AP Bio Unit 1-4 Recap
Unit 1
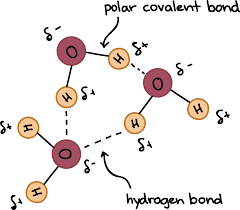
Water and its structure
Water is made up of 2 hydrogen and 1 oxygen, (H20). It is a polar covalent bond since it does not share electrons evenly. Therefore, leading to a partial negative and positive. Theres hydrogen bonds between different water molecules which bring them together. Oxygen is more electro negative than hydrogen.
Water properties
These things allow for water to have many properties. Cohesion where water is attracted to each other and Adhesion where water is attracted to other polar substances. Cohesion leads to there being a surface tension and this allows for a rock to bounce when thrown correctly. Since theres a force. Which also allows for animals to walk on water. Adhesion allows for water to move up a plants stem which is known as capillary action
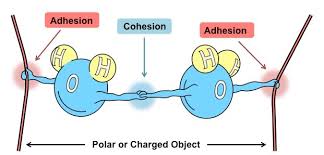
Water also has high specific heat. Meaning that it needs to take up a lot of heat in order to change its temperature. When water boils it needs lots of heat. This property allows our body to main body temp which is an example of homeostasis. Additionally, water's density is unique; it is less dense as a solid than as a liquid, which is why ice floats on water. This property is crucial for aquatic life during winter, as it insulates the water below and maintains a stable environment. The cohesive and adhesive properties of water also play a significant role in various biological processes, such as the transport of nutrients and waste in organisms, highlighting water's importance to life.
Macromolecules
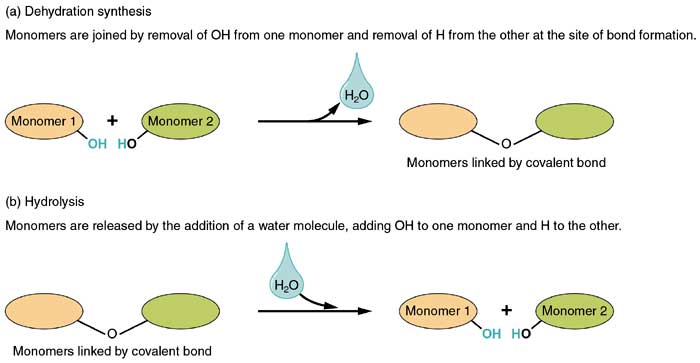
Hydrolysis breaks macromolecules by adding water (hydro-water) (lysis-break)
Example: Maltose turns into two glucose when u add water to break it down.
Dehydration synthesis builds up macromolecules by removing water.
Example: Two amino acids remove the H(hydrogen group) and OH (Hydroxile) to form a dipeptide
Monomers make up polymers and polymers are made up of monomers. Monomers are building blocks of polymers.
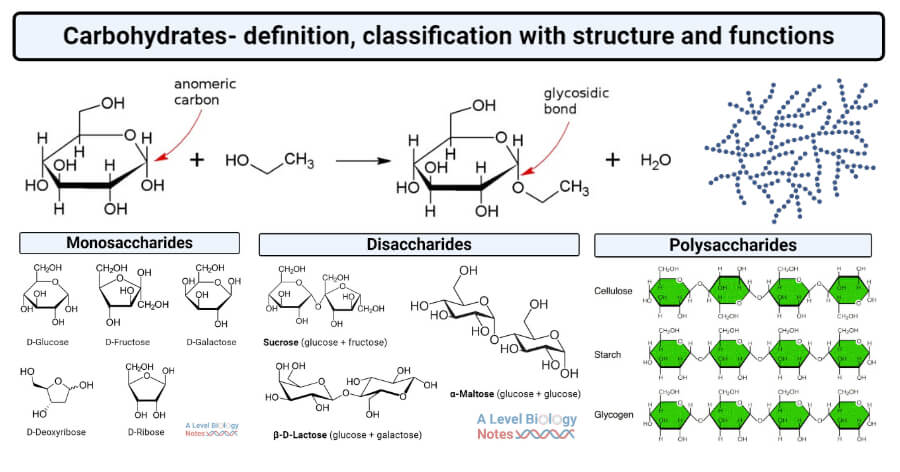
Carbohydrates: made up of carbon, hydrogen and oxygen. It has 1:2:1 ratio. 6 carbon, 12 hydrogen and 6 oxygen. Essential for energy storage; short term and it also provides and is used for structure in the cell membrane of a cell.
Monomer: Monosaccharide; simple sugar (Glucose and Fructose) these are the basic unit of a carbohydrate.
Disaccharides: Formed when two monosaccharides undergo a dehydration reaction, examples include sucrose and lactose; these serve as a quick energy source. In addition, carbohydrates can also be characterized by their functions in living organisms, including serving as signaling molecules and participating in cellular recognition processes. Additionally, carbohydrates play a crucial role in metabolism and are involved in various biochemical pathways that contribute to cellular processes.
Polymer: Polysaccharides: Complex carbohydrates formed from long chains of monosaccharides, important for energy storage energy
Starch: found in plant cells energy storage: linear chain
Glycogen: found in animal cells and used as an energy storage: highly branched
Cellulose: in plant cell walls
Citin: found in fungi cell walls and exoskeleton of arthropods
Bond: The bond that forms carbohydrates is called a Glycosidic Linkage (Covalent Bond)
Carbohydrates can be found in the form of a ring, branch and chain.
Lipids
made up of Carbon, Hydrogen, Oxygen and Phosphate group but only for phospholipids which make up the cell membrane of a cell. Lipids are not macromolecules.
Lipids serve a role of storage. Long energy storage and also they play a role in making up structures to keep cells alive. They are non polar which means that they don’t interact with water or dissolve they instead don’t mix with it.
Lipids: Fats, Steroids and Phospholipids.
Monomer: no true monomer
Fats: Give Energy storage and support cell function plus insulation
Fatty Acids: are long chain hydrocarbon with carboxyl group.
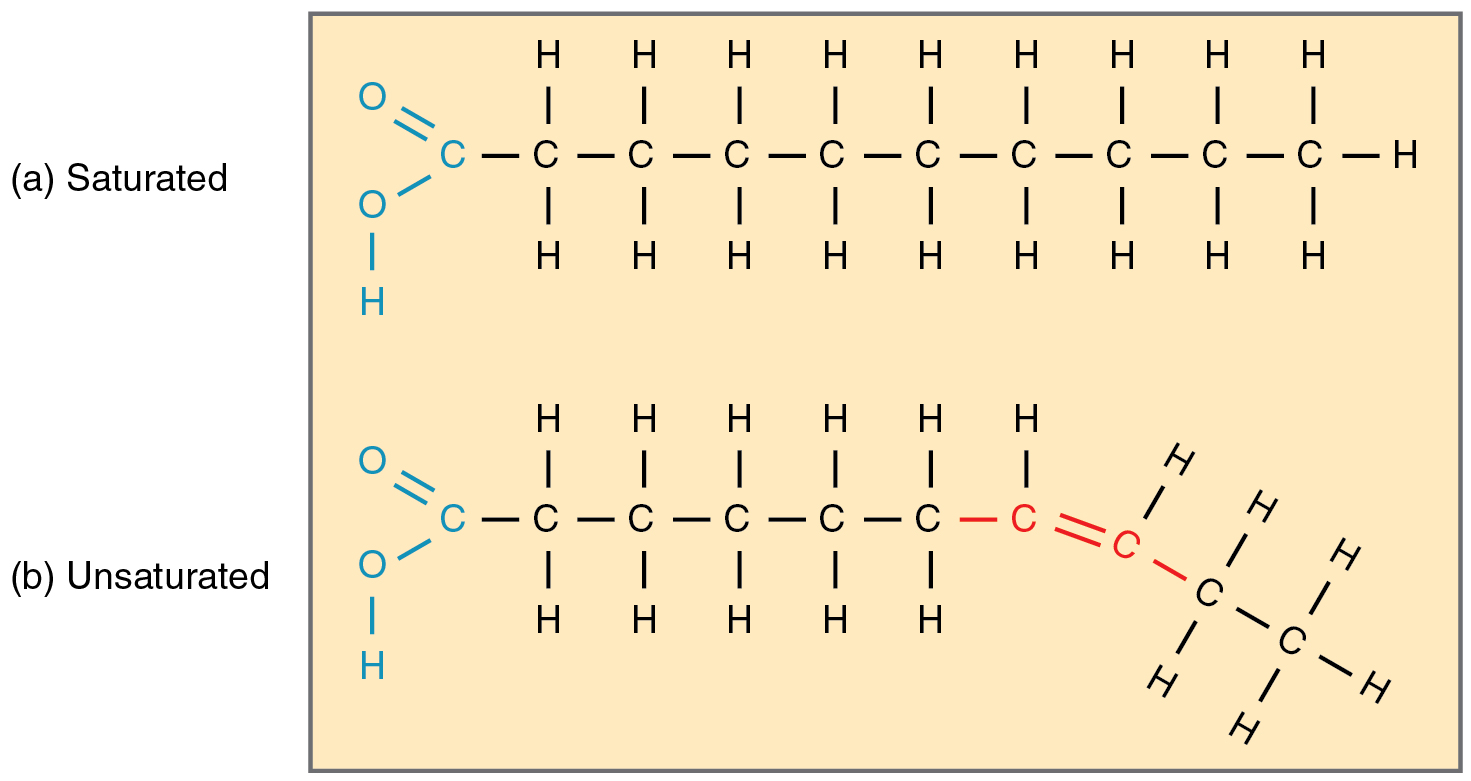
Saturated fatty acids: All single bonds, each carbon is saturated by hydrogen. As number increased its more solid. Example: butter. Unhealthier fats.
Unsaturated fatty acids: double bonds, Not all carbons are saturated by hydrogen. As number increased it becomes more liquid. These are considered healthy fats that cause less heart damage. Example: oil
Trans fats: These are industrially created fats that can be harmful to health, often found in processed foods.
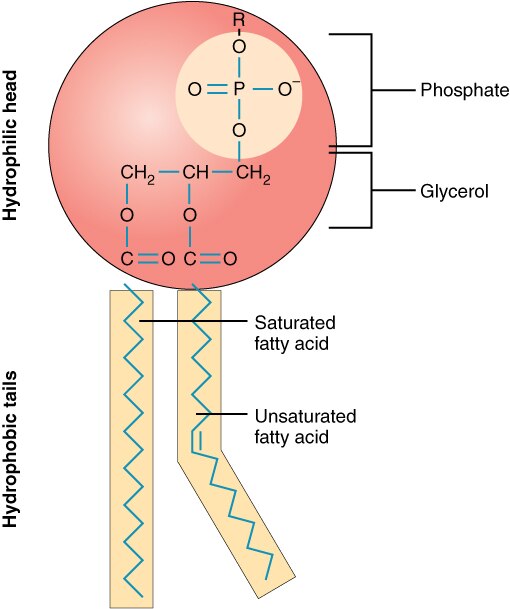
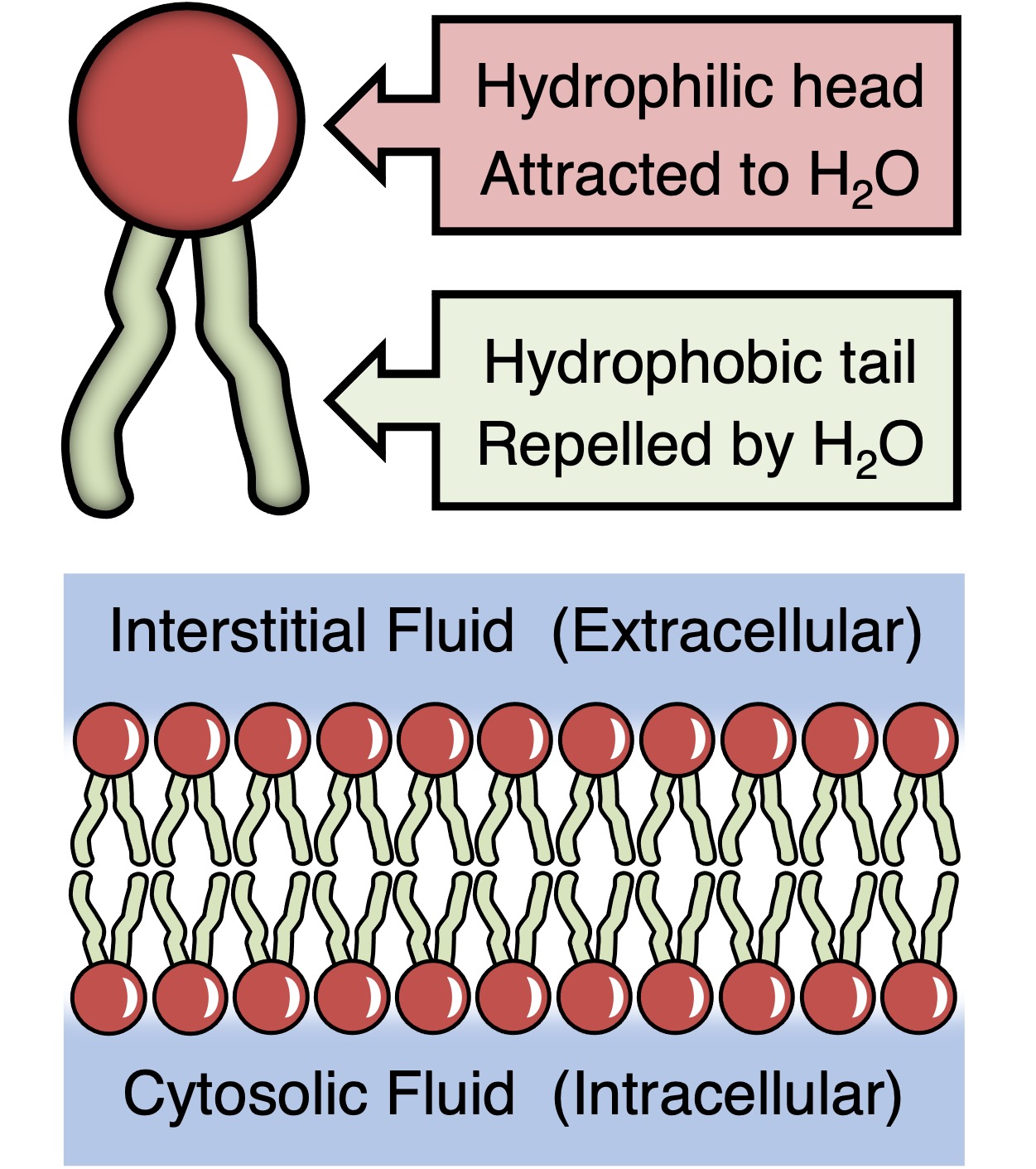
Phospholipids have a phosphate, glycerol and 2 fatty acids. Hydrophilic head and hydrophobic tail. Amphipathic is a word thats used to describe it. Because it has two different regions. One loves water and the other hates it.
Function: forms the lipids bilayer found in the plasma and cell membrane.
Steroids: are hormones and these are crucial for a variety of things for humans. It helps with growth and development. Also energy metabolism and homeostasis. Steroids are also cholesterol and this is found in the cell membrane which maintain fluidity.
Nucleic Acids
are made up of Carbon, Hydrogen, Oxygen and Phosphorous. They are made up of base, a phosphate and a sugar.
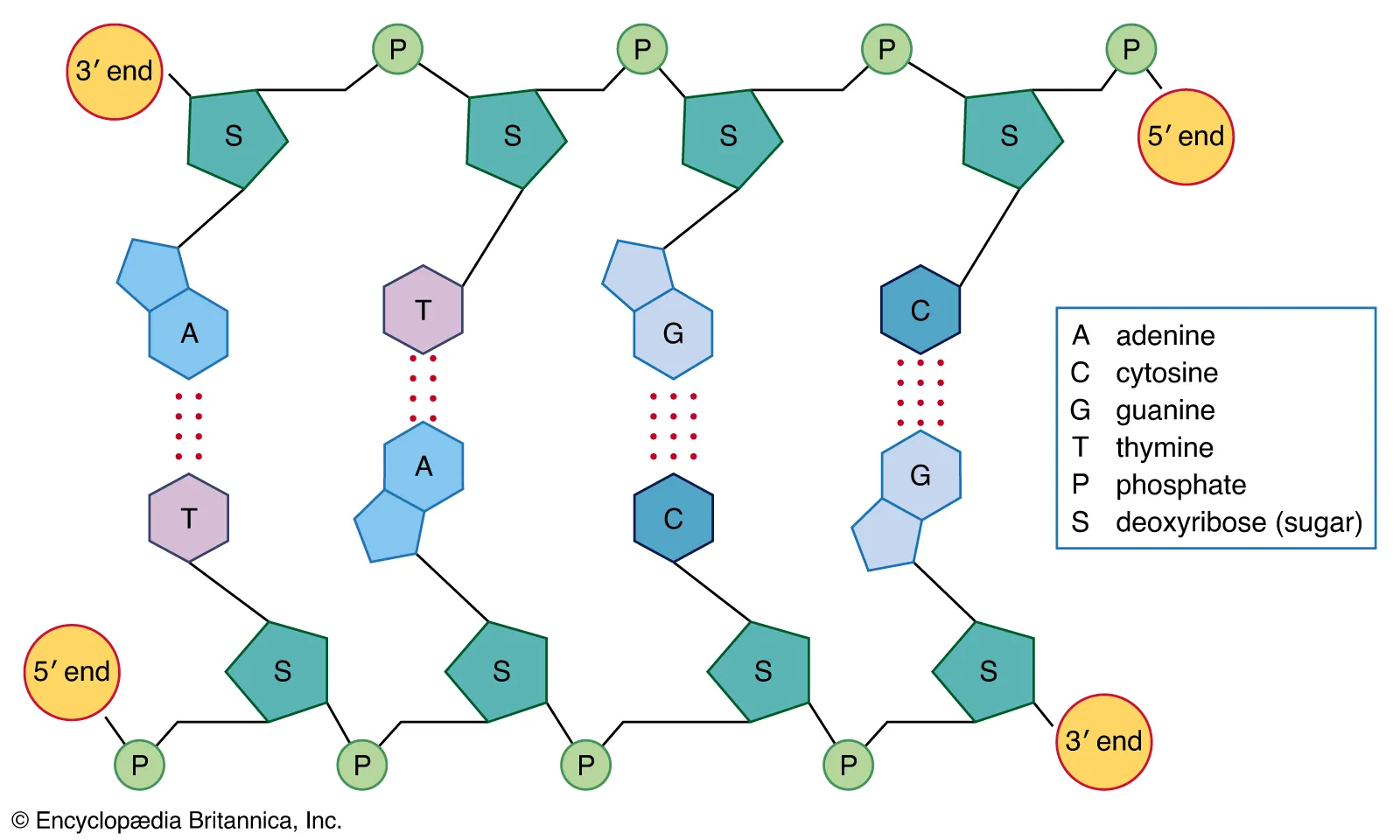
There are two different types DNA and RNA, Nitrogenous bases vary from adenosine, guanine, cytosine, and thymine in DNA, whereas RNA contains uracil instead of thymine. So A binds with T, C binds with G and in RNA, A binds with U. Double ring A,G which are purines and C,U,T are pyrimidine they are single rings. These bases are held by hydrogen bonds. A and T are held by two hydrogen bonds and C and G are held by three meaning that C and G are harder to break up.
Monomer: Nucleo tide
Bond: Phosphodiester linkage (between phosphate and hydroxyl, covalent bond)
Nucleic acids have a 5’ end and that goes down to a 3” end , which indicates the directionality of the DNA or RNA strand. This structural feature is crucial for replication and transcription processes. In addition to these properties, nucleic acids play a vital role in storing and transmitting genetic information across generations. Furthermore, the sequence of nucleotides in a nucleic acid determines the specific genetic code, which guides the synthesis of proteins and regulates cellular functions. Synthesis of DNA is 5 prime to three prim and the reading of it is 3 prime to 5 prime.
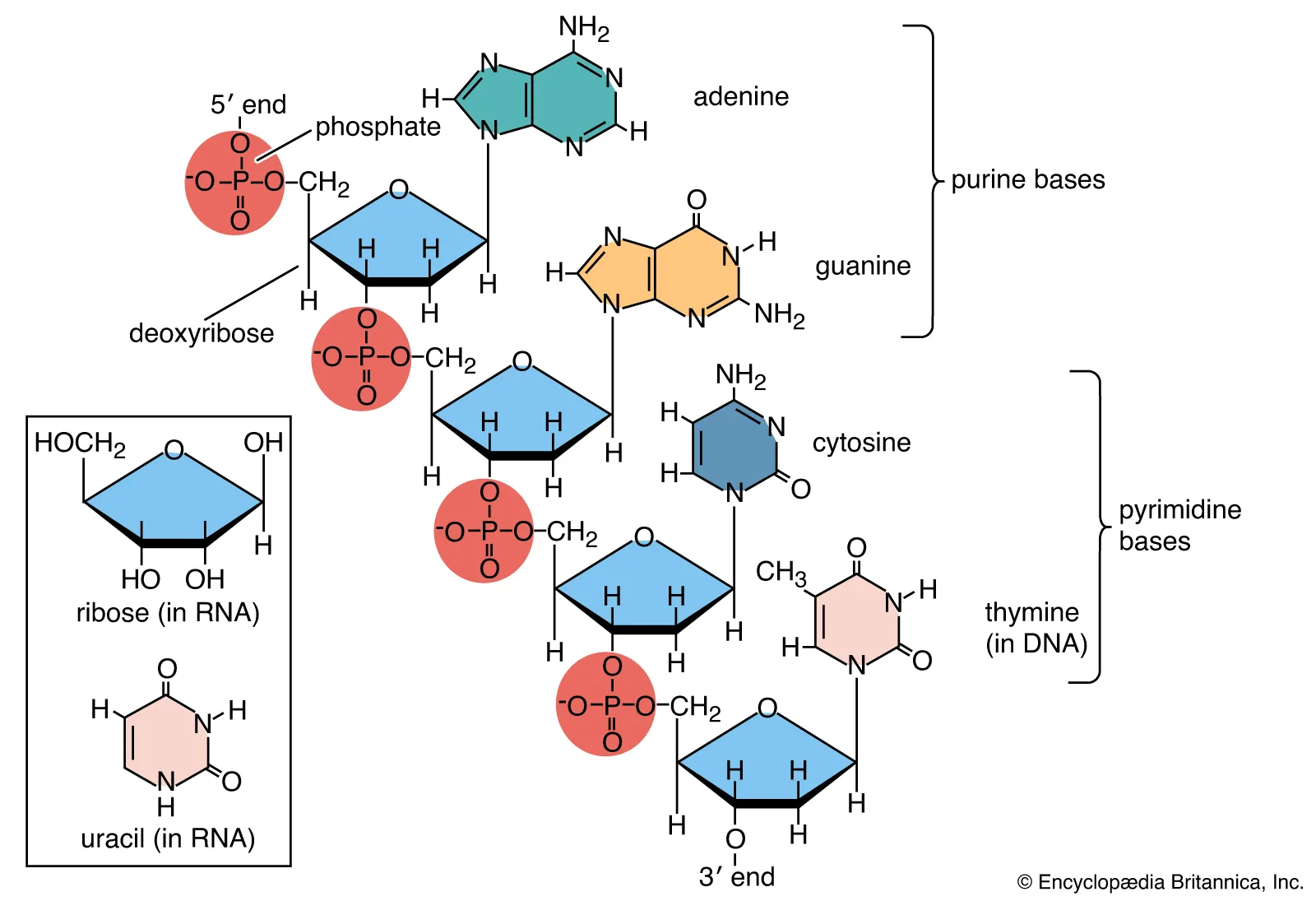
RNA- Ribose( different sugar, single stranded)
DNA- Deoxyribose( different sugar, double stranded)
Proteins
are made up Carbon, Hydrogen, Oxygen and Sulfur. It has different groups like a hydrogen an a R group and two ends an amino group and a carboxyl group. When amino acids come together the amino group binds to the carboxyl group.
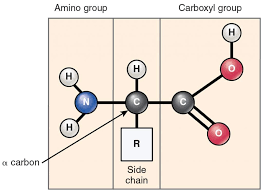
Monomer: Amino Acid
There are 20 different R groups which make up all 20 different amino acids.
R group
Hydrophilic- folds exterior
Hydrophobic- folds interior
Charged- folds exterior
Bond: Peptide Bond( N terminus amine group and C terminus carboxyl group)
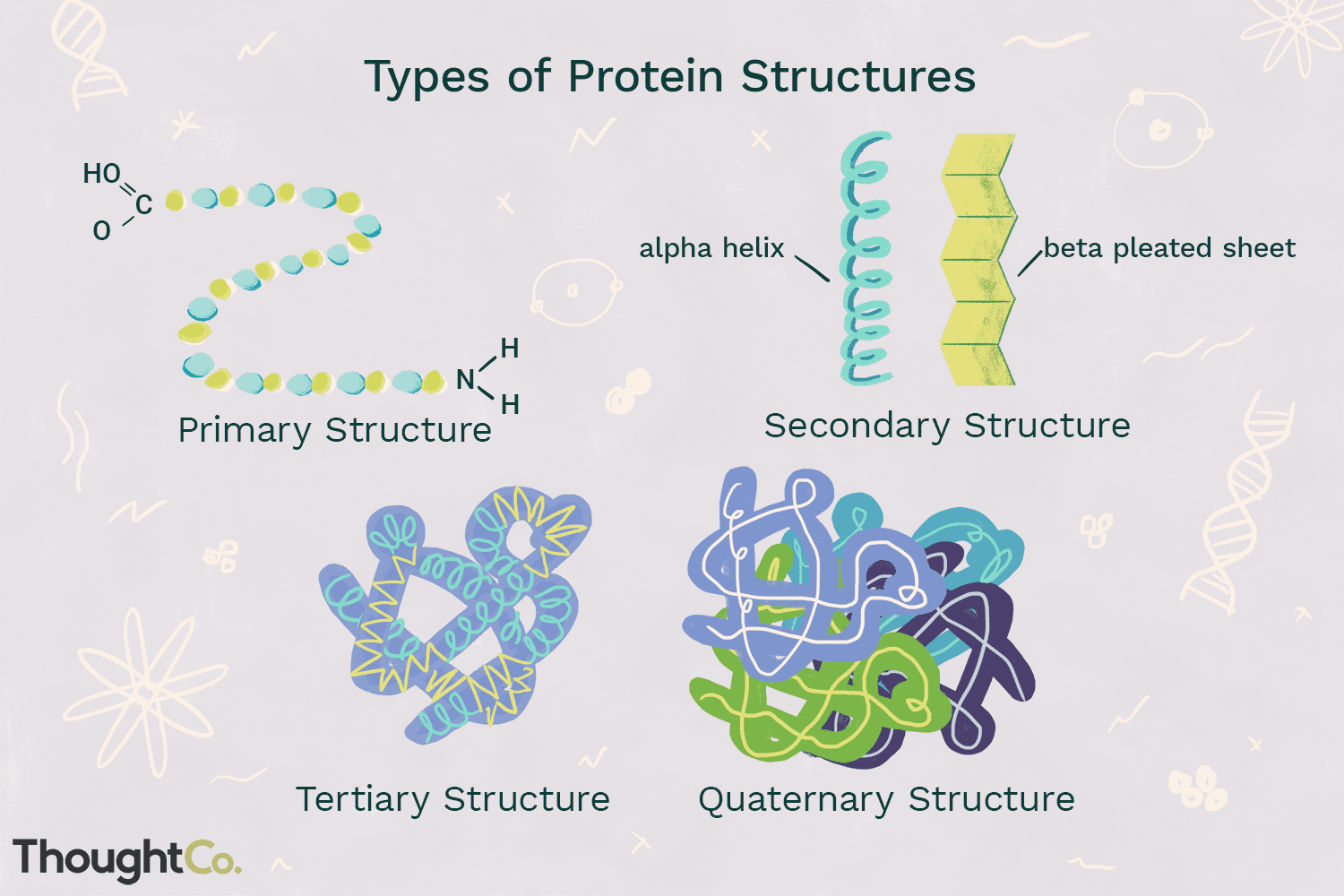
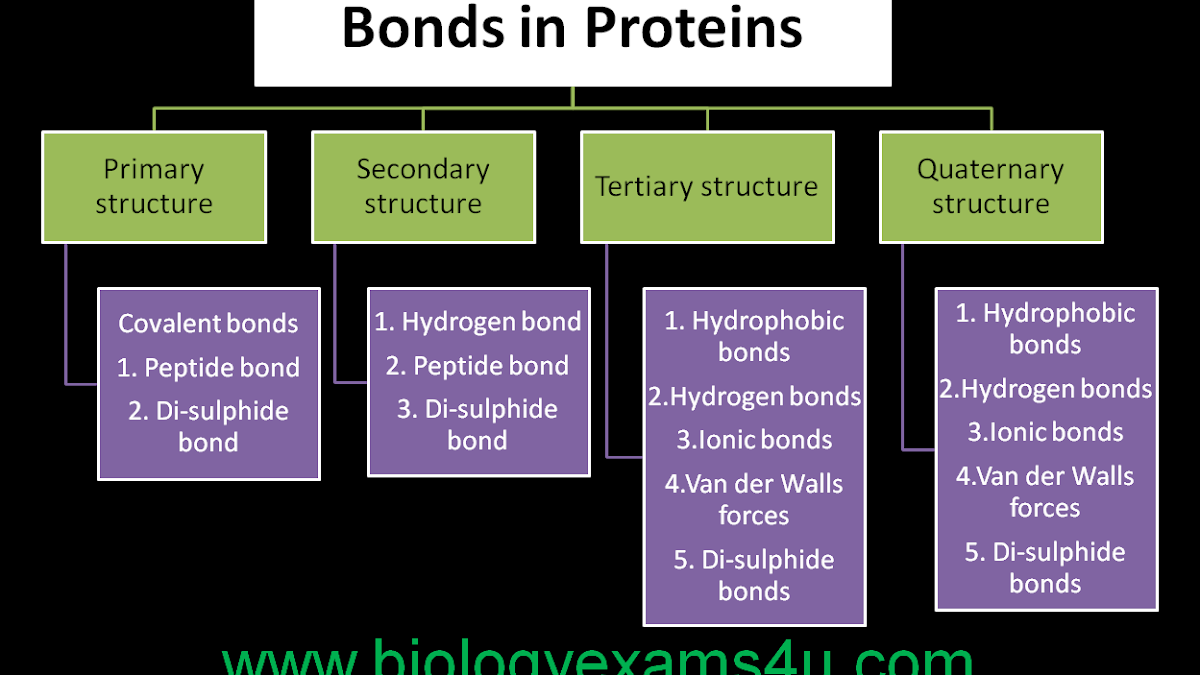
Proteins have four different structures
Primary- which is the peptide bond between amino acids. its a string of amino acids. Bonds and forces included are: Peptide bond
Secondary- Bonded by hydrogen bonds between the backbone and there is also alpha helix or beta pleated sheets which folds the protein, amino acids.
Alpha helix- curl
Beta sheet- waves
Tertiary: Bonded by hydrogen bonds, hydrophobic interaction, ionic interaction or disulfide bridges between R groups. 3D shape starts to form. Hydrophobic ions come together and move away from water.
Quaternary: Bonded by hydrogen bonds, covalent, Van Der Waals interactions, ionic interactions
Unit 2
Cell Structure and Function
A cell has many different organelles and it’s composed of a cell membrane, Smooth and Rough ER, Ribosomes, Mitochondria,Golgi Complex, Vacuole, Lysosomes, Nucleus and Chloroplast, and cell wall.
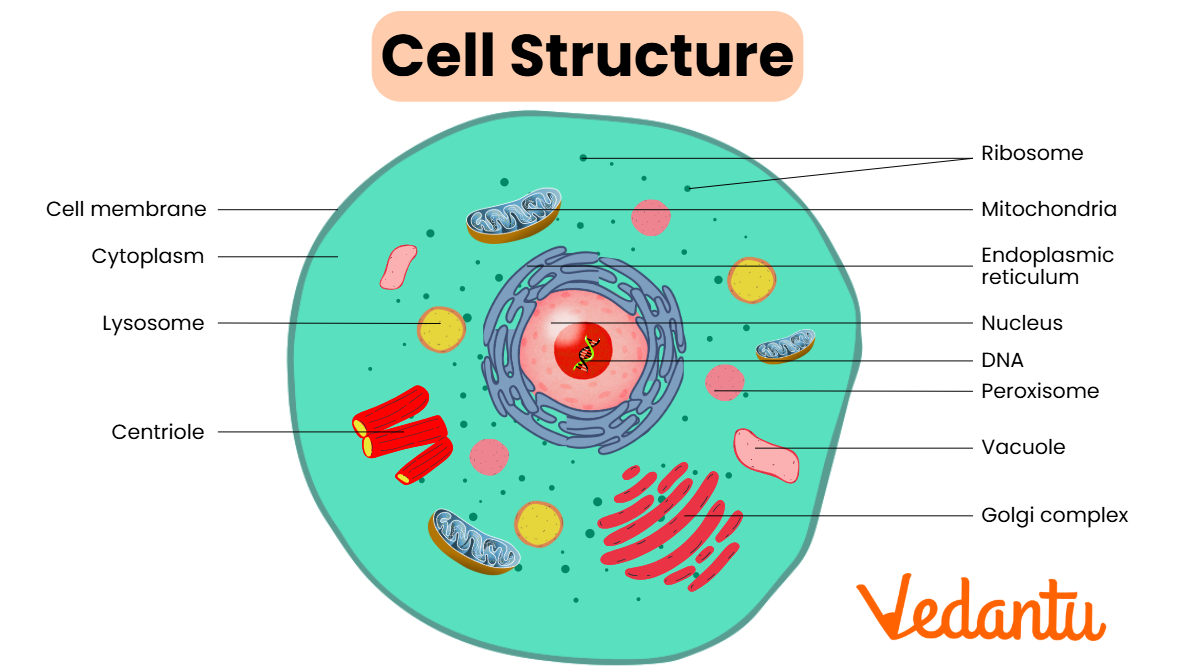
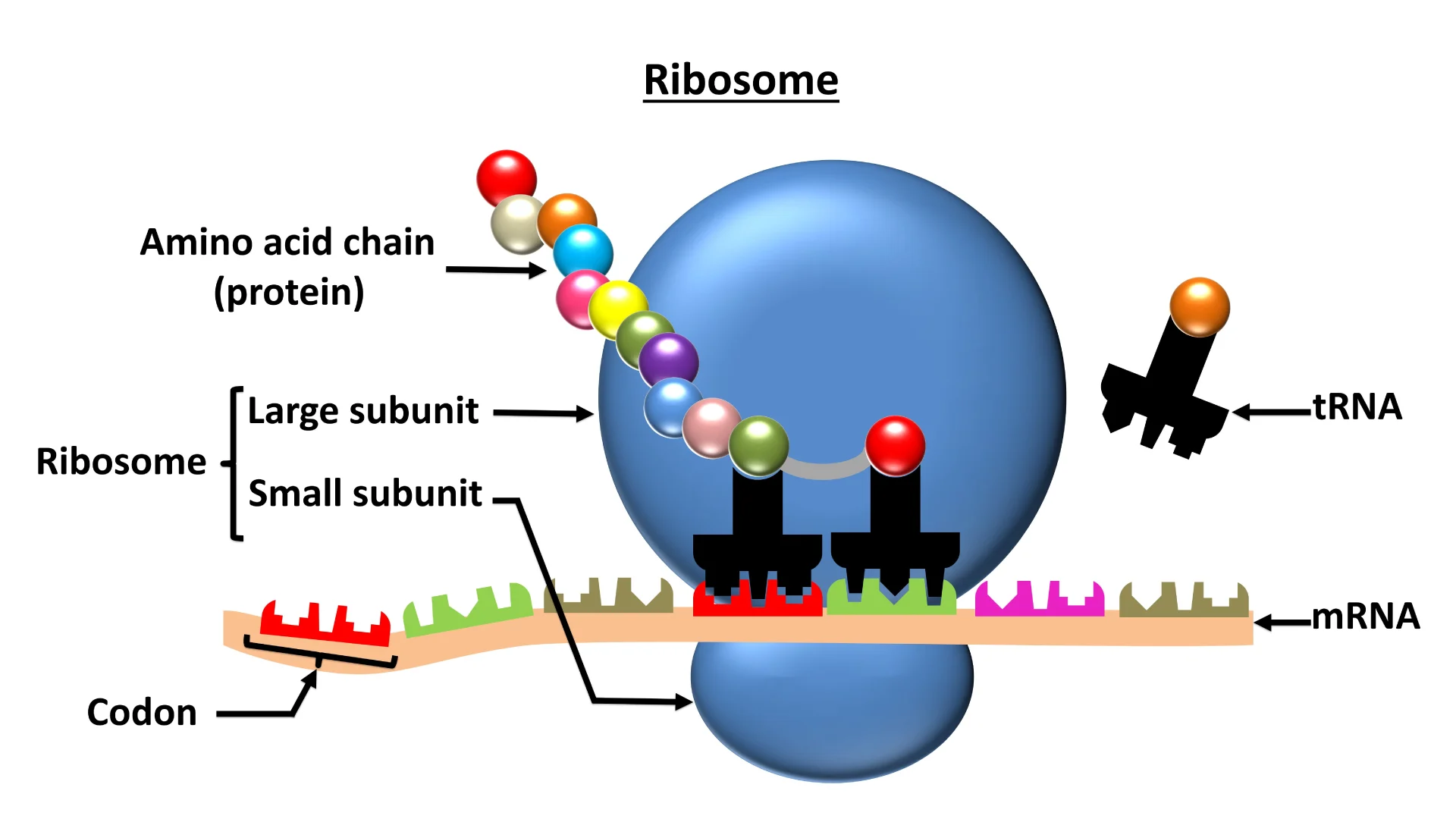
Ribosomes- are made up of ribosomal RNA (rRNA) and proteins non-membrane, sub cellular structures.
Free ribosomes: in cytosol and
Membrane bound ribosome: in the Rough ER
Ribosomes have a large subunit ( binding site of tRNA) and small subunit (binding site of rRNA)
Role: is to make proteins according to messenger RNA (mRNA) sequences.
tRNA brings the different amino acids to the ribosomes to make a protein. ( bringing the ingredients)
mRNA is the messenger RNA it brings the information from the nucleus out to be synthesized.
Found: in all cells and all forms of life and these reflect the evidence in common ancestry in all known life. Both Eukaryotic and Prokaryotic cells have them.
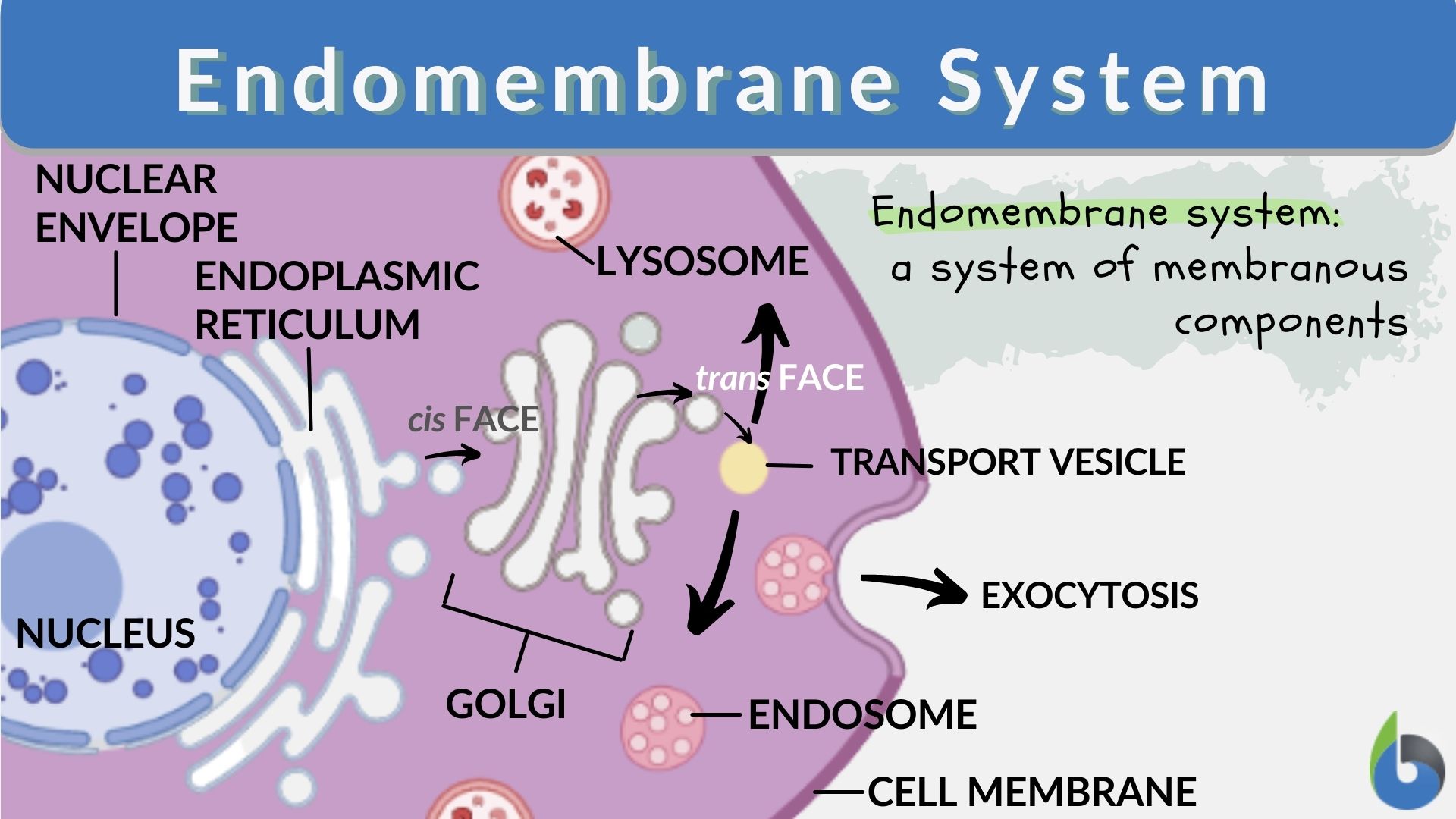
Endomembrane system is a group of membrane-bound organelles that are attached based on a membrane.
This consists of: Endoplasmicreticulium, Golgi complex, lysosomes, vacuoles, transport vesicles, nuclear envelope and plasma membrane.
These work together to modify, build, package and transport, lipids polysaccharide and proteins intercellularly.
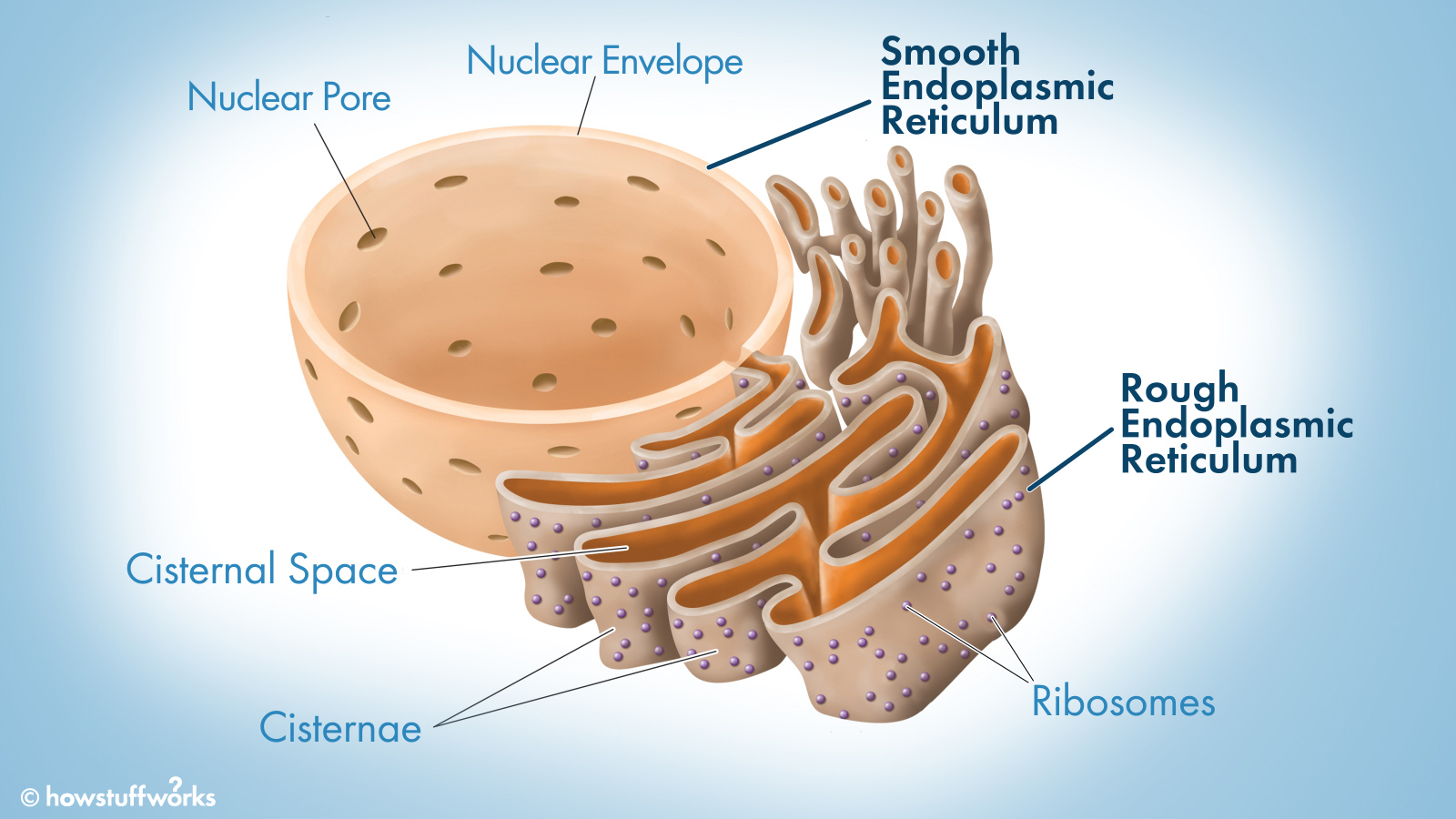
Endoplasmic reticulum: Helps maintain shape and structure in the cell and also allows for transport of materials.
Rough: has ribosomes stuck to it and is responsible for protein synthesis.
Smooth: detoxification of cells and also lipid synthesis.
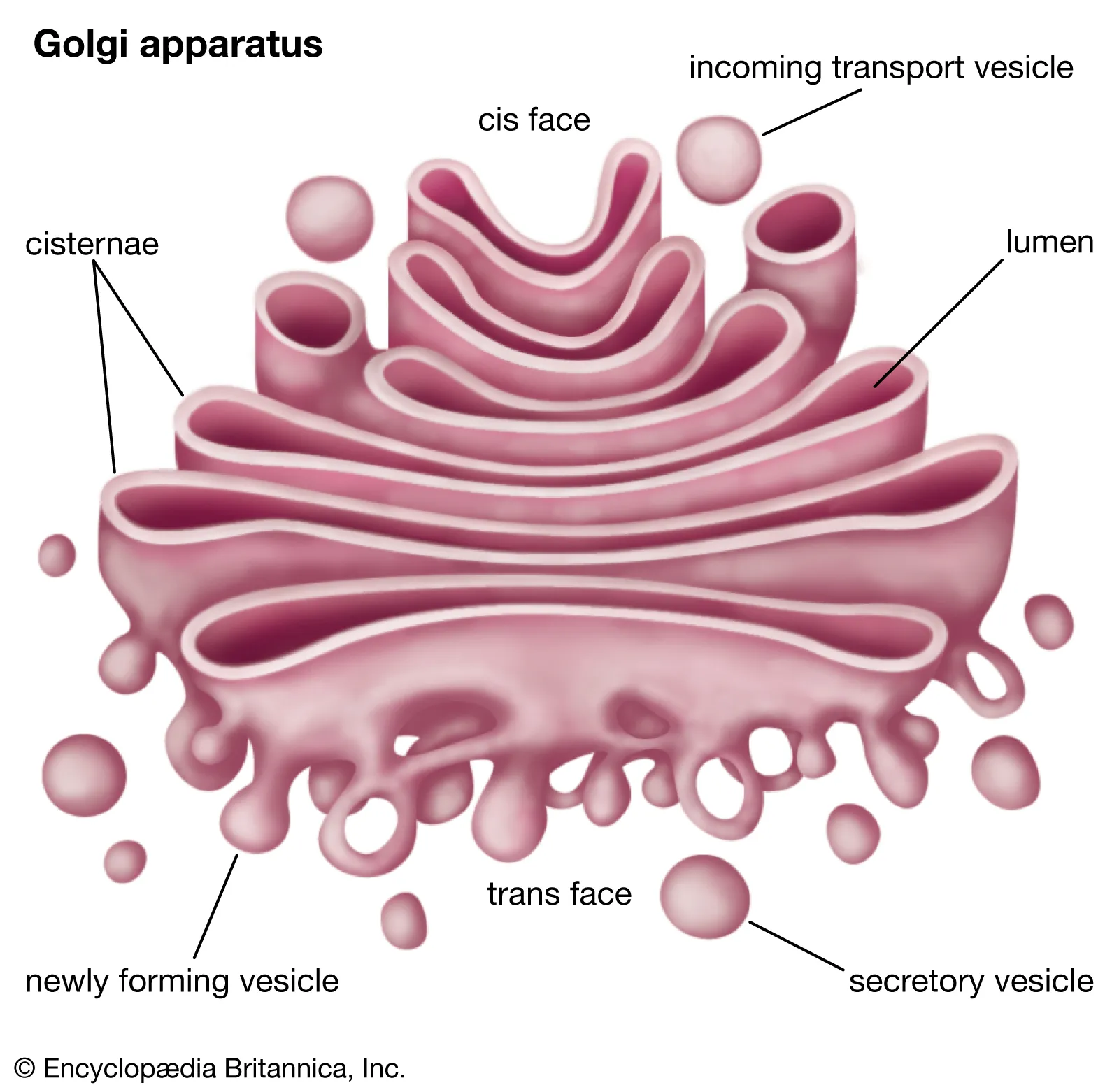
Golgi complex: highly folded membrane sacks. This shows high surface area.
Function: modifies and packages proteins into a vesicle for out the cell or where ever it’s needed.
Components
Cis face: near nucleus where materials come in
Trans face: where material come out
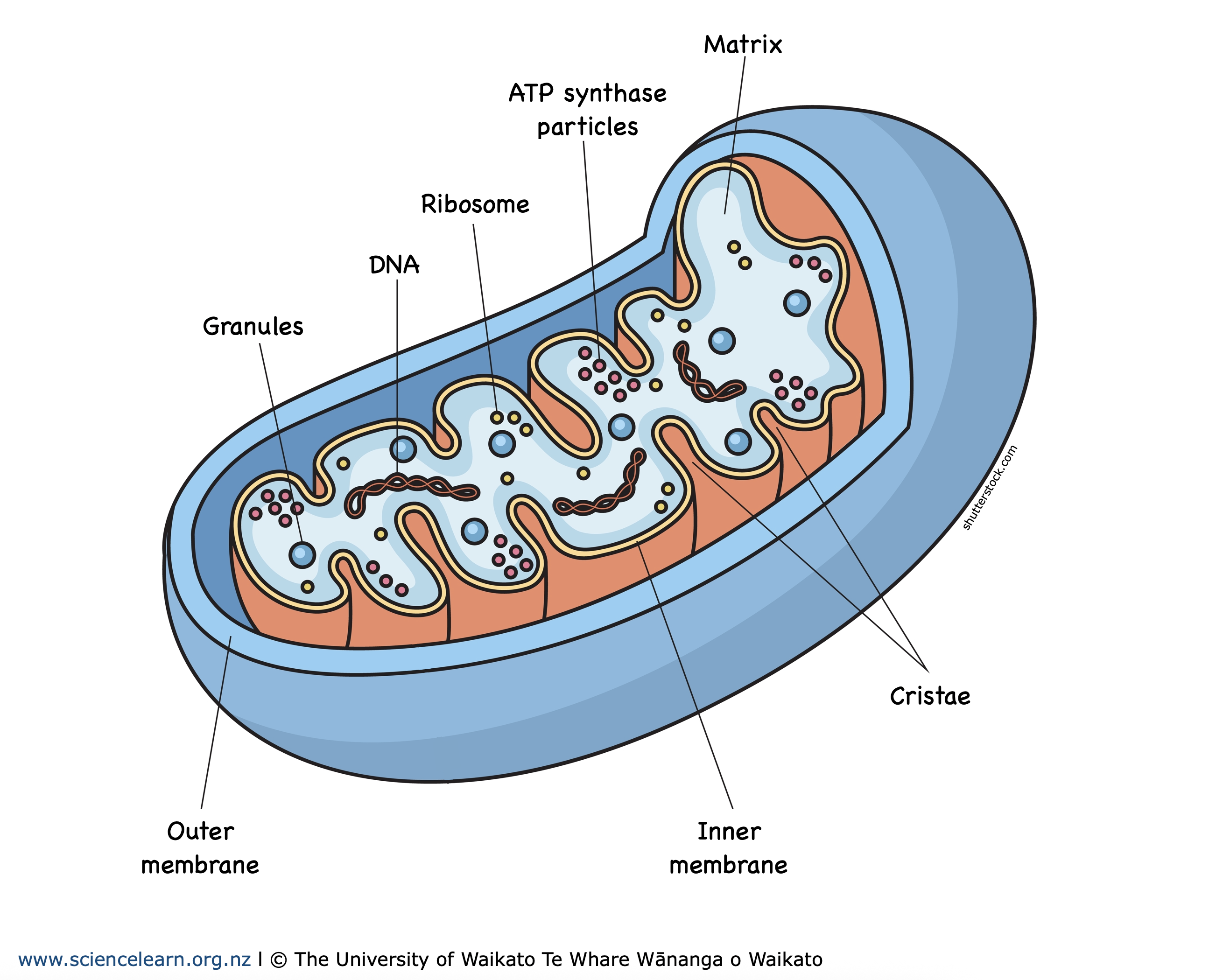
Mitochondria: Power house of the cell. Helps us with providing Energy and ATP for the cell to go through metabolic reactions. Higher Surface area because it’s highly folded membrane.
Originally: a prokaryotic
Structure:
Outer Membrane
Inner Membrane: is the Cristae; highly folded and it allows it so synthesis more ATP
Ribosomes: it’s own Ribosomes to make proteins
Circular DNA : it has its own DNA
Matrix :Kerbs cycle
Inter membrane Space
Cristae
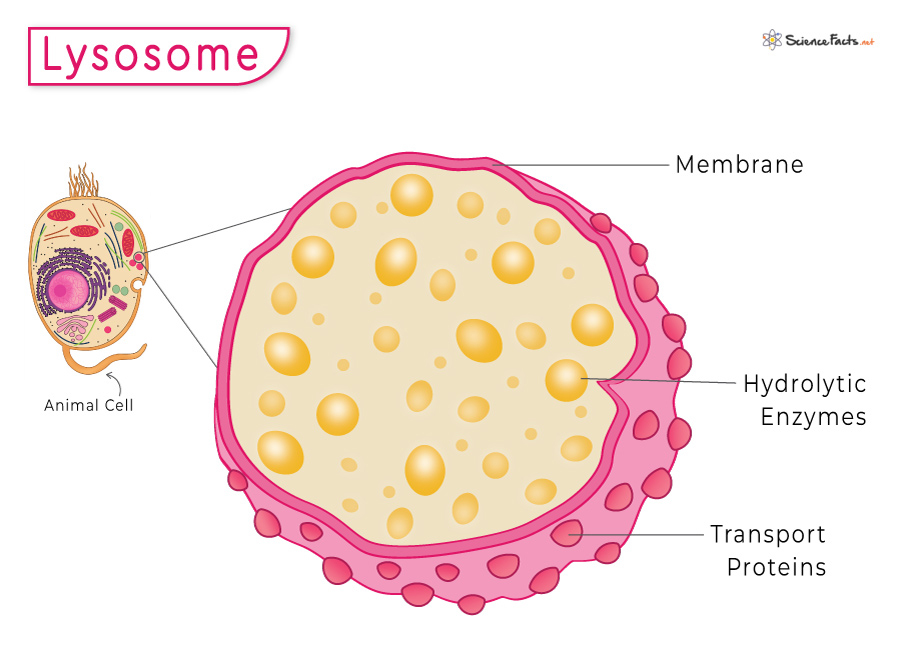
Lysosomes: membrane- bound organelle, has hydrolytic enzymes inside that break down things and has transport proteins, made from Golgi complex
Role/ function: get rid of waste, also apoptosis.
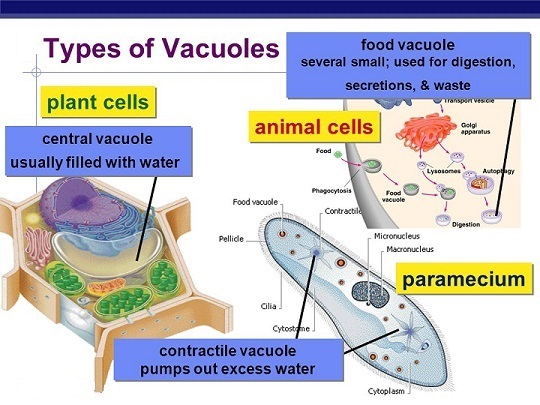
Vacuoles: membrane- bound sacs. Found in both animal cells and plants cells but more in plant cells. Smaller in animal cells and Larger in plant cells.
Central vacuole: found in plants cells and allows for maintaining turgor pressure allowing the cell to take in water and nutrients. Applies force to the cell wall.
Contractile vacuole: maintains osmoregulation in some single- felled organisms. Hypotonic environment where water rushes inside the cell and this vacuole helps push water out.
Food vacuole: formed from phagocytosis( cellular eating) and fuses with lysosomes.
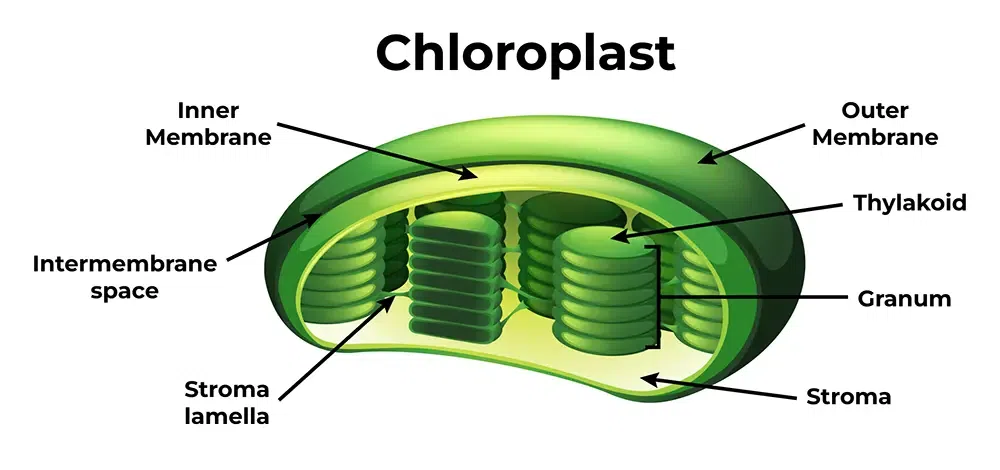
Chloroplast: specialized organelles found in plant cells and photosynthesis algae. It allows them to live and perform photosynthesis by getting food and energy.
Structure: has a double membrane, its own DNA, its own ribosomes, thylakoid sacs called Grana, Stroma and inner and outer membranes. It was once a prokaryotic cell, endosymbiosis
Cell Compartmentalization:
There’s membrane bound organelles found within the cytoplasm that allows to take up space in the cell and allows it to thrive and live.
Rough and smooth ER Golgi body/ complex Lysosomes Mitochondria Chloroplast Vacuole: each have their own jobs. This is why Eukaryotic cells are larger they have organelles that are membrane bound while prokaryotic cells don’t.
Internal Membranes: Help carry out cellular processes by increasing surface area
Mitochondria: Cristae where oxidative phosphorylation happens( makes as much ATP as possible) has high surface area
Chloroplast: Thylakoid where light reaction happens.
Cell size
Surface Area to Volume Ratio affect the obtain of nutrients, the elimination of waste products, exchange chemical/ energy with environment and acquire/ dissipate heat.
Large surface area and small volume you can work well in the cell and carry out its function by exchanging material within the environment.
Small cells have higher surface area to volume ratio and are more efficient when it comes to exchanging material in and out the cell.
Formulas:

Plasma membrane: made up of phospholipids, these phospholipids are polar and non polar meaning that they are amphipathic, they allow for certain things to come in and out the cell. Since the heads are polar and tails are non polar, polar substances can’t cross the cell membrane due to the tails inside. Only non polar substances can’t cross.
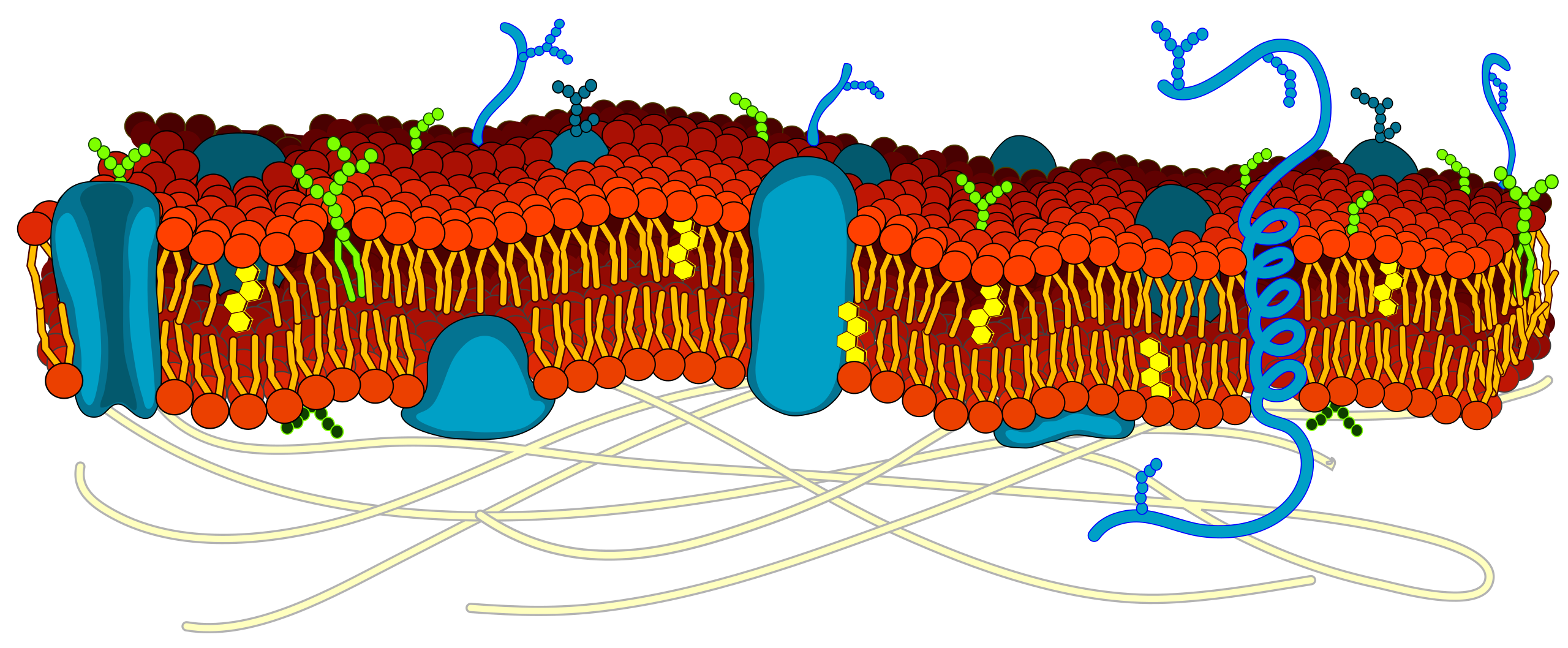
Heads- phosphate: face outside the cell and inside
Tails- fatty acids: face inside of the cell membrane itself.
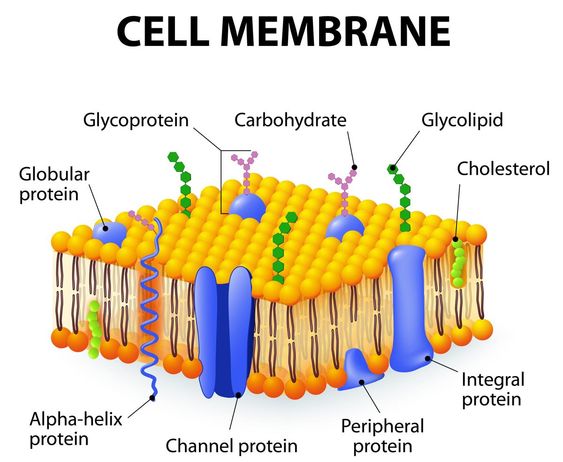
Also made up of membrane proteins, cholesterol, and carbohydrates; glycoprotein and glycolipids
Cholesterol : helps maintain the fluidity of the cell membrane, holds fatty acids together.
Membrane proteins: act as channels or receptors
Glycolipids: attached to lipids
Glycoproteins: attached to proteins
Cell wall: only found in plant cells or bacteria and Fungi which help with turgor pressure
Membrane Transport:
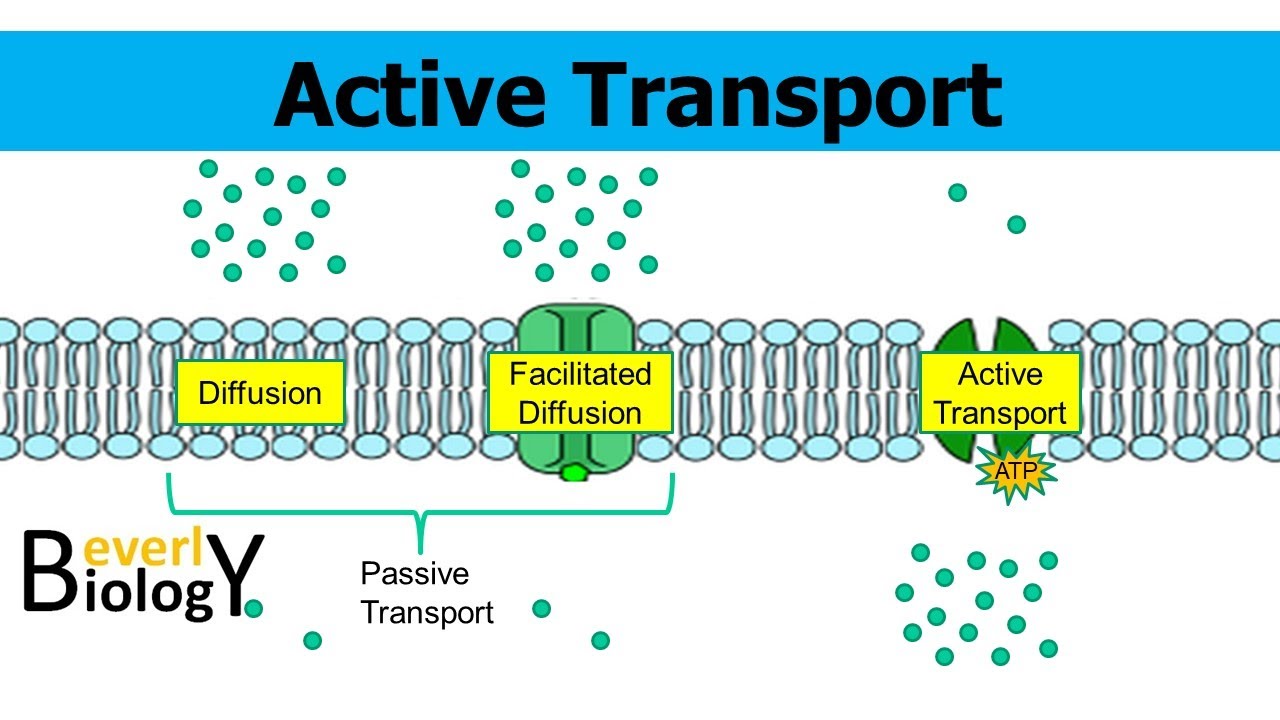
Simple diffusion: no energy required, its passive transport. When water passes through the membrane in tiny amounts or gases. Small or non polar substances, also steroids and hormones.
Facilitated diffusion: glucose or large polar substances can cross through the membrane with the help of a channel or protein without energy. It’s passive transport. Also small molecules. High to low concentration.
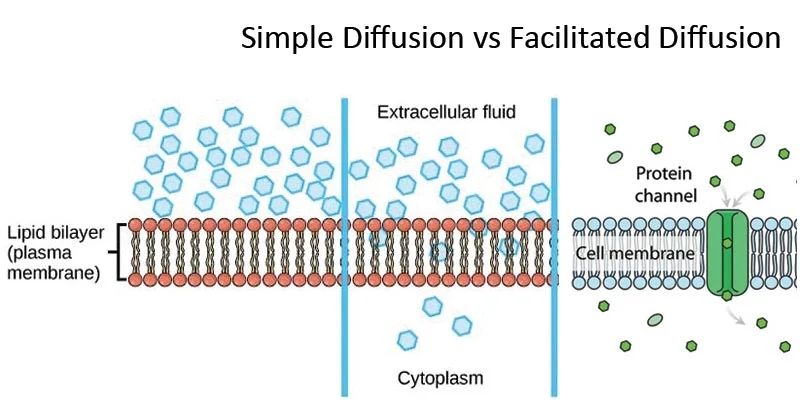
Concentration gradient for passive: high to low concentration
Concentration gradient for active: moves against the concentration low to high.
Active transport: needs energy (ATP) goes across the concentration gradient. Example: sodium and potassium pumps
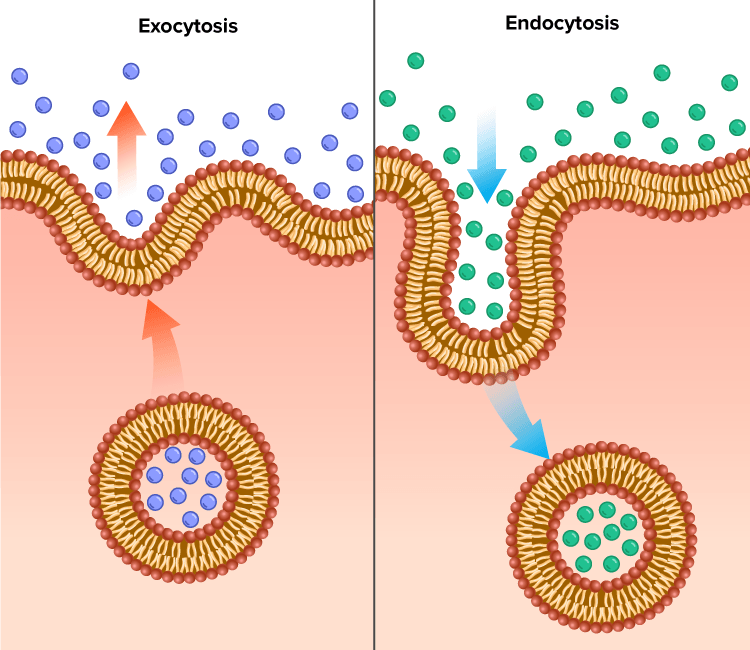
Exocytosis: is when inside vesicles release like materials from the inside of the cell to the outside of it and they do this by fusing with the cell membrane.
Pathway: Rough ER (synthesized), Golgi complex, Transport vesicle and plasma membrane
Endocytosis: the opposite of exocytosis and this is basically just when vesicles fuse with the plasma membrane and they take in Materials from the outside of the cell.
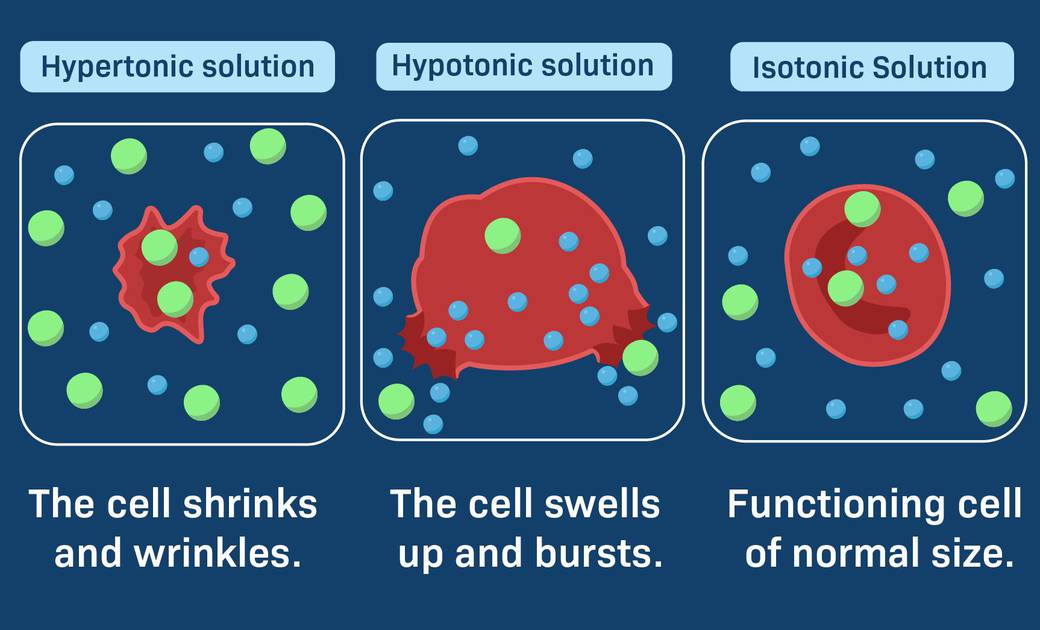
Tonicity and Osmoregulation:
Low solute concentration: more water and less solutes like salt or sugar
High solute concentration: less water and more solutes
Water moves from higher water that’s free to water lower free.
Hypertonic: High solute concentration, lots of free water. Gains water.
Hypotonic: low solute concentration, little free water. Loses water
Isotonic: equal solute concentration and water
Unit 3
Cellular Energetics
Enzymes: Are biological catalyst. They speed up chemical reactions and lower the activation energy for the process. They are specific to substrates, meaning each enzyme only catalyzes a particular reaction or type of reaction. They are proteins are not consumed during the reaction and they have no effect on the change in Gibbs Free Energy. They work non-stop and keep binding to substrates.
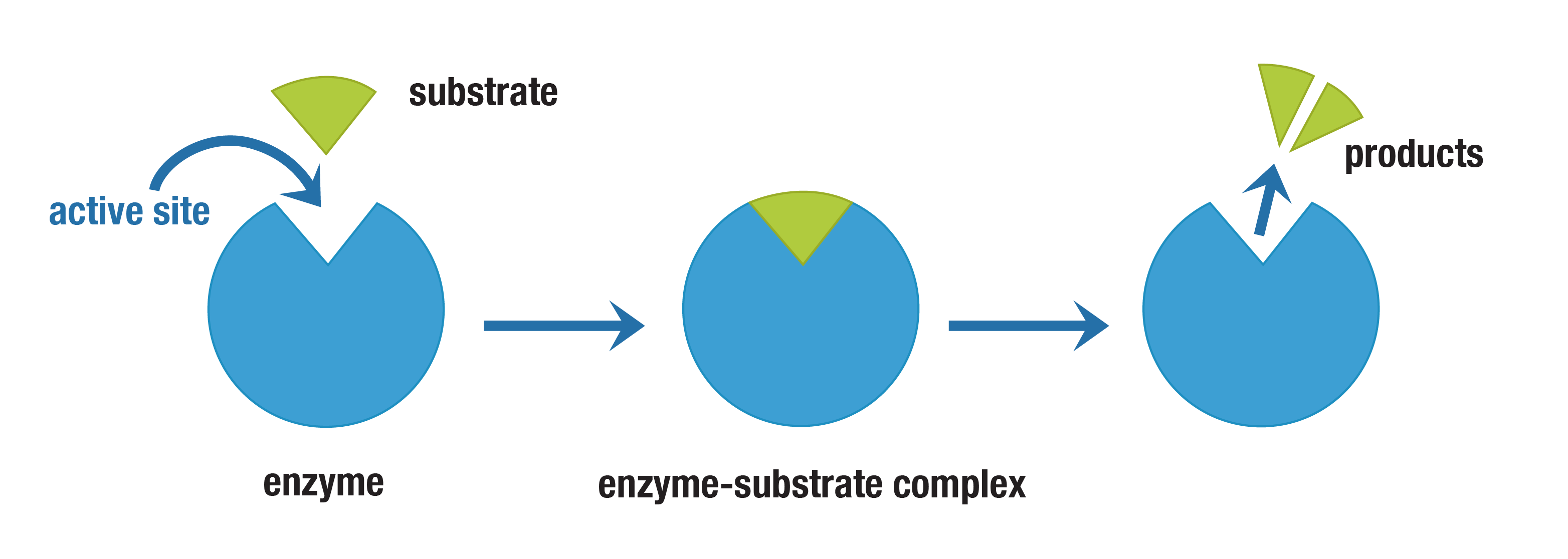
Factors: Enzymes can be affected by several factors including temperature, pH, enzyme concentration, and substrate concentration. Each of these factors can influence the rate of enzyme activity, thereby impacting overall cellular processes. If the pH they are not used to is exposed to them and also a temperature they can denature and come to ruins.
Inhibitors are also another factor. Sometimes they compete for the active site with the substrate and take it up. Another type is non-competitive inhibitors and these bind to the alloestirc site of the enzyme which changes the shape of the enzyme making it unable to bind the substrate.
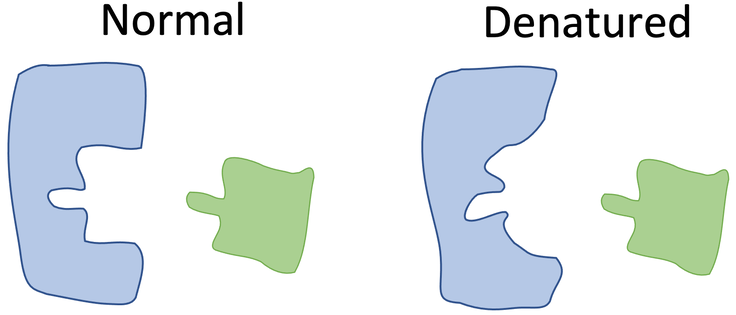
Denaturation: Change in structure of the protein. This happens when the enzyme is exposed to a temp that isn’t the optimal for its activity, resulting in a loss of its functional shape and ultimately preventing it from catalyzing reactions effectively. Different pH and temp messes with H+ ions and this alters the hydrogen bonds that make up the enzyme.
Energy
Endergonic reaction: needs energy is not spontaneous and has a positive free Gibbs energy.
Exergonic reaction: spontaneous, releases energy and has negative Gibbs free energy.
Role of energy: all living things and systems need energy
Photosynthesis: uses Carbon dioxide and water, sunlight to create energy and release oxygen into the atmosphere.
Evolution of it: prokaryotic organisms first started it, Cyanobacteria photosynthesis is responsible for oxygenated atmosphere. Prokaryotic photosynthesis pathway was the foundation for eukaryotic photosynthesis.
Organelle: Chloroplast.
Thylakoids is where the light dependent reaction happens. Where water is broken down and oxygen is released. NADPH and ATP are also produced for the light independent (Calvin cycle) reaction to happen which takes place in the stroma.
Light reaction: Water is broken down, oxygen is released and electrons then pass through the electron transport chain to absorb light and create ATP and NADPH. ATP is made with the help of ATP synthase and H+ ion concentration.
Calvin Cycle:
Takes place in the stroma and carbon dioxide is added to a 5 carbon molecule RUBP using an enzyme rubisco. It’s then broken into 2 sets of a 3 carbon molecule. It uses 6 ATP and 6 NADPH. Then 1 G3P molecules is used to make glucose and then the other is used to regenerate the 5 carbon molecule RuBP.
Cellular Respiration:
Process organism go through to create energy (ATP) using oxygen and glucose.
Organelle: used is mitochondria
First step: Glycolysis: happens in the cytoplasm. A glucose molecule is broke down into two pyruvate acids; which is a 3 carbon molecule structure. 2 ATP molecules and 2 NADH are also created
Second step: a pyruvate enters the matrix of the mitochondria and turns into an acetyl CoA and a carbon dioxide that’s released.
Third step: Oxidative Phosphorylation in the electron transport chain which is the mitochondria Cristae. The electron carriers FADH2 and NADH drop off electrons and this helps make ATP using ATPsynthase, with H+ ions. Oxygen acts as an electron acceptor and takes up some of these electrons and hydrogen ions and it makes water.
Chemiosmosis: makes ATP using the gradient.
Fermentation: allows glycolysis to proceed without oxygen and produces organic molecules alcohol and lactic acid. No mitochondria. Using NAD and NADH
Unit 1 Water and its structure Water is composed of hydrogen and oxygen atom (). It is held together by polar covalent bonds because oxygen is significantly more electronegative than hydrogen, leading to an unequal sharing of electrons. This results in a partial negative charge () on the oxygen and partial positive charges () on the hydrogens. Hydrogen bonds form between the partial positive hydrogen of one molecule and the partial negative oxygen of another, giving water its cohesive and adhesive properties.
Macromolecules 1. Dehydration Synthesis: Builds polymers by removing a water molecule. For example, two amino acids join to form a dipeptide by removing an from the amino group and an (hydroxyl group) from the carboxyl group. 2. Hydrolysis: Breaks macromolecules into monomers by adding a water molecule (hydro-water; lysis-break). For example, maltose is hydrolyzed into two glucose molecules. 3. Monomers vs. Polymers: Monomers are the building blocks that make up polymers.
Carbohydrates - Composition: Carbon, Hydrogen, and Oxygen in a ratio (). - Function: Primary source of short-term energy storage and structural support (cell walls). - Monomer: Monosaccharide (e.g., Glucose, Fructose). - Disaccharides: Sucrose and Lactose (formed via dehydration reaction). - Polysaccharides: - Starch: Energy storage in plants (linear components). - Glycogen: Energy storage in animals (highly branched). - Cellulose: Structural component of plant cell walls. - Chitin: Found in fungal cell walls and arthropod exoskeletons. - Bond: Glycosidic Linkage (covalent).
Lipids - Composition: Carbon, Hydrogen, and Oxygen (and Phosphorus in phospholipids). - Note: Lipids are not true polymers because they lack a repeating monomeric unit. - Types: - Fats (Triglycerides): Long-term energy storage and insulation. Consist of glycerol and three fatty acids. - Saturated Fatty Acids: All single bonds; carbons are saturated with hydrogen. Solid at room temperature (e.g., butter). - Unsaturated Fatty Acids: Contain at least one double bond, causing a 'kink'. Liquid at room temperature (e.g., oil). - Phospholipids: Amphipathic molecules (polar head, non-polar tail) that form the cell membrane. - Steroids: Four-fused ring structures like cholesterol and hormones.
Nucleic Acids - Composition: C, H, O, N, and P. - Monomer: Nucleotide (Sugar, Phosphate, Nitrogenous Base). - Bases: - Purines: Adenine () and Guanine () (Double ring). - Pyrimidines: Cytosine (), Thymine ( - DNA), and Uracil ( - RNA) (Single ring). - Pairing: In DNA, pairs with ( hydrogen bonds), and pairs with ( hydrogen bonds). This makes pairs harder to break.
Proteins - Composition: C, H, O, N, and Sulfur (). - Monomer: Amino acids (containing an amino group (), a carboxyl group (), and a variable group). - Structure: - Primary: Sequence of amino acids held by peptide bonds. - Secondary: -helices and -pleated sheets (hydrogen bonds). - Tertiary: Overall 3D fold ( group interactions). - Quaternary: Multiple polypeptide chains joining.
Unit 2 Cell Structure and Function Endomembrane System Consists of the nuclear envelope, ER, Golgi complex, lysosomes, vacuoles, and plasma membrane. They work together to package and transport proteins and lipids. - Endoplasmic Reticulum (ER): - Rough ER: Studded with ribosomes; site of protein synthesis. - Smooth ER: Detoxification and lipid synthesis. - Golgi Complex: Modifies, sorts, and packages proteins for secretion. - Mitochondria: Site of ATP production via cellular respiration. Evidence for Endosymbiotic Theory: Contains circular DNA, ribosomes, and a double membrane. - Chloroplast: Site of photosynthesis in plants and algae. Also contains its own DNA and ribosomes. - Lysosomes: Contain hydrolytic enzymes for intracellular digestion and apoptosis. - Vacuoles: Storage sacs. Plants have a large Central Vacuole for turgor pressure.
Cell