Proteins
The elements that make up a protein - Carbon, hydrogen, oxygen, nitrogen and often sulphur
Proteins are polymers made from amino acids
- polymer - long repeating chain of smaller molecule joined together
2 amino acids joined together is called a dipeptide
Many amino acids joined together is called a polypeptide
The structure of an amino acid
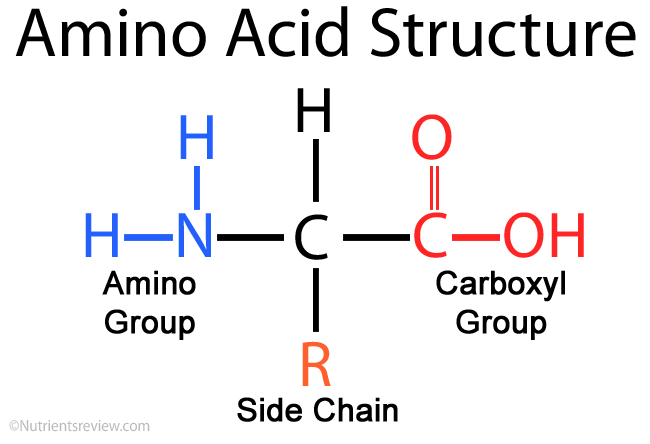
- There are 20 amino acids in the human body
- The bonds between amino acids are called peptide bonds
- They form between the amino group of one amino acid and the carboxyl group of another. They form by a condensation reaction where a molecule of water is formed
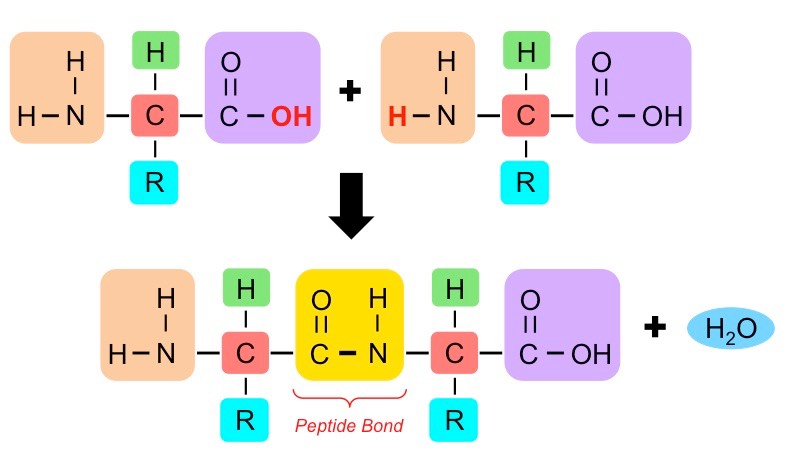
They are broken by hydrolysis where a molecule of water is added to break the bond
Structures
Primary
- The chain of amino acids coded for by the DNA
Secondary
- The simple structures formed when the ammino acid chain's fold and twist
- They form alpha helix and beta pleated sheet shapes, held together by hydrogen bonds
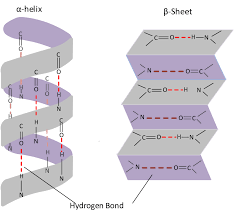
- The hydrogen bonds form between the negative carboxyl group and the positive amino group
Tertiary
- The proteins fold the 2 secondary shapes into complex 3d shapes
- They are held together by
- Ionic bonds between the strongly positive and negative R groups of amino acids
- Disulphide bonds which are covelant bonds between 2 sulphur containing groups
- Hydrogen bonds which are very weak but form almost everywhere
The tertiary structure is important to the function as in enzymes if the shape alters then the active site will alter causing the enzyme to no longer be complementary and thus not function
Quaternary
- Quaternary structure occurs when a protein has several different polypeptide chains and then they fold up to fit together, this is a quaternary structure
- An example of this is the protein haemoglobin which has 4 polypeptide chains
- A proteins structure can be broken by conditions which are too hot, wrong pH, too saline or too many heavy metals