5.4 Hess' Law
Hess’ Law: the enthalpy change for the conversion of reactants to products is the same whether the conversion occurs in one step or several steps.
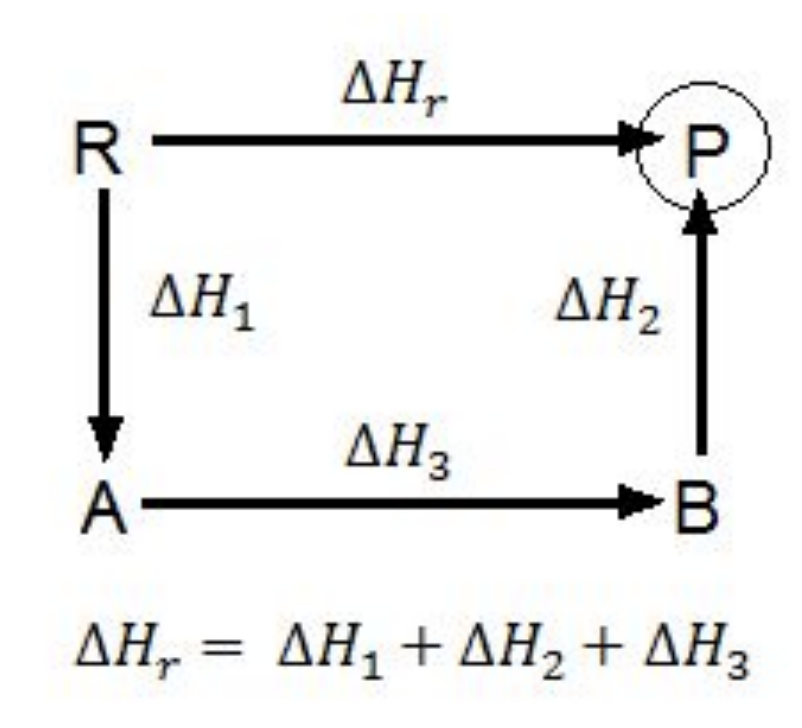
Rules for Enthalpy Changes
- If you reverse a chemical reaction, you must also reverse the sign of ΔH.
- The magnitude of ΔH is directly proportional to the number of moles of reactants and products. If you multiply a balanced equation by a factor, the value of ΔH is multiplied by the same factor.