Nuclear Chemistry
- Elements undergo chemical changes so it can become more stable.
- Stability depends on the proton to neutron ratio.
- Atomic numbers < 20 are the most stable because the ratio is about 1:1
- Henri Becquerel called emission from decay uranic rays because they were emitted from uranium.
- Marie Curie discovered Po (Polonium) and Ra (Radon) which emitted uranic rays, changing uranic rays to radioactivity.
\n
Radioactivity: emission of subatomic particles or high energy electromagnetic radiation by nuclei. such atoms/isotopes said to be radioactive
\n
Nuclear Decay:
- protonsnucleons, (neutrons + protons)X
- An unstable nucleus undergoes a change and reduction in energy to become more stable.
- The four types of nuclear decay (+ gamma)
- Alpha decay
- largest ionizing power, lowest penetrating power
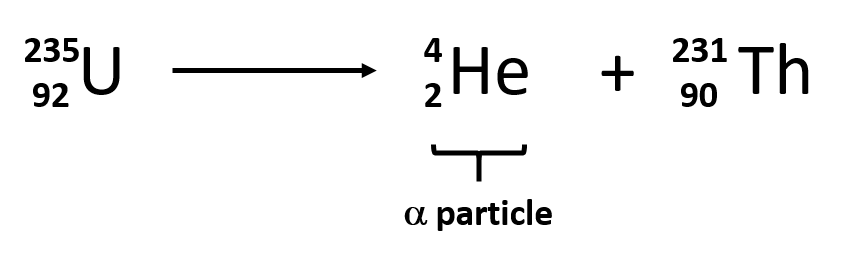
- can penetrate matter
- nucleus emits alpha particle
- # nucleons must =, # protons must =
- 24He is the alpha particle. It is always one of the daughter particles.
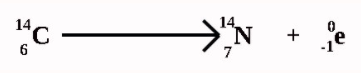
- Beta decay
- high speed electron is ejected from a nucleus
- turns neutron into proton
- it has a lower ionizing power than alpha particle but a higher penetration power
- Positron emission
- positron is antiparticle of e
- collision with e causes gamma ray emission
- cause: too little neutrons
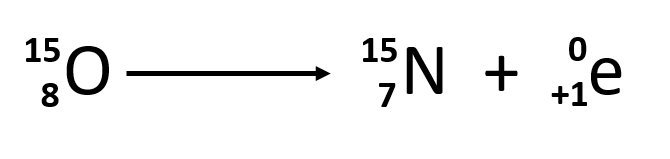
- proton converted into neutron, emits positron
- Gamma decay
- electromagnetic radiation
- high-energy photons
- no charge, no mass
- usually emitted in conjunction with other radiation types
- lowest ionizing power, highest penetrating power
- requires several inches lead shielding
- Electron capture
- particle absorbed by unstable nucleus
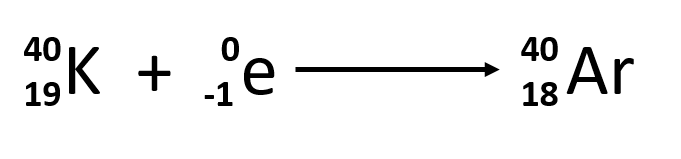
- converts proton into neutron
- different from beta decay! 2 reactants, 1 product
- Nuclear decay series:
- Uranium has an atomic number greater than 83. Therefore it is naturally radioactive.
- Uranium decays into Thorium by alpha decay.
- Thorium decays into Protactinium by alpha decay.
- Protactinium decay next and so on until we reach a stable non-reactive element.
\n
\n
\n
Nuclear half-life:
- Unstable nuclei emit either an alpha, beta, or positron particle to try to shed mass or improve their neutron to proton ratio.
- You cannot predict when an oculus will disintegrate for individual nuclei, but you can when you look at large # of atoms.
- The half-life of any nuclide does not depend on temperature, pressure, or amount of material left. It depends on the N/P ratio.
- Loss of mass to decay
- ** \n **
| Amount of Beanium | 64 | 32 | 16 | 8 | 4 | \n |
|---|---|---|---|---|---|---|
| Fraction left | 1 | 1/2 | 1/4 | 1/8 | 1/16 | 1/2^x |
| # Half life | \n | 1 | 2 | 3 | 4 | x |