C4: Chemical Changes
Metal Ores
Metals are found in costal areas
Depending on the reactivity of the metal, they are often chemically combined with another element such as oxygen
We must separate them in order to obtain the pure metal

Acids and Bases
A base is a substance that will neutralise an acid. An alkali is a soluble base.
Not all bases are soluble
HCl | Hydrochloric acid | Chloride |
HNO3 | Nitric acid | Nitrate |
H2PO4 | Sulphuric acid | Sulphate |
H3PO4 | Phosphoric acid | Phosphate |
Acid + Base —> Salt + water
Acid + Metal —> Salt + Hydrogen
Acid + Any Metal Carbonate —> Salt + carbon dioxide + water
e.g. Hydrochloric Acid + Sodium Hydroxide —> Sodium Chloride + Water
e.g. Nitric Acid + Calcium Carbonate + Calcium Nitrate + Carbon Dioxide + Water
Required Salt Practical
Independent variable - the one you change
Dependent variable - the one you measure / record
Control variables - the ones you keep the same - make sure test is valid
Method
Measure 20ml of 0.5M HCl into a measuring cylinder and pour into a beaker
Weigh 5g of Sodium Oxide into the beaker and stir until no more can be dissolved
Heat gently with a Bunsen burner, then remove solution
Filter solution into an evaporating dish and place back on the heat
Heat solution until 2/3 has evaporated
Remove dish from heat and wait until it has evaporated at room temp
Dry crystals with a paper towel
Strong and Weak Acids
The formation of the H+ ions is the reason something will be acidic
pH = potential of H+ the H+ ions only form when the acid is dissolved in water, called an aqueous solution
e.g. HCl —> H+ + Cl
The formation of OH- ions in water is the reason something will be alkaline
The OH- ions only form when the base is dissolved in water, called an aqueous solution
e.g. H+ + OH- —> H2O
Electrolysis
Breaks down ionic compound (salts) into their elements using electricity
The solution need to be molten so the ion can move to the electrodes
Ions must be free to move, for current to flow
Electrical current = movement of charge
Positive electrode = Anode
Negative electrode = Cathode
Positive Anode Negative Is Cathode - PANIC
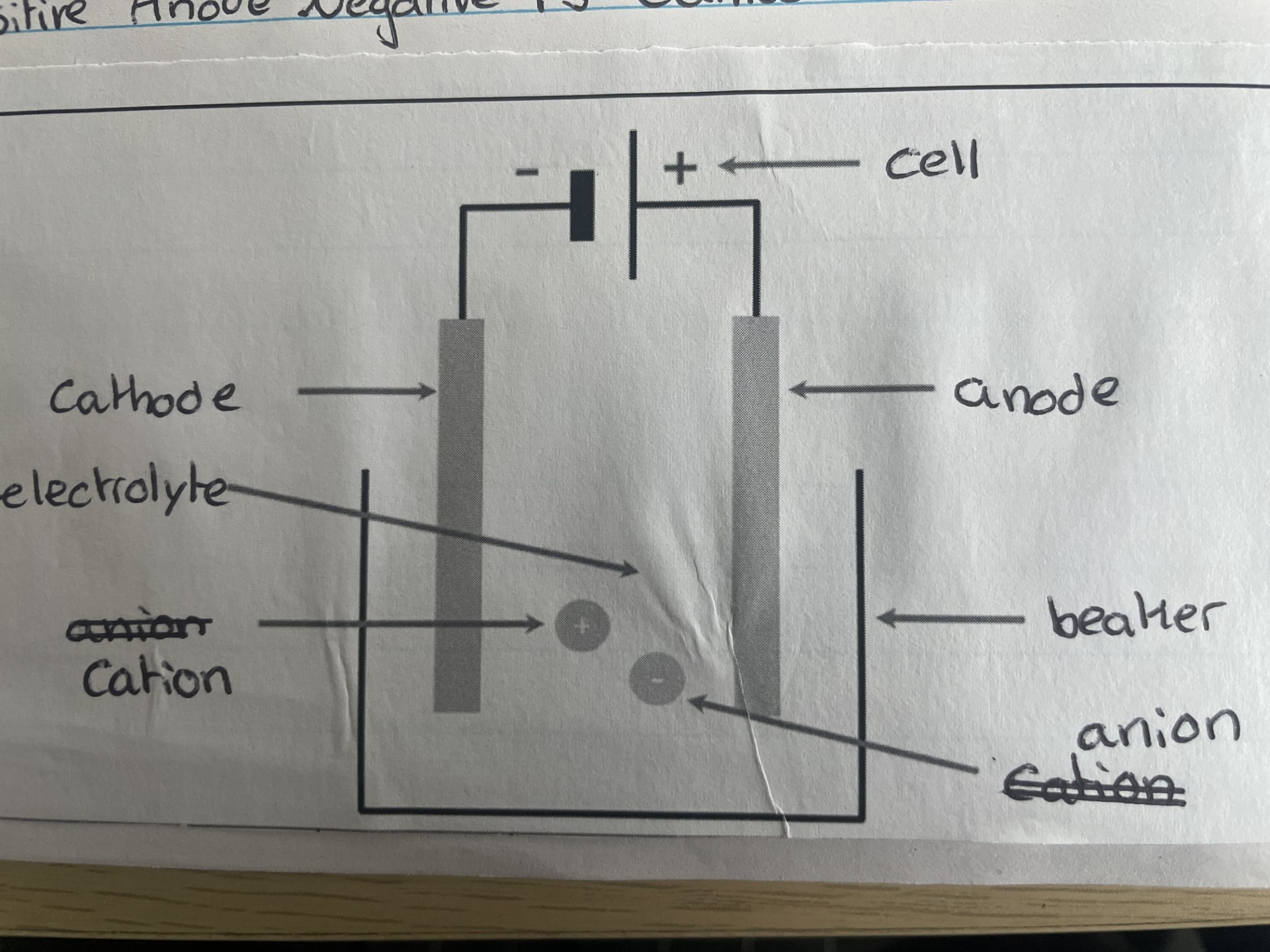
During electrolysis of molten ionic compounds, the metal element is formed at the cathode and the non metal at the anode
Aluminium Extract
Aluminium is found naturally within the ore bauxite
Pure Al2O3 (alumina) is purified from bauxite and melted (with cryolite)for electrolysis
The melting point of alumina is 2070oC. The temperature and energy cost is too high
Alumina is mixed with cryolite (a mineral) which reduces the melting point of the mixture and Al2O3. Lowering the temperature (and energy requirements) for ions to move.
Electrolysis of Solutions
During electrolysis of solution (aq), a small percent of the the H2O molecules break up into ions: H2O —> H+ + OH-
These ions could be discharged (a process where ion gain or lose electrons to form neutral atom or molecules) in electrolysis of ionic compound dissolve in water.
Cathode (negative)
The ions of the least reactive element will be discharged
Anode (positive)
If the negative ion is a Halide (Cl, Br, I) it is discharged
If any other anion is present, O2 gas will be produced from the OH- ions
The half equation: 4OH- —> O2 + 2H2O + 4e-
Electrolysis Required Practical
Independent variable - different metals ions in the electrolyte e.g. copper chloride and sodium chloride
Dependent variable - products of electrolysis using observations and test for gases
Control Variables - volume and concentration