Movement of particles

Important definitions:
Diffusion: The net movement of particles from an area of high concentration to an area of low concentration down a concentration gradient. It is passive and random
Osmosis: The net passive vacuum movement of water from an area of high water potential to an area of low water potential across a partially permeable membrane until equilibrium is reached.
Active transport: Movement of particles from an area of low concentration to an area of high concentration until the particles have accumulated (built-up) - it requires energy in the form of ATP (chemical energy) which comes from respiration inside mitochondria.
Partially permeable: Some particles can fit through according to size whereas some are too large. The cell membrane in animal and plant cells is partially permeable e.g. small molecules (water) can fit, and large molecules (polysaccharides) cannot fit.
Net: The overall effect such as particles moving from high to low concentration in diffusion
Equilibrium: Even the spread of particles after diffusion
Passive: Process that does not extra energy (particles use their natural kinetic energy to move).
Active: Process that requires extra energy (energy comes from respiration in mitochondria and releases chemical energy called ATP).
Turgid: When plant cells take in water by osmosis and the water pushes against the cell wall – the cell is firm
Flaccid: When a plant cell loses water by osmosis, and the cell contents is reduced – the cell is floppy
Plasmolysed: When a plant cell loses all of its water and the cell membrane comes away from the cell wall
Movement of particles:
Molecules can move in 3 different ways:
• Diffusion
• Osmosis
• Active Transport
Diffusion occurs because:
Particles in a gas or liquid are constantly moving at random due to their kinetic energy
If you have a high concentration of particles in one area then this random movement will cause them to spread out.
They will continue to spread out until they are evenly distributed.
We say they have reached equilibrium
The particles will move at random in all directions but most will move from an area of high concentration to an area of low concentration.
We say there is a net movement of particles from the higher to lower concentration.
The difference between the high and low concentrations is known as the concentration gradient
As you do not need to put more energy into the system the process is passive
The rate of diffusion depends on:
The size and mass of the particles – the smaller the particles the faster they move
The concentration gradient – diffusion is the movement of particles from a high to a low concentration of molecules. The greater the difference in concentration the faster diffusion occurs. We call this difference the concentration gradient - the larger the difference the steeper the gradient.
The temperature – the higher the temperature the more kinetic energy the particles have so the faster they move
Importance of diffusion in living organisms:
Nutrients and oxygen diffuse into living cells and waste products like carbon dioxide diffuse out of the cells through the cell membrane
Carbon dioxide for photosynthesis also diffuses into the leaves through openings called Stomata
Molecules moving across the placenta e.g. oxygen and glucose • Gas exchange at the lungs
Glucose molecules enter the blood in the small intestine during digestion
Surface area to volume ratio:
Oxygen enters a cell by diffusion across the cell membrane
The more membrane the cell has, the more oxygen can diffuse into it
This is the cell’s surface area
However, the amount of O2 needed by the cell depends on its volume
The larger the cell, the more O2 it needs
The larger the ratio, the easier it is for an object cell or organism to take in or give out chemical or heat.

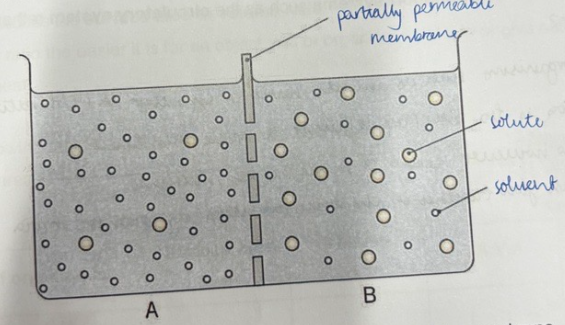
Osmosis:
Osmosis is the diffusion of water across a partially permeable membrane
Distilled water has the highest water potential
This means there is a high concentration of water
A concentrated sugar solution has a low water potential
This means it has a low concentration of water
The water particles are small enough to fit through the holes in the membrane
The sugar particles are too big to move through the membrane.
As in diffusion, there will be a net movement of particles from where there is a high concentration of water (a high water potential) to where there is a low concentration of water (a low water potential)
This will continue until the water potential is equal on either side of the membrane – it has reached equilibrium
Osmosis is passive as it doesn’t require energy
Further osmosis:
Plant and animal cells have a partially permeable membrane – the holes are very small
Water molecules are small and can pass across the membrane
Most solute molecules e.g. sugar or salt, are too large to pass across the membrane
Water moves from a region of high water concentration (water potential) to low water concentration (water potential) across a partially permeable membrane – the solutes cannot move, so the volume of water inside the cell changes
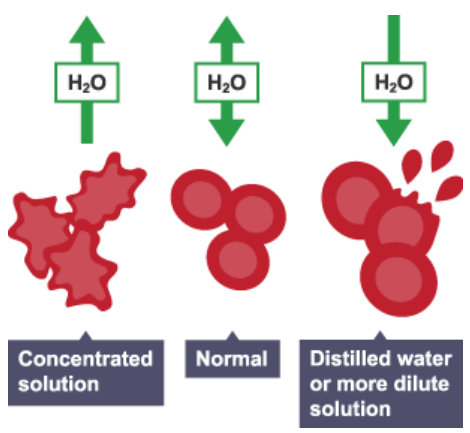
Osmosis in animal cells:
Animal cells have no cell wall, so water moving into the cell can cause the cell to burst (or lyse) – this happens when the animal cell is in a dilute solution
Water moving out of the animal cell can cause the cell to shrink (or crenate) – this happens when the animal cell is in a concentrated salt/sugar solution
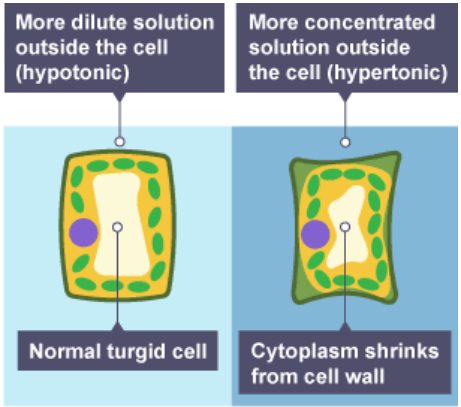
Osmosis in plant cells:
Plant cell walls are freely permeable to all molecules as the holes are big (compared to cell membranes)
Plant cells also have large, sap-filled vacuole that can take in more water
Plant cell in distilled water and a concentrated solution:
Plant cells left in dilute solutions – water moves into the cell, the cell becomes firm or turgid – the cytoplasm and vacuole push against the cell wall
Plant cells left in concentrated salt/sugar solutions – water moves out of the cell, and the cell becomes floppy or flaccid
Plant cells left in very concentrated salt/sugar solutions – so much water moves out of the cell that the cell membrane pulls away from the cell wall – the cell is now plasmolyzed
Importance of osmosis in plant cells:
1. Turgor in plants cells provides support to the green parts of plants.
2. Uptake of water through the root hairs and movement through the root.
Experiment 1:
Observing diffusion in gelatine:
Aim: To investigate the diffusion of substances into a test tube of gelatine. Equipment:
A test tube of 3% gelatine • A test tube of 3% gelatine and universal indicator.
Methylene blue • 0.5M HCl
Two boiling tubes
Method:
Measure 2 cm3 of methylene blue and put it into a boiling tube.
Take a test tube containing only gelatine, invert it and lower it into the boiling tube containing the methylene blue
Measure 2 cm3 of HCl and put it into another boiling tube.
Take a test tube containing gelatine and universal indicator, invert it, and lower it into the boiling tube containing the HCl
Results:
The tube containing methylene blue had the gelatine go blue a little way up with liquid remaining at the bottom
The tube with hydrochloric acid has diffused much more and has travelled further up the tube making it red due to its acidity against the universal indicator
Conclusions:
Diffusion in jelly happens quite slowly
In 48hours the blue dye/acid has moved less than halfway up the tube
The hydrochloric acid diffuses much faster and easier than the methylene blue as it travels further up the tube. The rate of diffusion is slow.
The rate of diffusion depends on the size of the particles, the blue dye diffused more slowly due to its particles being large
WHY?:
The rate of diffusion depends on the size of the particles (the blue dye particles are larger and have moved less)
The temperature has also been quite low, therefore the particles have had less kinetic energy

An experiment to demonstrate osmosis in eggs:
Two raw eggs have had their shells removed by soaking them in acid. This leaves the membrane surrounding the white and yolk intact.
The volume of the eggs will be measured using a displacement can. Egg A will then be put in a beaker of distilled water and Egg B in a beaker of concentrated salt solution.
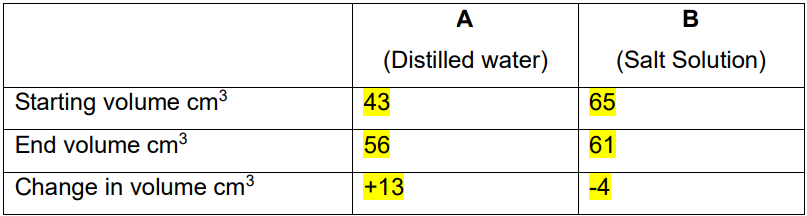
WHAT HAPPENED TO THE EGGS?
EGG A WAS LEFT IN WATER
THIS MEANS THERE WAS MORE WATER IN THE BEAKER THAN INSIDE THE EGG
WATER MOVED INTO THE EGG BY OSMOSIS FROM HIGH WATER POTENTIAL (WATER CONCENTRATION) TO LOW WATER POTENTIAL
THEREFORE, EGGS GAINED MASS
EGG B WAS LEFT IN SALT SOLUTION
THIS MEANS THERE WAS MORE WATER INSIDE THE EGG THAN INSIDE THE BEAKER
WATER MOVED OUT OF THE EGG (HIGH WATER POTENTIAL) AND INTO THE SALT SOLUTION (LOWER WATER POTENTIAL)
THEREFORE, EGG B LOST MASS
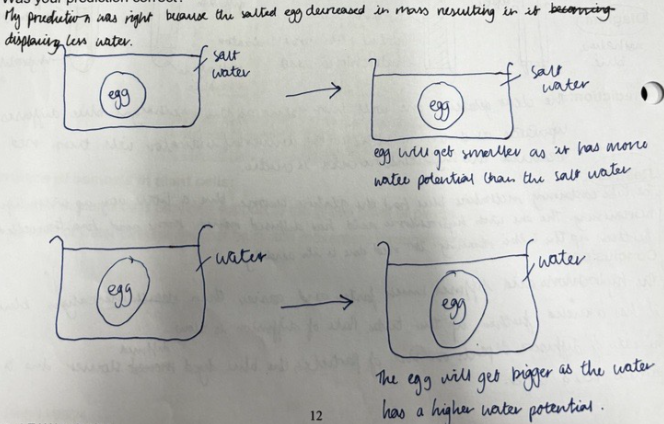
The experiment investigating the rate of osmosis using the Visking tube
Visking tubing is a synthetic partially permeable membrane.
It allows small molecules (e.g. water) to pass through but not large molecules (e.g. sugar)
The apparatus was set up as shown below:
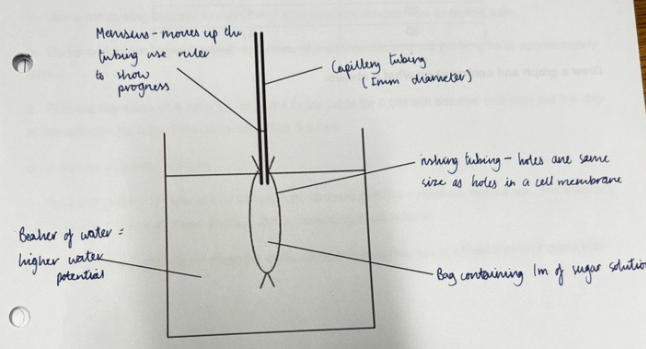
The experiment investigating osmosis in living tissues:
1. Label 6 boiling tubes as follows:
0.0M salt solution (Distilled water)
0.1M salt solution
0.2M salt solution
0.3M salt solution
0.4M salt solution
0.5M salt solution
2. Use a measuring cylinder to add 20cm3 of the correct solution into its boiling tube.
3. Use a cork borer to cut 6 potato cylinders. Cut off the skin and cut the lengths to approximately 3cm.
4. Find out the mass of a cylinder, record it in the table for 0.0M salt solution and then put the chip in the appropriate tube. Repeat for the other 5 tubes.
5. Leave for at least 24 hours.
6. Tip each potato cylinder out of its solution. Do not get the cylinders mixed up. Pat them dry using tissue paper and measure their mass, recording it in the table.
7. Work out the % change in mass for each cylinder (i.e. difference in mass/original mass x100
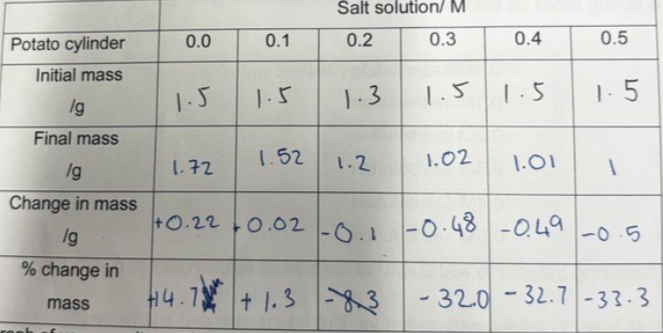
Describe and explain your results using scientific knowledge. Include terms such as water potential, osmosis, and concentration gradient:
The Graph shows the movement of water from an area of high water potential to an area of low water potential using a concentration gradient. Regarding the experiment, it can be categorized as osmosis since the potato cylinders have a partially permeable membrane and water diffusion occurs from the salt solution to the potato cylinder. The less salt is present the higher the water potential.
Why did we work out the percentage change of mass?
In order to make the denomination the same instead of it varies. In this case, make it 100 to work out %.
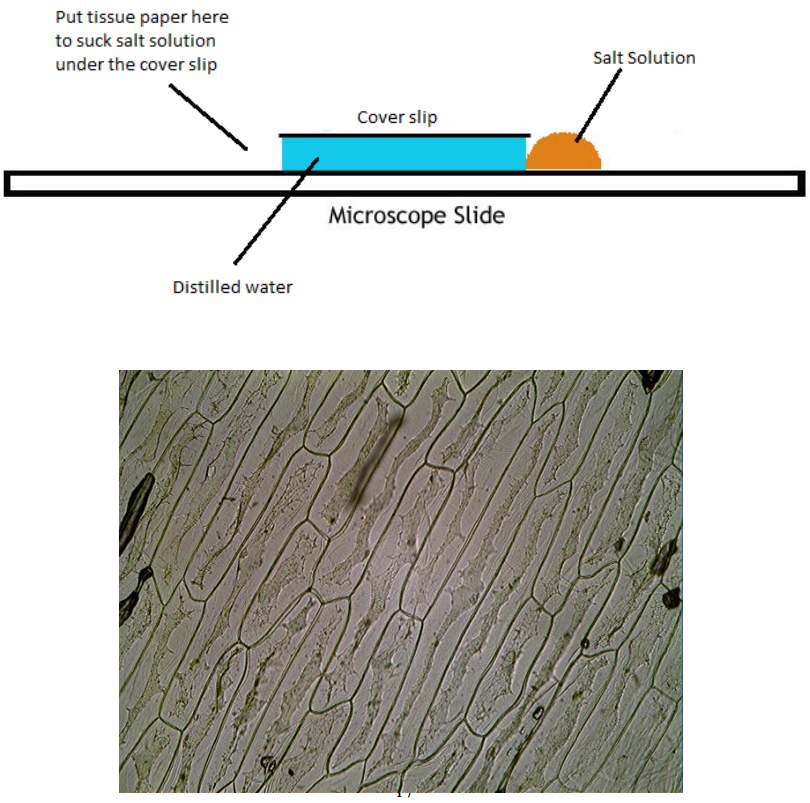
Experiment to demonstrate plasmolysis in onion epidermis
1. Remove a small square of onion epidermis measuring from the inside of one of the fleshy leaves of onion bulb.
2. Using forceps mount this epidermis on a slide and using a pipette add a few drops of water.
3. Gently lower a cover slip over the epidermis using a mounting needle.
4. Remove any excess water off the slide with filter paper.
5. Place the slide on the stage of the microscope and observe with x4 objective. Focus and then view on x10 and x 20 objectives.
6. Draw what you see in one onion cell. Label and annotate.
7. Now irrigate the slide by placing a few drops of concentrated salt solution on one side of the cover slip.
8. Place filter paper on the opposite side of the cover slip and soak up the water under the cover slip so that the salt solution is drawn under it. – as shown below.
Active transport:
The concentration of minerals such as nitrates is normally higher in the roots of the plants than in the water surrounding the soil. Why is this a problem for the plant?
Diffusion occurs from high to low concentration
Diffusion would mean that nitrates would move from the roots back into the soil which is not good for the plant
Nitrates form proteins (proteins are needed for growth)
Active transport is needed to accumulate nitrates in the roots
A plant has to use energy to absorb these minerals, the process is called active transport.
The movement of particles from low to high concentration, against the concentration gradient. This requires energy in the form of ATP.
The energy is provided by respiration. Therefore, cells that carry out active transport will contain a lot of Mitochondria
Why would a plant take up fewer mineral ions if it was in water-logged soil?
Normally, soil particles are surrounded by oxygen, when it is waterlogged, the soil particles are surrounded by water particles and there is not enough oxygen for respiration. There is not enough ATP produced, therefore not enough active transport happening to move the minerals.
Summary:
How do the particles move:
Diffusion: Passively from an area of high to low concentration
Osmosis: Passive through a partially permeable membrane
Active transport: Active
High to low or low to high:
Diffusion: High to low
Osmosis: High to low
Active transport: Low to high
Down or against the concentration gradient:
Diffusion: down
Osmosis: Down
Active transport: Against
Does it require energy:
Diffusion: No
Osmosis: No
Active transport: Yes