nuclear chemistry
%%nucleus review:%%
- %%atomic nuclei%% are made up of positively charged protons and neutral neutrons
- # of protons determines the element
- %%isotopes%% are atoms of the same element with different # of neutrons
- nuclear chemistry is chemistry that deals with changes to the nucleus of atoms
- this is the very first type of “%%reaction%%” that we will deal with
%%stability of nuclei:%%
- not all atomic nuclei are stable, meaning that they cannot exist forever when left undisturbed
the full explanation why deals with some extremely complicated physics (%%short answer: stability = minimal amount of energy, instability = not yet at minimum%%)
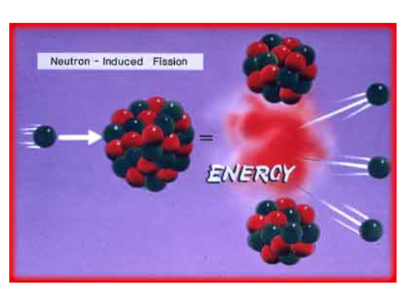
- an unstable nucleus is %%radioactive%%—it releases energy by emitting high-speed particles (%%radiation%%)
- any isotope of an element that has an unstable nucleus/is radioactive is known as a %%radioisotope%%
type 1: very heavy elements—any element above 82 is radioactive
type 2: bad ratio of protons and neutrons
chemical vs. nuclear rxns:
- chemical
- rearrange atoms (%%share & transfer e-%%)
- the nucleus is unchanged
- 2(H2) + O2 → 2(H2)O
- affected by the changes in T, P, or catalysts
- nuclear
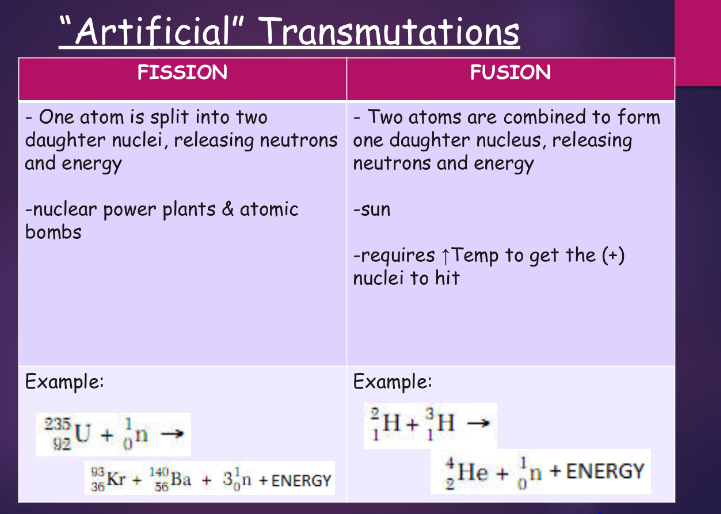
- change 1 element into another transmutation
- the nucleus is changed
- not affected by the changes in T, P, or catalyst
REFERENCE: TABLE O
%%natural decay:%%
before/parent nucleus → after/daughter nucleus
natural decay: alpha (α)
speed - slow
mass - 4 amu
charge - +2
%%α-particle = 4/2 He (emission)%%
natural decay: beta (β-)
speed - medium
mass - 0 amu
charge - -1
%%β--particle = 0/-1 e (emission)%%
natural decay: positron (β+)
speed - medium
mass - 0 amu
charge - +1
%%β+ - particle = 0/+1 e (emission)%%
natural decay: gamma (γ)
speed - fast
mass - 0 amu
charge - 0
penetrating powers rank (increasing):
- alpha
- beta
- gamma
- neutron
using table n: shows natural decay processes of several radioisotopes
- radioactive decay is the process by which an %%unstable atom loses energy%% via radiation
- natural decay—naturally occurring, not human-induced
important information about natural decay:
- there is always going to be one reactant (starting material)—%%the original radioisotope%%
- there will always be two products—%%the emitted particle and the new isotope%%
- mass and charge must balance out
finding radioactive decay products:
- determine what type of decay the given isotope undergoes
alpha: loses 2n and 2p
beta-: 1n becomes 1p
beta+: 1p becomes 1n
- identify the %%radioisotope%% and %%all necessary atomic information (mass number, atomic number)%%
- apply the change from the proper decay type
- show the change with proper notation
(reactant) → (product 1) + (product 2)
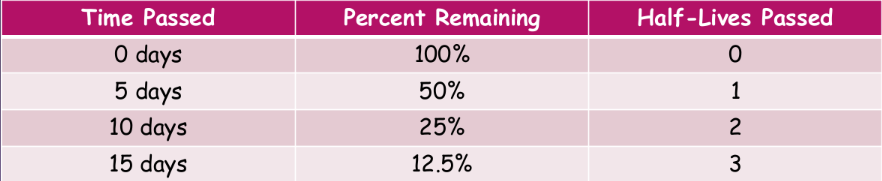
%%half-life:%%
can we calculate when a radioactive atom decays?
we can’t, you can’t influence radioactive decay by changing the conditions surrounding an atom, and it is entirely random
- our best guess is to measure a sample of radioactive atoms to determine how long it takes for half of the atoms in them to decay into the product
- this is the half-life of a radioactive isotope—the amount of time it takes on average for half of the atoms in a sample to radioactively decay
%%transmutations:%% atoms of one element changing to atoms of a different element
two kinds: natural & artificial
natural - happens spontaneously, radioactive decay, 1 reactant
ex.: uranium-238 → thorium-234 + alpha particle
artificial - requires shooting particles at another particle, 2 reactants
some general physics knowledge:
E = mc^2
E = energy
m = mass
c = the speed of light
(this equation tells us that mass could be converted to energy)