Types of Reactions + Predicting Products
Synthesis: Two things combining into one. More reactants than products.
Decomposition: One thing falling apart into two. More products than reactants.
Combustion: Burning. Usually reacts Hydrocarbon and O2 to create CO2 and H2O
Single Replacement: Swapping one element. Reactants: One Element and a Compound. Products: One Element and One Compound, but different from reactants.
Double Replacement: Swapping two elements. Reacts 2 compounds and produces 2 different compounds.
In Replacement RXNs, must replace cations with other cations and anions with other anions.
All neutral compounds must have a cation and an anion when finished, and in the cation then anion order.
New subscripts needed
Remember diatomic elements
Predicting Products
If given words, convert into formulas.
Cross over neutral compounds
Diatomics
Identify reaction
Write Products
Balance Equation
Activity Series of Metals
Metals can replace other metals if they are above the metals they are trying to replace.
Metals above hydrogen can replace hydrogen in acids.
Metals from sodium upward can replace hydrogen in water.
Lithium
Potassium
Calcium
Sodium
Magnesium
Aluminum
Zinc
Chromium
Iron
Nickel
Lead
Hydrogen
Bismuth
Copper
Mercury
Silver
Platinum
Gold
Activity Series of Halogens
Halogens can replace other halogens in compounds if they are above the halogen they are trying to replace. This is shown in the periodic table.
Fluorine
Chlorine
Bromine
Iodine
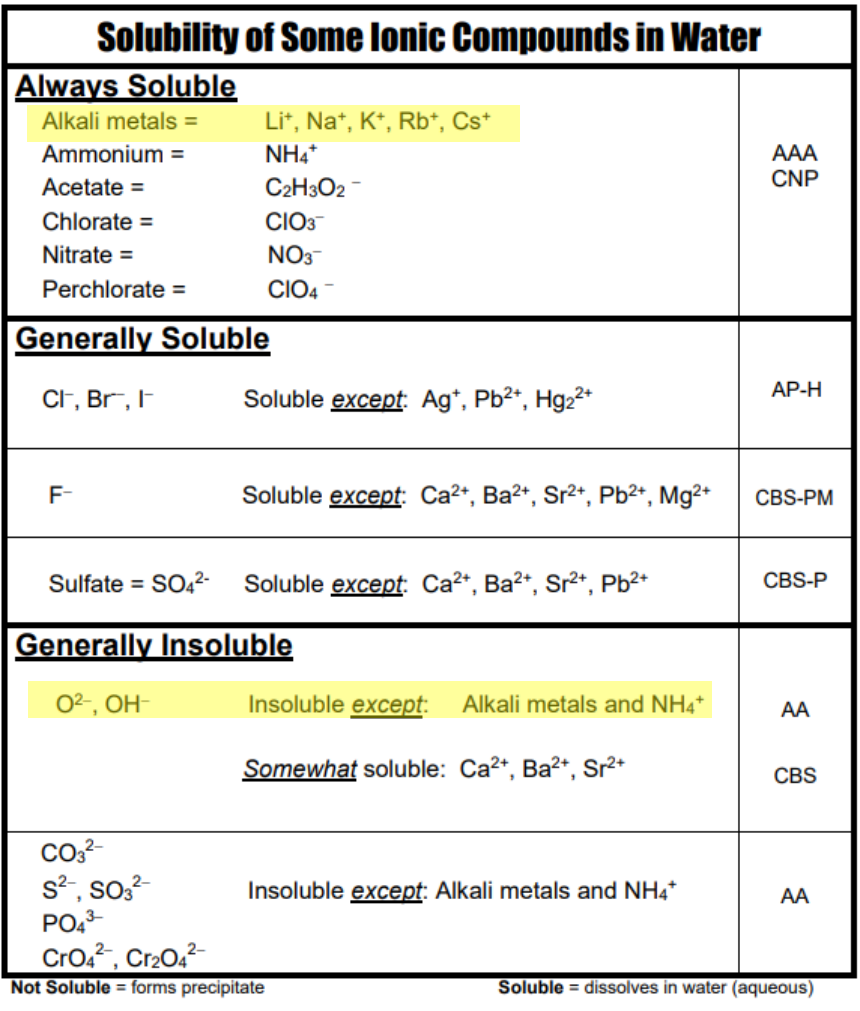
Net Ionic Equations

