1.4 Ethers
- General formula: R-O-R
Properties of Ethers
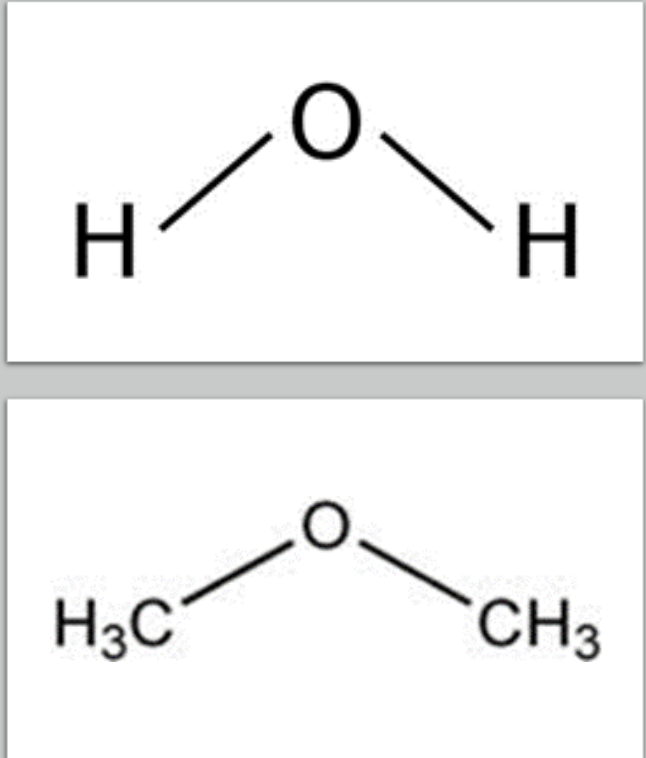
- Like water, ethers take the V shape due to paired electrons on oxygen
- This V shape makes the ether molecules more polar than hydrocarbons, increasing intermolecular attraction between ether molecules compared to hydrocarbons but weaker than alcohols (no H-bonds)
- The boiling points of ethers are slightly higher than those of analogous hydrocarbons, but lower than those of alcohols.
* Ethers are good solvents for organic reactions since they mix with polar (due to C – O bond) and nonpolar (due to alkyl groups) substances.
* Dipole-Dipole, not a hydrogen bond!
Condensation/Dehydration Synthesis Reaction
- Prepares ethers from TWO alcohols
- Requires a catalyst (hydrogen)
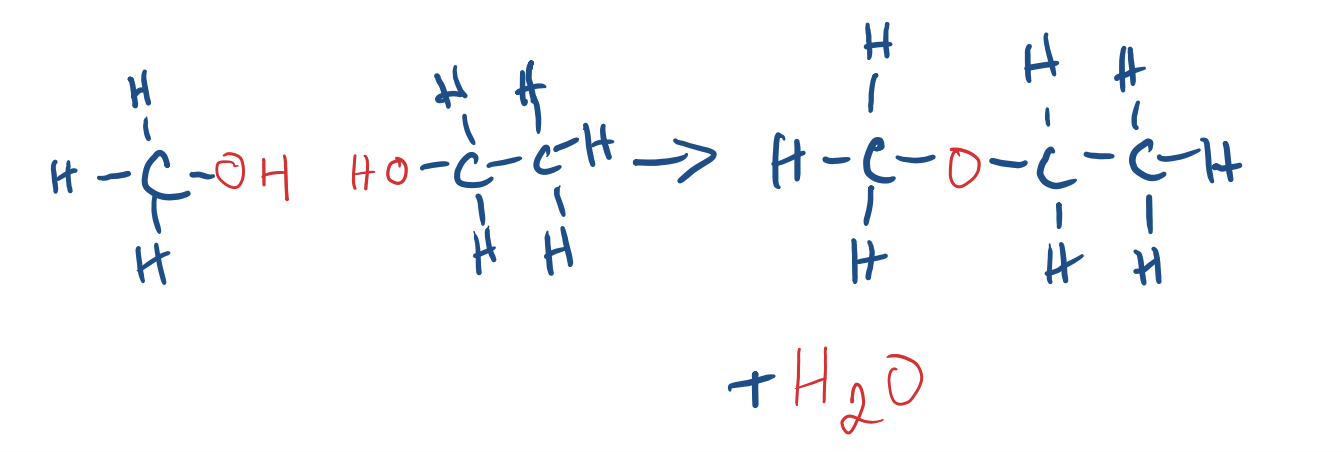
Naming Ethers
- longer chain becomes parent chain (alkane name). Shorter chain get the “oxy” suffix and is treated like a branch.