom. it applies to anything that can be counted in moles.
Gram atomic mass: the atomic mass of an element expressed in grams
Relative masses
Law of definite proportion
- The mass of each element in a given compound has constant composition
- The ratio of the masses of the elements in a compound is always constant
Law of conservation of mass
- Mass within a closed system remains the same over time
- Total mass of reactants = total mass of products
- Mass can neither be created, nor destroyed
- Mass of A+B(products)= Mass of C+D(reactants)
- A+Bà C+D
For gases- standard temperature
Molar volume of 1 mol = 22.7dm3
Concentration in stoichiometry
- Amount of molecules in a given volume
- C = V/N
- Where c = concentration
- N= no. of moles
- V= total volume of solution in dm3
Limiting reagent à reagent which is present in small amounts as compared to ratios of a balanced chemical equation. It decides how much product is formed.
Formulas
- No of moles = given mass / molar mass
- No. of moles = given volume / 22.7 dm3
- No of moles = no. of particles / 6.02 x 1023
- Percentage yield = (actual/theoretical) x 100
- Concentration = (no. of moles g / volume of solution dm3)
- Unit for concentration = dm-3
- Empirical formula = percentage / molar mass = x
- Smallest value of x will be below in division for another x’s. the number you get is the ratio and is put in the formula.
- 1 mole of atoms/molecules = 6.022x1023 (Avogadro’s number)
- 1 mole of gas (volume) = 22.7dm3 (reacting volumes)
- Percentage purity = mass of pure sample / total mass x 100
- Molecular formula= molecular mass/ empirical formula
Unit 4- Acids, bases and salts
Unit details-
KC- Systems
RC- Patterns, interactions
GC- Scientific and technical innovation
SOI- Patterns in properties and interactions of systems, can help understand processes and develop possible solutions
KWL Chart-
Know | Wonder | Learn |
|
| (will be done at the end of the unit- reflection) |
pH scale simulator
Substance | pH | Acidic/basic |
Drain cleaner | 13.0 | Basic |
Plasma | 7.4 | Basic |
Milk | 6.5 | Acidic |
Chicken soup | 5.8 | Acidic |
Coffee | 5.0 | Acidic |
Beer | 4.5 | Acidic |
Soda pop | 2.5 | Acidic |
Battery acid | 1.0 | Acidic |
What is pH?
- A numerical measure of the acidity or alkalinity of a solution,
usually measured on a scale of 0 to 14. - pH stands for Potential of Hydrogen as it describes the concentration
of the hydrogen ions in a solution, which is the indicator of the
acidity or basicity of the solution. - The pH scale measures the strength or how acidic an object is.
The scale has values ranging from zero (the most acidic) to 14
(the most basic). - High concentration of hydrogen ions (H+ ions) indicate that the
substance is more acidic, whereas a lower concentration indicates
that the substance is basic. - A substance with a pH of 7 is neutral, a substance with a pH lower
is an acid, and higher is a base.
Importance of pH-
- pH is used to measure the basicity and acidity of a solution.
- The amount of hydrogen ion concentration in a solution determines its pH.
- Chemical reactions: pH plays a crucial role in determining the rate and extent of a chemical reaction. for example, in acid-base reactions, the pH determines the equilibrium position of the reaction
- Biological systems: In living organisms, the pH affects the activity of the enzymes. This affects the enzyme activity, protein structure and cell function.
- Environmental monitoring: pH is a key parameter in assessing the health and quality of natural environments such as lakes, rivers and oceans. A change in the pH can indicate pollution, acid rain or other environmental problems.
- Agriculture: pH is vital in agriculture, specifically in soil management and crop production. By adjusting soil pH through liming or acidification, farmers can optimize crop yields and improve soil fertility.
- Water treatment: pH is also helpful in the treatment of water. Adjusting the pH of water helps disinfection, coagulation, and precipitation of contaminants.
Factors affecting pH-
- Concentration of Hydrogen Ions (H+): The primary factor influencing pH is the concentration of hydrogen ions. More hydrogen ions lead to increased acidity (lower pH), while fewer ions result in alkalinity (higher pH).
- Industrial and Agricultural Activities: Human activities, such as industrial discharges and agricultural runoff, can introduce various chemicals into water bodies, affecting their pH. For example, the use of fertilizers and the release of acidic pollutants can alter the pH of soils and water.
- Buffers: Buffers are substances that resist changes in pH by either accepting or releasing hydrogen ions. They help stabilize the pH of a solution. Common biological buffers include bicarbonate ions in blood.
- Dissolved Gases: Certain gases, such as carbon dioxide (CO2), can dissolve in water to form carbonic acid. This reaction can lower the pH of water, making it more acidic. In contrast, the removal of dissolved gases can contribute to an increase in pH.
Acids & Bases
Acids | Bases |
Corrosive- can break things down Burn skin Turn blue litmus paper red pH below 7 sour and can be found in fruits and juices good conductors of electricity | Corrosive- can break things down pH above 7 turn red litmus paper blue slippery to touch can be found in interior of DNA double helix good conductors of electricity |
Other characteristics:
- measured using the pH scale
- The pH scale ranges from 0-14 (however, there can be ones which are outside this range)
- 0-7= acidic
- 7-14= basic
- pH of 7 is neutral
- Strong acids and bases can be combined to form salt and water
Arrhenius concept of acids and bases-
Acids are substances that disassociate in water to produce H+ ions and bases are substances that disassociate in water to produce OH+ ions.
- A chemical compound which gives H+ in an aqueous solution is an acid
- A chemical compound which gives OH- in an aqueous solution is a base
The Arrhenius concept of acids and bases, proposed by Swedish chemist Svante Arrhenius in 1884, is one of the fundamental theories in chemistry for understanding the behaviour of acidic and basic substances. According to this concept:
- Acids: Hcl(aq)àH+(aq)+Cl- (aq)
Hcl (hydrochloric) , HNO3 (nitric) , H2SO4 (sulphuric), H3PO4 (phosphoric acid)
Arrhenius defined acids as substances that dissociate or ionize in water to produce hydrogen ions (H+H+). In other words, an acid increases the concentration of H+H+ ions in an aqueous solution. Examples include hydrochloric acid (HClHCl), sulfuric acid (H2SO4H2SO4), and acetic acid (CH3COOHCH3COOH). The general reaction can be represented as:
acid→H++anionacid→H++anion - Bases: NaOH(aq)àNa+(aq)+OH- (aq)
According to Arrhenius, bases are substances that dissociate or ionize in water to produce hydroxide ions (OH−OH−). Bases increase the concentration of OH−OH− ions in an aqueous solution. Examples include sodium hydroxide (NaOHNaOH), potassium hydroxide (KOHKOH), and ammonia (NH3NH3). The general reaction can be represented as:
base→cation+OH−base→cation+OH− - Neutralization Reaction: When an acid reacts with a base, they undergo a neutralization reaction, forming water and a salt. The H+H+ ion from the acid combines with the OH−OH− ion from the base to form water (H2OH2O), while the remaining ions form a salt. The general equation for a neutralization reaction is:
acid+base→water+saltacid+base→water+salt
Brensted Lowry concept-
- Acid (H+ giver) –
are substances which give H+ ions in aqueous medium and are proton donors
example for acid: NH3+H2O→NH4++OH− - Base (H+ receiver) –
substance which accepts a proton (H+)
example for base: NH3+HclàNH4++Cl-
Acid-base conjugate pair differs by H+ ion
Indicators
- An indicator is a chemical compound that changes its colour based on the presence of an acid or a base
- Indicators are generally derived from plant pigments and are mildly acidic or basic in nature
Types of indicators-
Natural indicator | Artificial indicator | Universal indicator |
Litmus | Phenolphthalein | pH paper |
Red cabbage juice | Methyl Orange | Universal indicator solution |
Bromothymol Blue | ||
Methyl Red |
Alkali & Bases
- All bases are not soluble in water.
- However some of them do dissolve in water such as sodium hydroxide, potassium hydroxide. These bases are called alkalis.
- Hence it can be said that- alkali are those bases which are soluble in water.
- Therefore, it is clear that- all alkali are bases but all bases are not alkalis.
Strength of an acid/base:
- Strong acid/base: if all the molecules of acid or base ionize completely in a aqueous solution, then it is called a strong acid or base
- Weak acid/base: if only few molecules of acid or base ionize (partially) in an aqueous solution, they are known as weak acids or bases
Acids | Bases | |
Strong |
|
|
Weak |
|
|
The strength of an acid or base is often expressed using qualitative terms such as "strong" or "weak." However, it can also be quantitatively measured using parameters such as the acid dissociation constant (Ka) for acids or the base dissociation constant (Kb) for bases. Strong acids and bases have large dissociation constants, indicating complete dissociation, while weak acids and bases have small dissociation constants, indicating partial dissociation.
Concentration-
It can be either concentrated or dilute
Concentration is relative
Concentrated | Dilute |
solute is more and solvent is less to prepare same volume of solution 5 moles/dm3 2 moles /dm3 | solute is less and solvent is more to prepare same volume of solution 2 moles /dm3 1 mole /dm3 |
Chemical properties of acids-
- When a metal reacts with an acid, the products are a salt and hydrogen gas
- The salt produced depends on the metal and type of acid involved in the reaction
- Metal+ Hcl à metal chloride
- Metal+ sulfuric acid à metal sulphate
- Metal + nitric acid à metal nitrate
Reaction of acids
- Acid + metal = salt + H2
- Acid + metal oxide= salt + H2O
- Acid + base (metal hydroxide) = salt + H2O (neutralization reaction)
- Acid + metal carbonate = salt + H2O + CO2
- Acid + metal bicarbonate = salt + H2O + CO2
- Acid + hydroxide = salt + H2O
- Acid + carbonate = salt + CO2 + H2O
Neutralization reaction: acid + base
- Whenever acid reacts with base, a neutralization reaction occurs and the product is salt and water.
- Example: all carbonates react with acids to form a salt, carbon dioxide and water.
- Calcium carbonate + hydrochloric acid à calcium chloride + carbon dioxide + water
CaCO3+2HCl→CaCl2+ CO2+H2O - Sodium carbonate + sulphuric acid à sodium sulphate + carbon dioxide + water
Na2CO3+H2SO4→Na2SO4+CO2+H2O - Copper carbonate + nitric acid à copper nitrate + carbon dioxide + water
CuCO3+2HNO3→Cu(NO3)2+ CO2+H2O
Word equation examples:
- Sodium carbonate + nitric acid à sodium nitrate + carbon dioxide + water
- Calcium carbonate + nitric acid à calcium nitrate + carbon dioxide + water
- Zinc carbonate + sulfuric acid à zinc sulphate + carbon dioxide + water
Reactions of bases:
- Ammonia+ base à salt + ammonia gas + water
- Base + acid à salt + water
- Base + metal à salt + hydrogen gas
- Base + CO2 à carbonate salt + water
- Base + fat/oil à soap + glycerol
Detecting that hydrogen gas is produced with the salt:
Burning of hydrogen gas with a pop sound indicates its presence
Chemical properties of alkalis
- Alkalis heated with ammonium salts give off ammonia gas
- General equation: alkali + ammonium salt à ammonia + salt + water
Soil acidity determines the availability of mineral nutrients for your vegetables
- In alkali soils, phosphorus, iron and zinc are limited
- In acidic soils, calcium and magnesium are less available to plants
Oxides
Basic oxides (and hydroxides):
| Acidic oxides: Examples: Sulphur trioxide + water = H2SO4
|
Neutral oxides:
| Amphoteric oxides:
|
Identifying what type an oxide is:![]()
Salts
- Salts are a class of ionic compounds that can be produced when an acid and a base react
- Acid + base à salt + water (neutralisation reaction)
- There are 2 types of salts ; soluble (sodium hydroxide) and insoluble (lead chloride)
Method 1 | Method 2 |
|
|
Soluble or insoluble?
- The salts like copper sulphate and zinc sulphate are soluble in water. However, some salts are insoluble.
- The insoluble salts can be made by mixing 2 soluble compounds.
- The solid obtained when solutions of 2 soluble compounds are mixed is called precipitate.
- It is important to know which compounds are soluble in water and which are not. This is done by a rule called solubility rule.
Solubility rule:
Soluble | Insoluble |
All salts of group 1 elements | Chlorides, bromides, and iodides of silver and lead |
All nitrates | Sulphates of calcium, barium and lead |
All ammonium salts | Most hydroxides and carbonates |
Most chlorides, bromides and iodides | Most metal oxides |
Most sulphates | |
Group 1 hydroxides and carbonates | |
Group 1 and 2 oxides react with water |
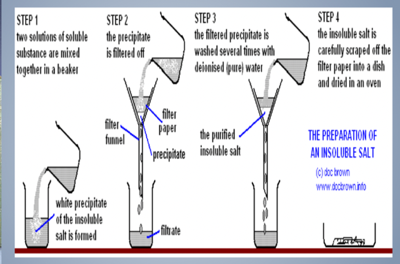
To make an insoluble salt: lead chloride
- Identify the ions present in insoluble salt- lead and chloride
- Use the solubility rules to choose soluble compounds including these ions- for example, lead nitrate for the lead and sodium chloride for the chloride.
- Add one solution to the other.
- Filter off the precipitate then wash and dry the solid.
What happens in a precipitation reaction?
- In a solution of an iconic compound, the ions are free to move so the solution of lead nitrate contains lead ions and nitrate ions which are separate from each other.
- They are able to move freely and randomly throughout water. The water molecules help keep them in solution. A similar thing happens with the solution of sodium chloride. When the solutions are mixed, an insoluble salt and lead chloride is precipitated.
- The lead ions in the solution have a greater attraction for the chloride ions than the water molecules to keep them in the solution. So the lead and chloride ions come together in large numbers to form a 3D ionic lattice. The sodium ions and nitrate ions remain in solution and are the spectator ions.
- Spectator ions: exists in the same form on both the reactant and product sides, it is left unchanged and does not affect equilibrium.
Preparation of salts:
Soluble Copper sulphate | Insoluble Lead iodide |
CuO(s) + H2SO4(aq) à CuSO4(aq) + H2O(g) | Pb (NO3)2(aq) + NaI(aq) à PbI2(s) + NaI(aq) |