Reactions of Alkenes
Electrophilic Addition
π bond in the C=C bond breaks and atoms or groups join the carbon atom, this happens because the C=C bond is rich in electrons and is target for electrophiles
Examples of electrophiles - Polar molecules and halogens
Addition of Halogens
Halogens react with alkenes in an electrophilic addition reaction to form dihaloalkanes
E.g C2H4 + Br2 → C2H4Br2
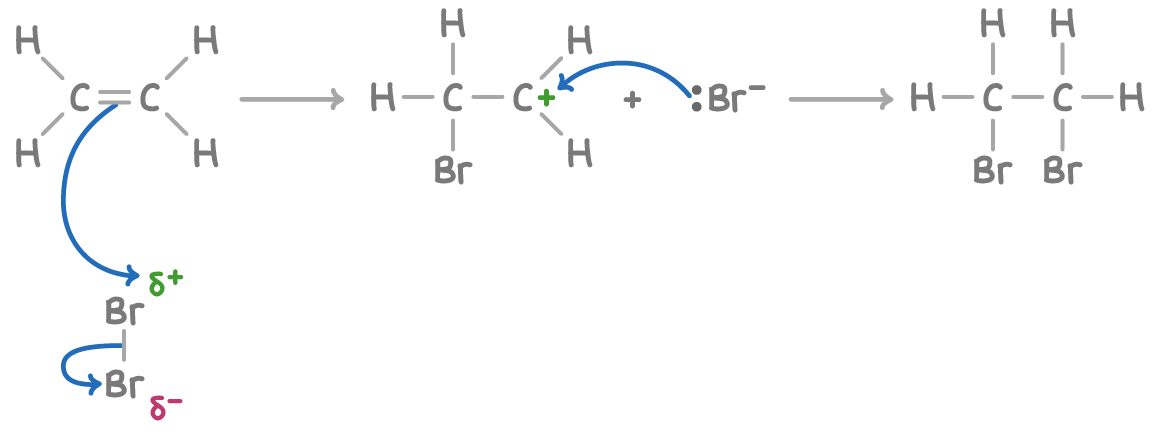
High electron density of C=C bond repels electrons in the bromine molecule, polarising the bond between bromine atoms
Heterolytic fission of the bromine atom, where one atom takes the pair of electrons, the newly created positive bromide ion bonds to one of the C atom
Positive charged carbocation forms, the negatively charged bromide ion joins the positive C ion
The final product, 1,2-dibromoethane has a bromine atom bonded to each of the original alkene carbon atom
Addition of hydration halides
Hydrogen halides like HCl and HBr add across the C=C to form a haloalkane
In symmetrical alkenes the double bonded carbons are identical leading to a single product
In unsymmetrical alkene with two different alkyl groups, hydrogen halides create two isomers, e.g propene forms a mixture of 1-bromopropane and 2-bromopropane, the ratio of the products depend on the stability of the carbocation intermediate
e.g ethene + hydrogen bromide
C2H4 + HBr → C2H5Br
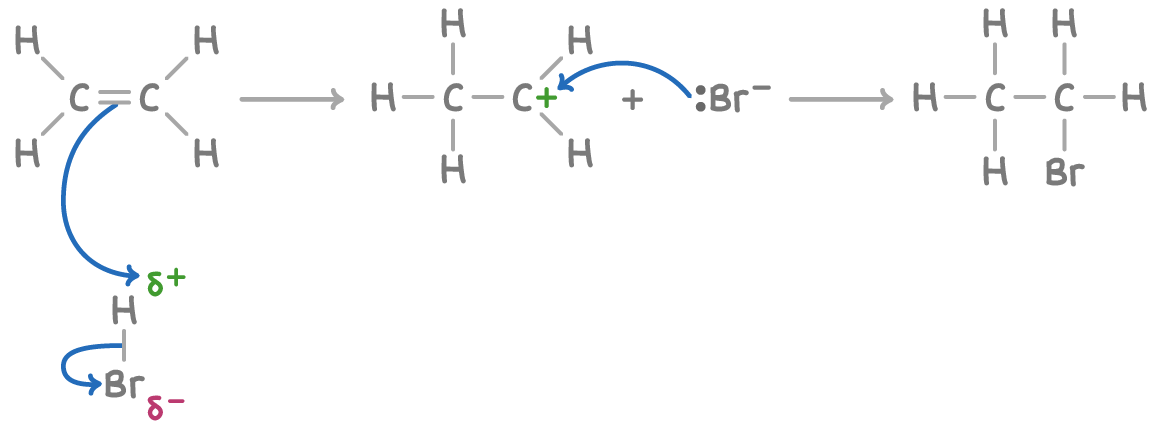
Minor and major products
The major product in a hydrogen halide addition to unsymmetrical alkenes can be predicted by carbocation stabilty
The stability of the carbocation increases with the number of alkyl groups
Primary Carbocations - One alkyl group attached to the positive carbon, least stable
Secondary Carbocations - Two alkyl groups attached to positive carbon (more stable)
Tertiary Carbocations - Three alkyl groups and is the most stable
Most stable carbocation forms the major product. e.g. propene + HBr forms the major product 2-bromopropane
Addition of concentrated sulfuric acid
Cold concentrated H2SO4 adds across the C=C forming an alkyl hydrogen sulfate
E.g ethene + H2SO4 → ethyl hydrogen sulfate
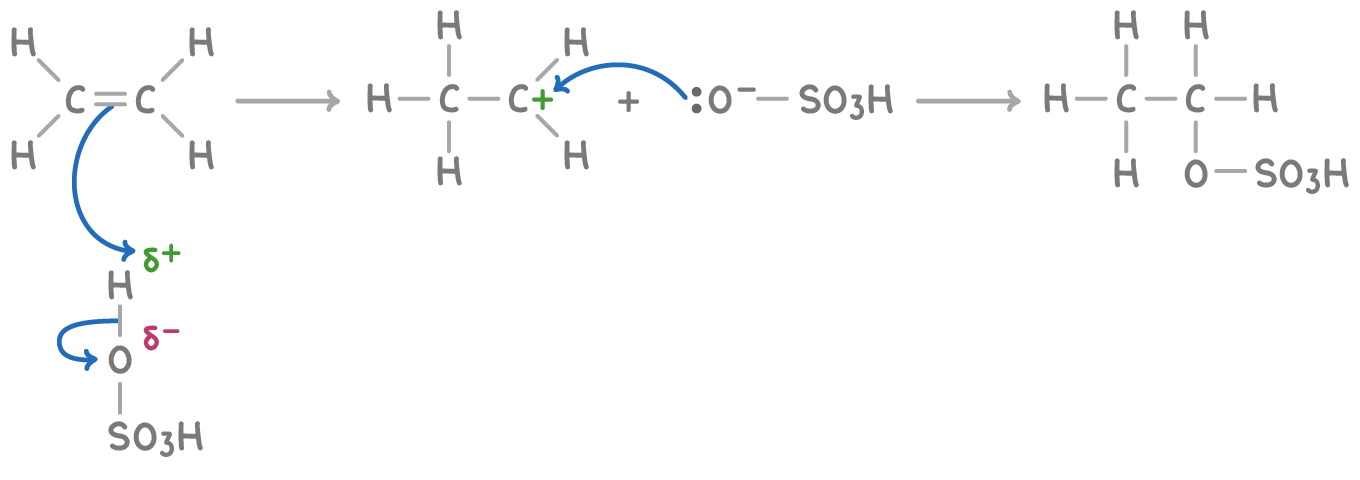
Reaction with unsymmetrical alkenes lead to a mix of products
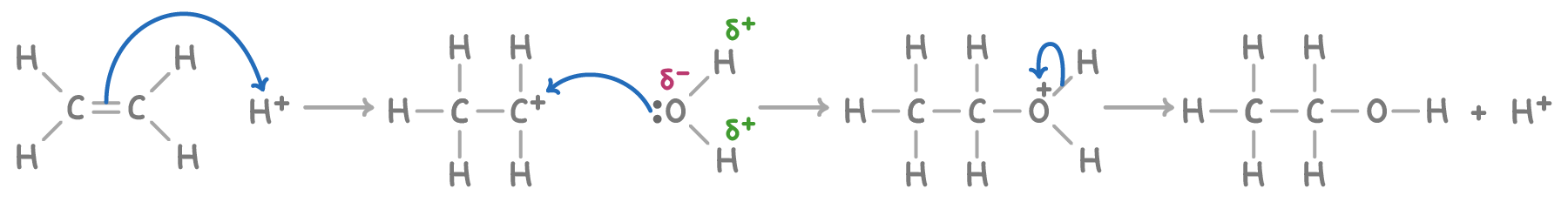
Hydration to form alcohols
Under the influence of a phosphoric acid catalyst, alkenes react with steam at 300°C to form an alcohol
E.g ethene + H2O → ethanol