01: The Foundations of Biochemistry
Principles
cells are the fundamental unit of life
exceptions could be viruses or prions
cells use a relatively small set of carbon-based metabolites to create polymeric machines, supramolecular structures, and information repositories
why carbon??? maximum 4 bond/ can be double bond, triple bond, etc. (can provide very large flexibility structure for the type of molecule) longer life span??
living organisms exist in a dynamic steady state, never at equilibrium with their surroundings
cells are highly organized- to fight against entropy increment, energy is needed (entropy= how chaotic an environment is)
cells have the capacity for precise self-replication and self-assembly using chemical information stored in the genome
recall the central dogma of genetics (dna replication)
living organisms change over time by gradual evolution
have dinosaurs truly extinguished?
What is Biochemistry?
uses chemical approaches to describe the structures, functions, mechanisms, and chemical processes (metabolism= synthesis + breaking down) shared by all organisms
Plasma Membrane = defines the periphery of the cell
composed of lipid and protein molecules
thin, flexible, hydrophobic barrier around the cell
contains embedded transport proteins, receptor proteins, and membrane enzymes
compartments = different functions, different locations
Cytoplasm Contains Cytosol and Suspended Particles
cytoplasm= internal volume enclosed by the plasma membrane (everything in cell !!)
composed of the cytosol (an aqueous solution) and a variety of suspended particles (such as mitochondria, chloroplasts, ribosomes, and proteasomes)
cytosol = highly concentrated solution (soup)
contains enzymes, RNA, amino acids, nucleotides, metabolites, coenzymes, and inorganic ions
The Nucleoid or Nucleus of a Cell Stores the Genome
genome = complete set of genes, composed of DNA
bacteria and archaea (formally grouped as prokaryotes) store their genome in a nucleoid
eukaryotes store their genome in a membrane-enclosed nucleus
Cellular Dimensions are Limited by Diffusion
cells are microscopic:
animal and plant cells:
5 to 100 um in diameter
unicellular microorganisms: 1 to 2 um long
upper limit of cell size is likely set by the rate of transport and the need to deliver O2 to all parts of the cell
as size increases, surface-to-volume ratio decreases
longest cell? biggest cell?
Archaea and Bacteria Subgroups are Distinguished by Their Habitats
aerobic = plentiful supply of O2; organisms transfer electrons from fuel to O2 for energy. die without oxygen
anaerobic = devoid of O2; organisms transfer electrons to nitrate; sulfate or CO2 for energy
obligate anaerobes = die when exposed to O2
facultative anaerobes = can live with or without O2
Ex. Question
Which organelle is not membrane-bound?
a. lysosome
b. peroxisome
c. ribosome
d. the Golgi complex
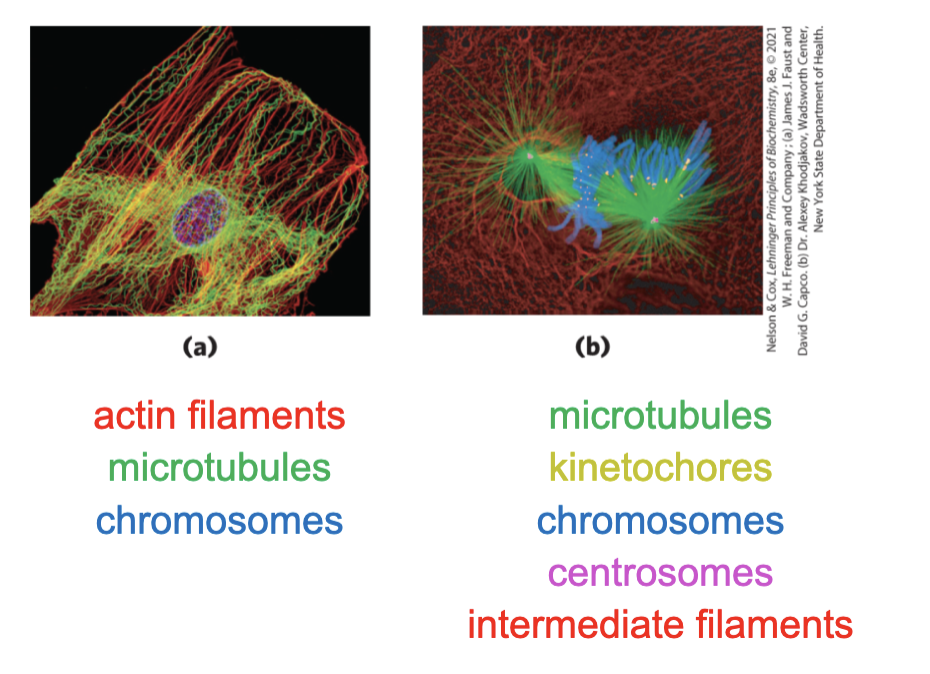
The Cytoplasm is Organized by the Cytoskeleton and is Highly Dynamic
cytoskeleton = 3D network of protein filaments in eukaryotic cells
actin filaments
microtubules
intermediate filaments
filaments undergo constant disassembly into their protein subunits and reassembly into filaments
shape and morphology, matter transportation
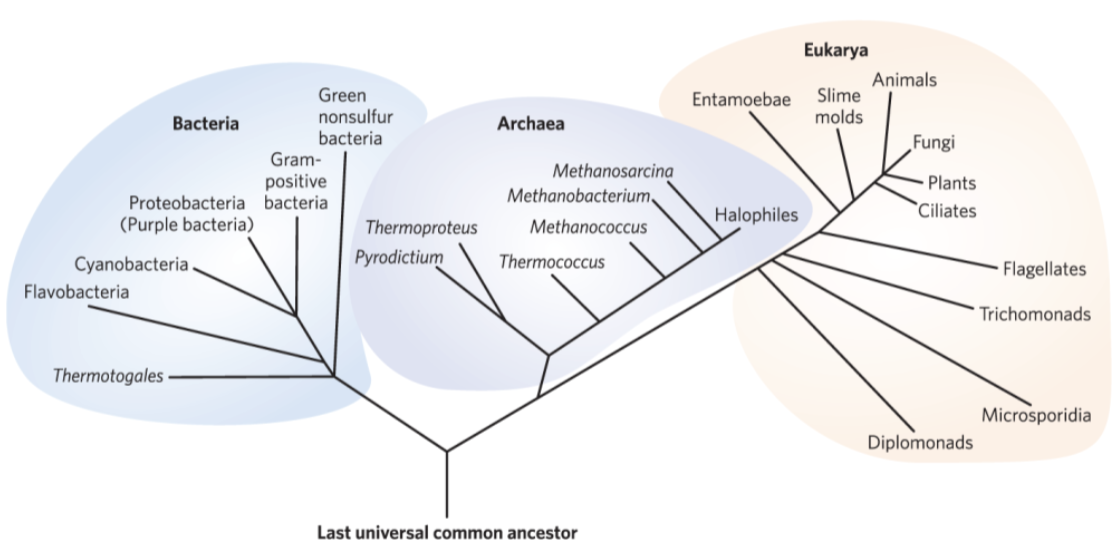
Organisms Belong to Three Distinct Domains of Life
bacteria = inhabitat soils, surface waters, and the tissues of other living or decaying organisms
archaea = inhabit extreme environments
eukarya = all eukaryotic organisms
more closely related to archaea than bacteria
Phylogeny of the Three Domains of Life
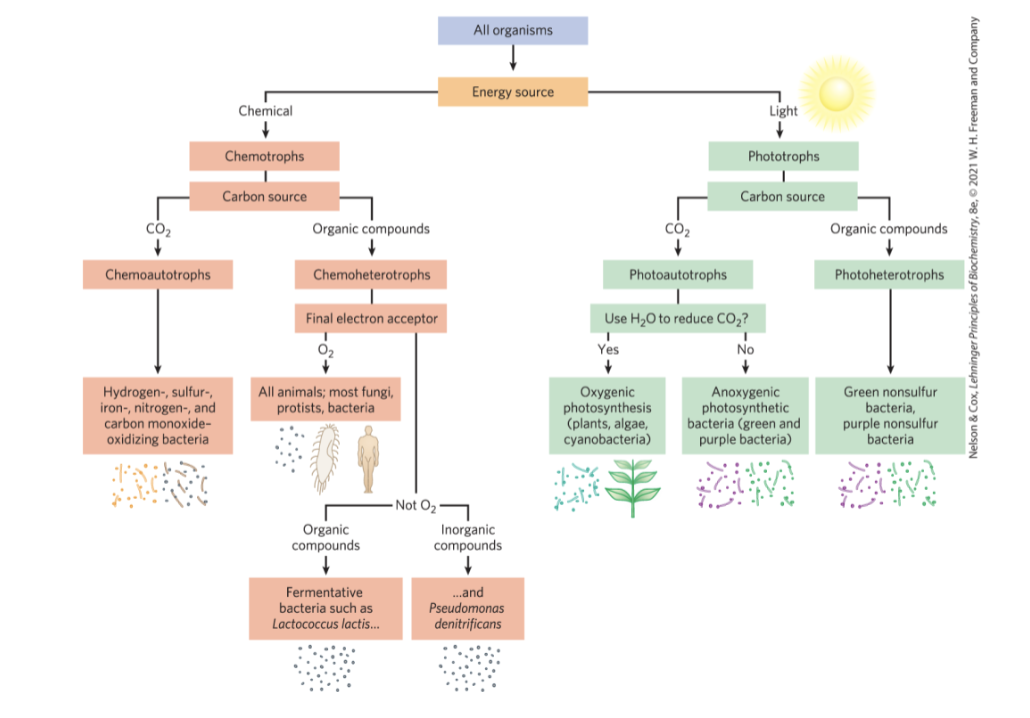
Organisms Differ Widely in Their Sources of Energy and Biosynthetic Precursors
phototrophs = trap and use sunlight
chemotrophs = derive energy from oxidation of a chemical fuel
autotrophs = can synthesize all their biomolecules directly from CO2
heterotrophs = require some preformed organic nutrients made by other organisms
Classifying Organisms According to Their Source of Energy
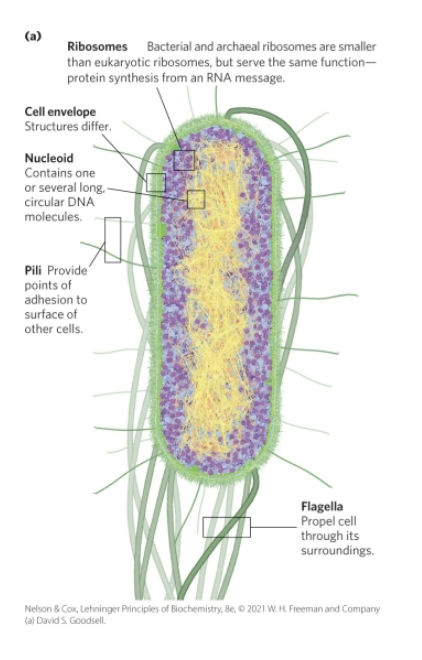
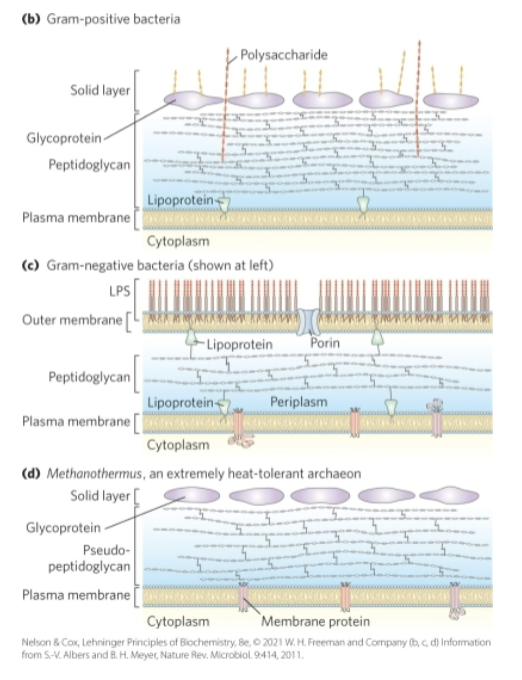
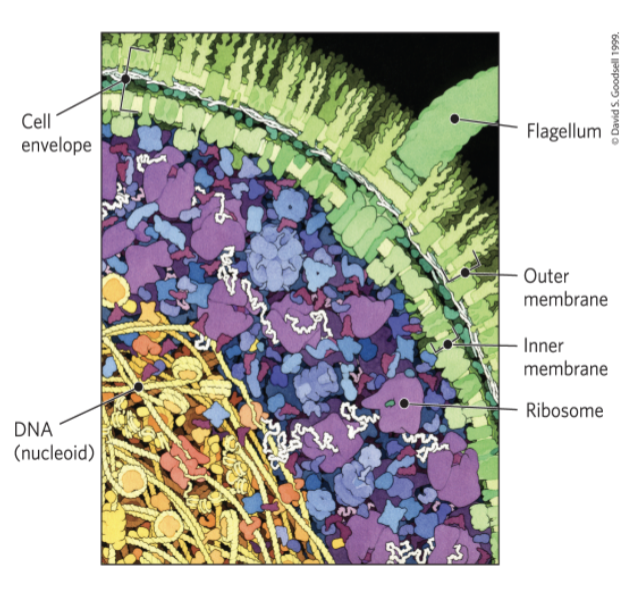
Bacteria and Archaeal Cells Share Common Features but Differ in Important Ways
cell envelope = composed of plasma membrane, outer membrane, and peptidoglycan (high molecular weight polymer)
gram-positive bacteria:
colored by gram’s stain
thick peptidoglycan layer outside plasma membrane
lack an outer membrane
gram-negative bacteria:
outer membrane composed of a lipid bilayer
archaea:
layer of peptidoglycan or protein confers rigidity on their cell envelopes
The E. coli Cytoplasm
contains ribosomes, enzymes, metabolites, cofactors, and inorganic ions
the nucleoid contains a single, circular molecule of DNA
plasmids = smaller, circular segments of DNA that confer resistance to toxins and antibiotics in the environments
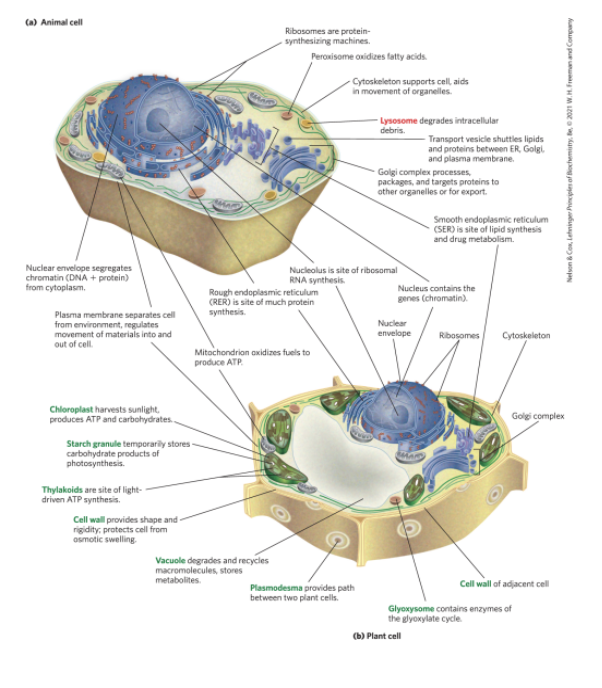
Eukaryotic Cells Have a Variety of Membranous Organelles, Which Can Be Isolated for Study
mitochondria = the site of most of the energy-extracting reactions of the cell
endoplasmic reticulum and golgi complexes = play central roles in the synthesis and processing of lipids and membrane proteins
peroxisomes = site of the oxidation of very-long-chain fatty acids and detoxification of reactive oxygen species
lysosomes = filled with digestive enzymes granules or droplets containing stored nutrients, such as starch and fat
Plant Cell Organelles
vacuoles = store large quantities of organic acids
chloroplasts = where sunlight drives the synthesis of ATP (adenosine triphosphate) in the process of photosynthesis
Eukaryotic Cell Structure
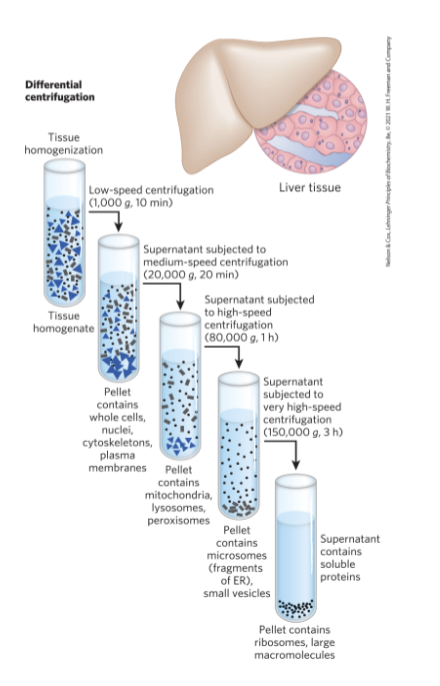
Subcellular Fractionation of Tissue
first step = gently disrupt cells or tissues by physical shear to rupture the plasma membrane
second step = centrifuge the homogenate
organelles differ in size and sediment at different rates
Cytoskeletal Filaments
The Structural Organization of the Cytoplasm
endomembrane system = segregates specific metabolic processes and provides surfaces on which certain enzyme-catalyzed reactions occur
exocytosis and endocytosis = mechanisms of transport (out of and into cells, respectively)
involve membrane fusion and fission
provide paths between the cytoplasm and the surrounding medium
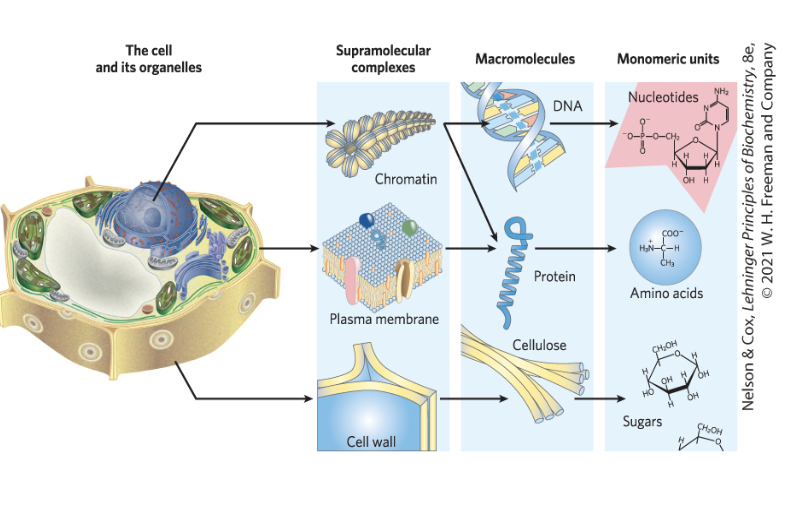
Cells Build Supramolecular Structures
held together by noncovalent interactions (hydrogen bonds, ionic interactions, van der waals interactions, and the hydrophobic effect)
why noncovalent??
In Vitro Studies May Overlook Important Interactions Among Molecules
in vitro = ‘in glass’
in vivo = ‘in the living'
molecules may behave differently in vivo and in vitro
1.2 Chemical Foundations
Biomolecules are Compounds of Carbon with a Variety of Functional Groups
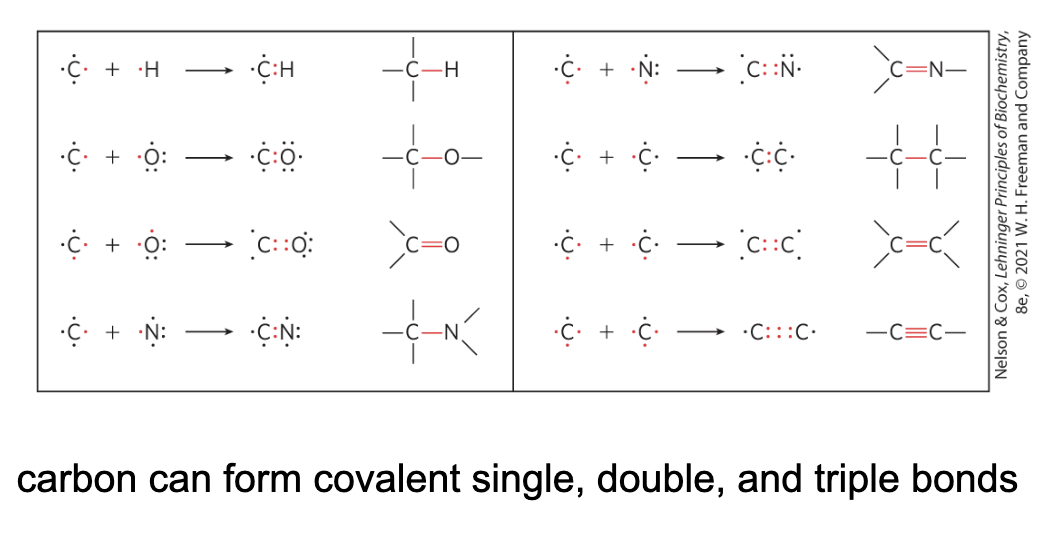
Geometry of Carbon Bonding
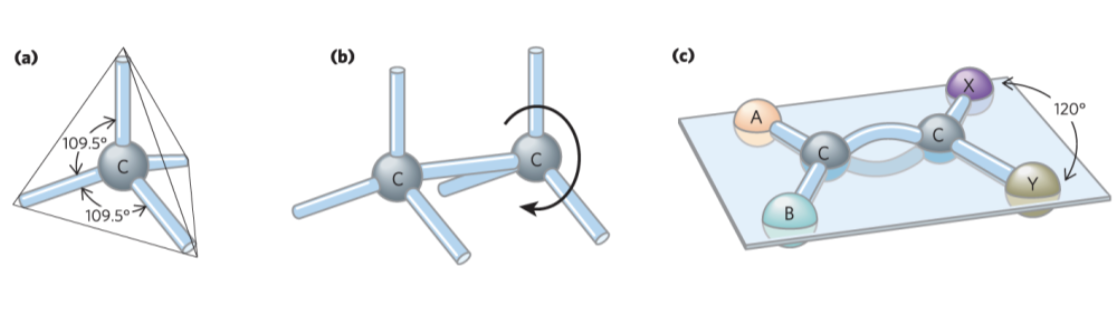
carbon atoms have a characteristic tetrahedral arrangement of their four single bonds
free rotation around each single bond
limited rotation about the axis of a double bond
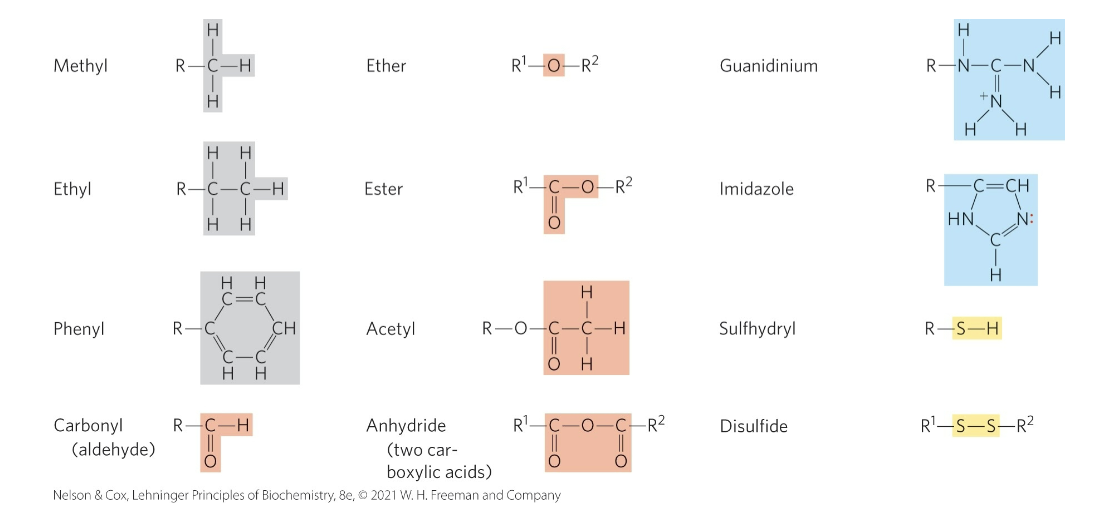
Common Functional Groups of Biomolecules
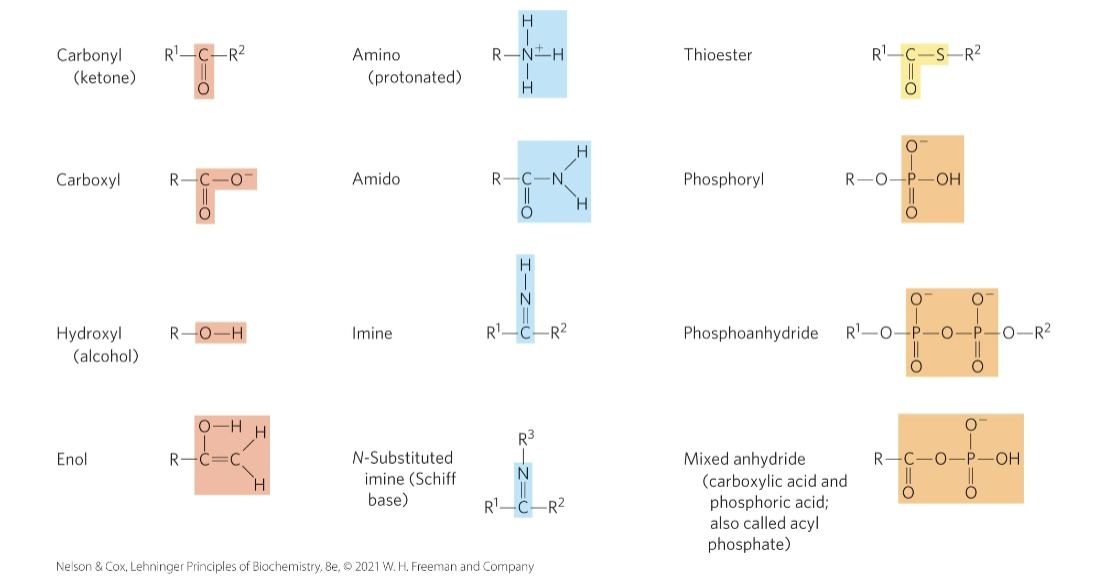
Additional Functional Groups of Biomolecules
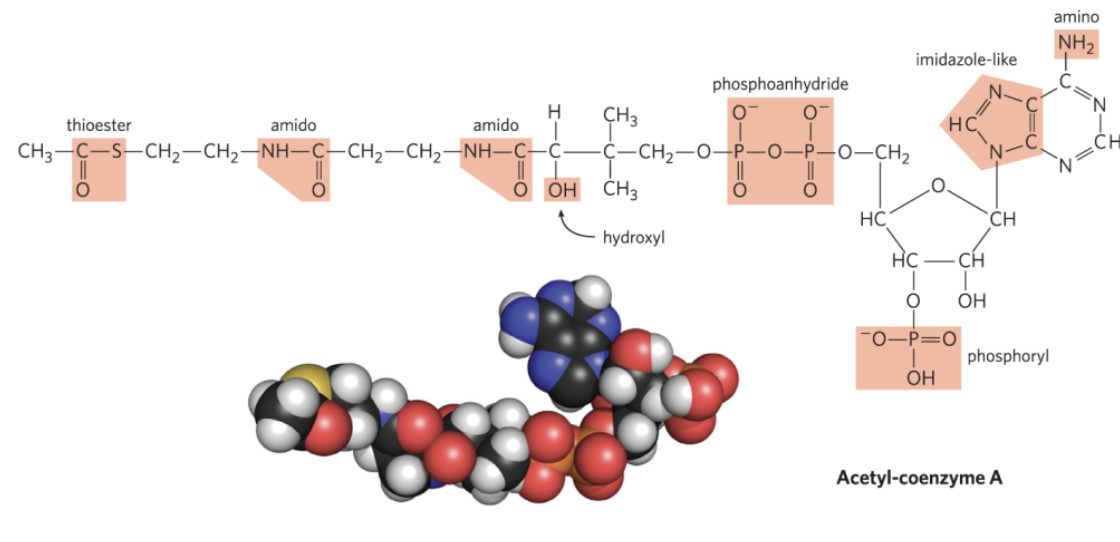
Many Biomolecules are Polyfunctional
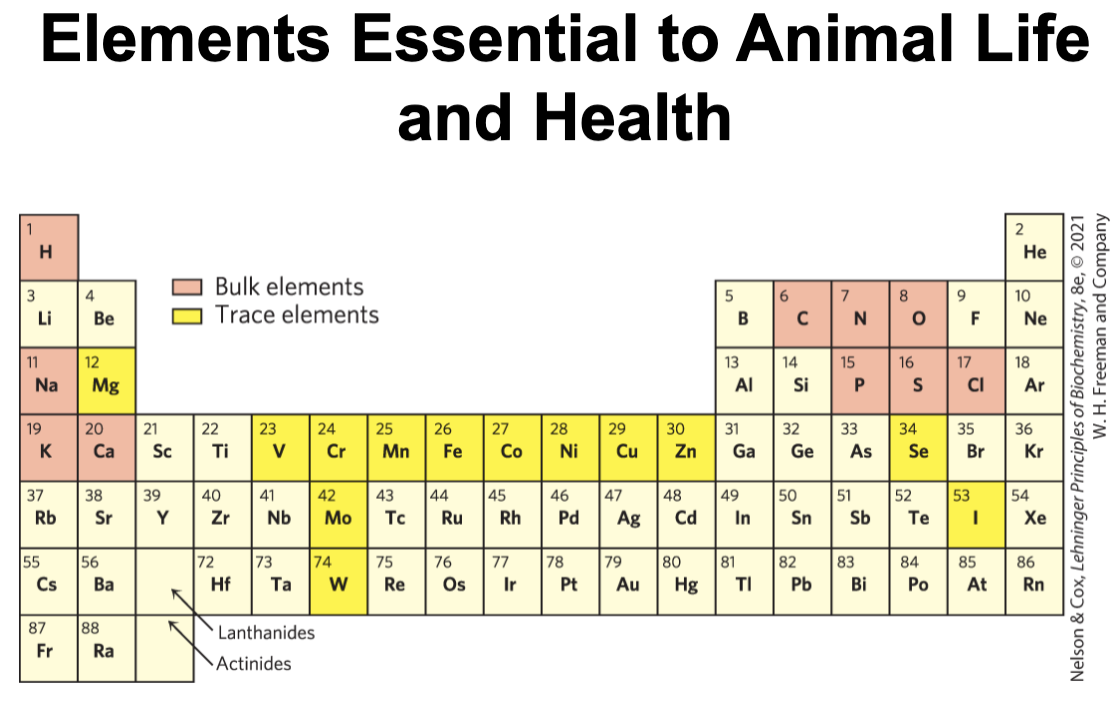
Cells Contain a Universal Set of Small Molecules
central metabolites:
common amino acids
nucleotides
sugars and their phosphorylated derivatives
mono-, di-, and tricarboxylic acids
secondary metabolites = specific to the organism
metabolome = entire collection of small molecules in a given cell under a specific set of conditions
metabolomics = the systematic characterization of the metabolome under very specific conditions
Macromolecules are the Major Constituents of Cells
macromolecules = polymers with molecular weights above ~5000 that are assembled from relatively simple precursors
proteins
nucleic acids
polysaccharides
oligomers = shorter polymers
informational macromolecules = name for proteins, nucleic acids, and some oligosaccharides, given their information-rich sequences
Protein Macromolecules
proteins = long polymers of amino acids
can function as enzymes, structural elements, signal receptors, transporters
proteome = sum of all the proteins functioning in a cell
proteomics = the systematic characterization of this protein complement under a specific set of conditions
Nucleic Acid Macromolecules
nucleic acids = DNA and RNA = polymers of nucleotides
store and transmit genetic information
some RNA molecules have structural and catalytic roles in supramolecular complexes
genomes = entire sequence of a cell’s DNA or RNA
genomics = the characterization of the structure, function, evolution, and mapping of genomes
Polysaccharide Macromolecules
polysaccharides = polymers of simple sugars
energy-rich fuel stores
rigid structural components of cell walls (in plants and bacteria)
extracellular recognition elements that bind to proteins on other cells
glycome = entire complement of carbohydrate-containing molecules
Lipid Molecules
lipids = water-insoluble hydrocarbon derivatives
structural components of membranes
energy-rich fuel stores
pigments
intracellular signals
lipidome = the lipid containing molecules in a cell
Building Blocks of Biochemistry
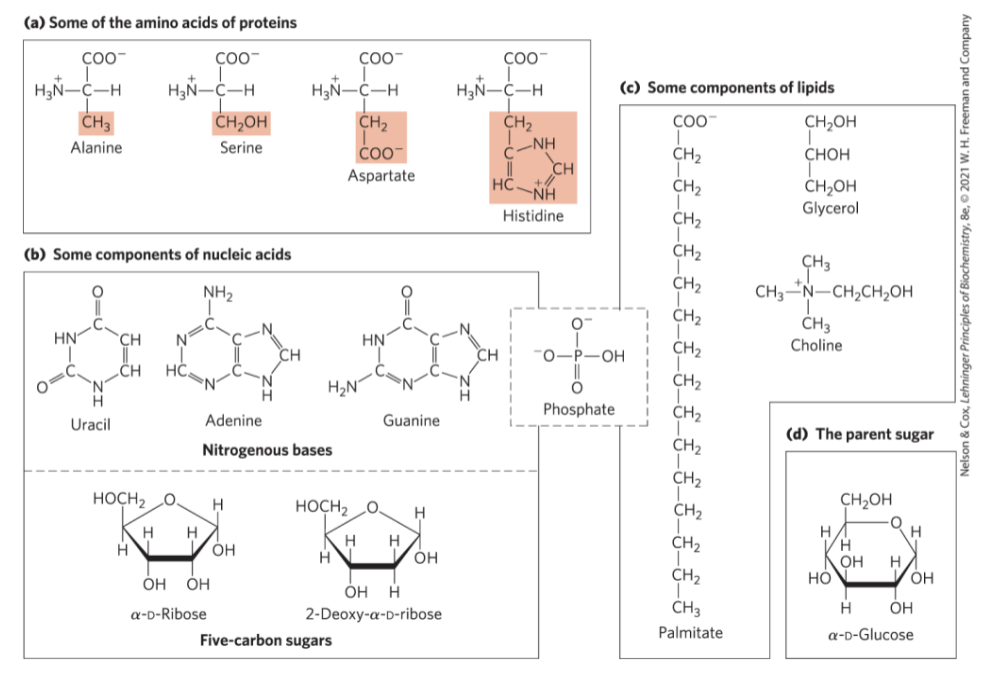
Major Classes of Biomolecules in E. coli Cells
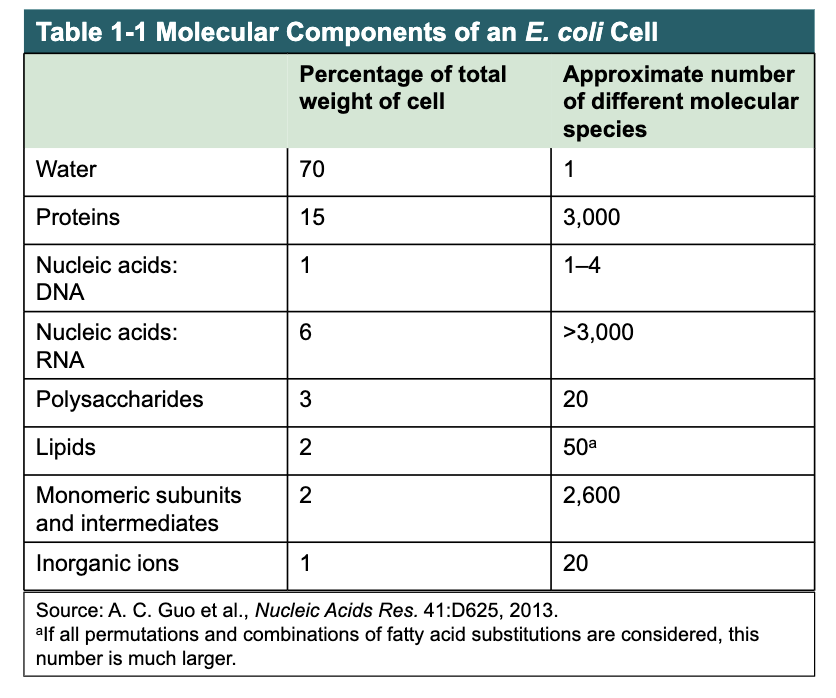
3 Dimensional Structure is Described by Configuration and Conformation
configuration = the fixed spatial arrangement of atoms
stereoisomers = molecules with the same chemical bonds and same chemical formula
stereospecific = requiring specific conformations in the interacting molecules
describes typical interactions between biomolecules
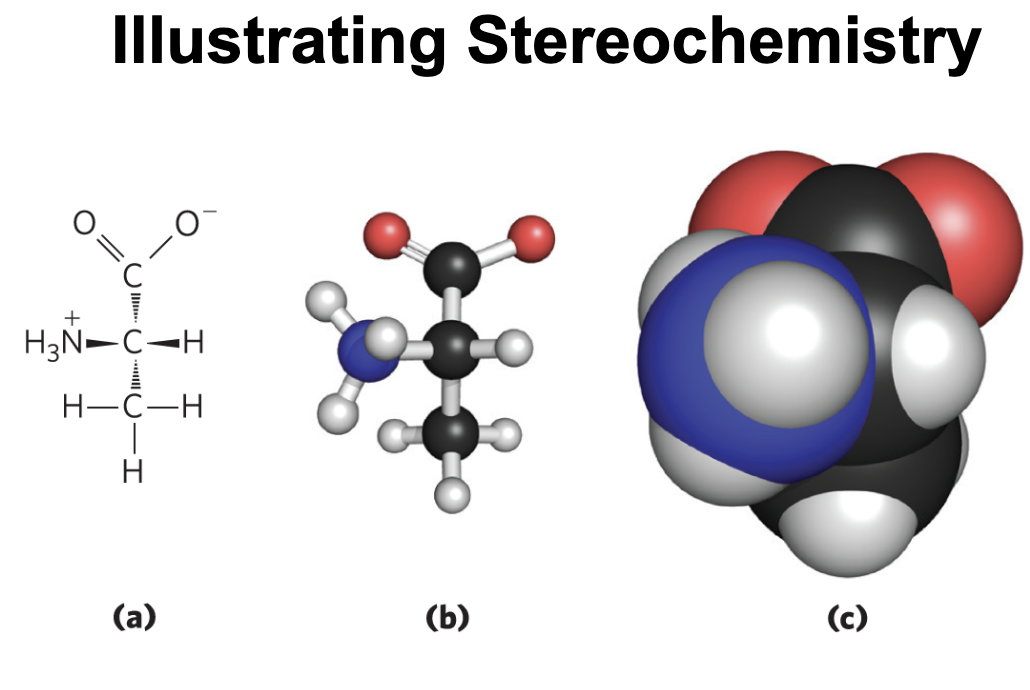
Configurations of Geometric Isomers
geometric isomers, or cis-trans isomers = differ in the arrangement of substituent groups with respect to the double bond
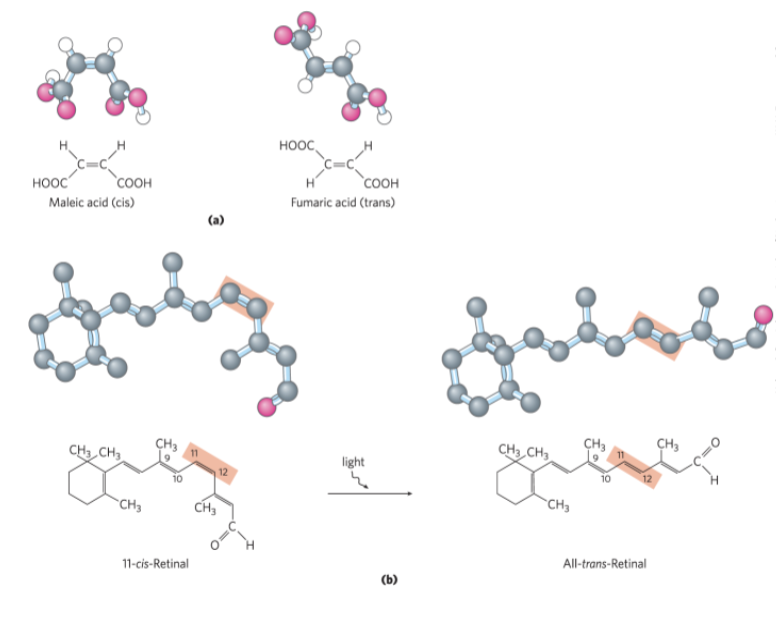
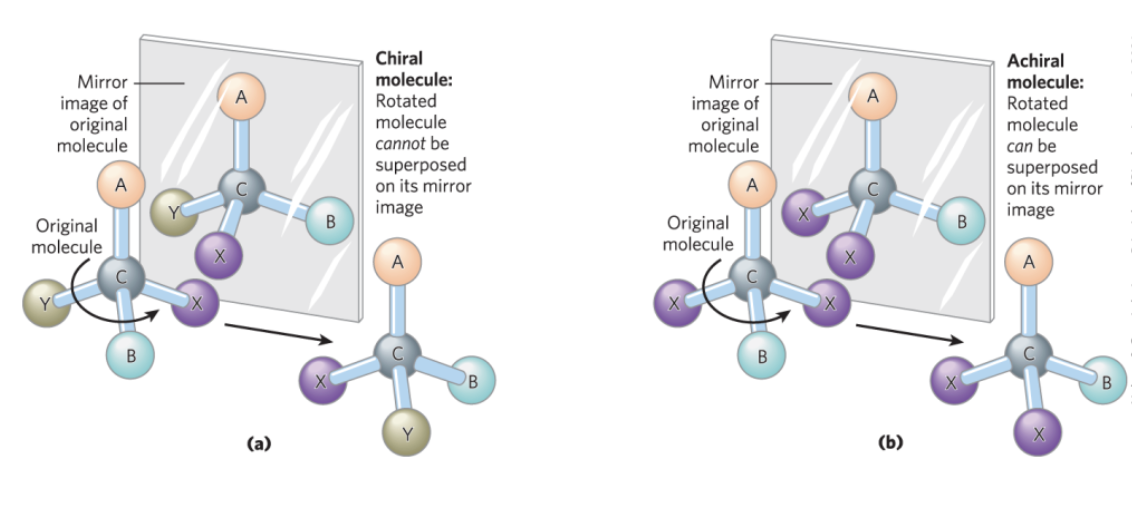
Chiral and Achiral Molecules
chiral centers = asymmetric carbons
a molecule can have 2n stereoisomers, where n is the number of chiral carbons
Enantiomers and Diastereomers
enantiomers = stereoisomers that are mirror images of each other
diastereomers = stereoisomers that are not mirror images of each other
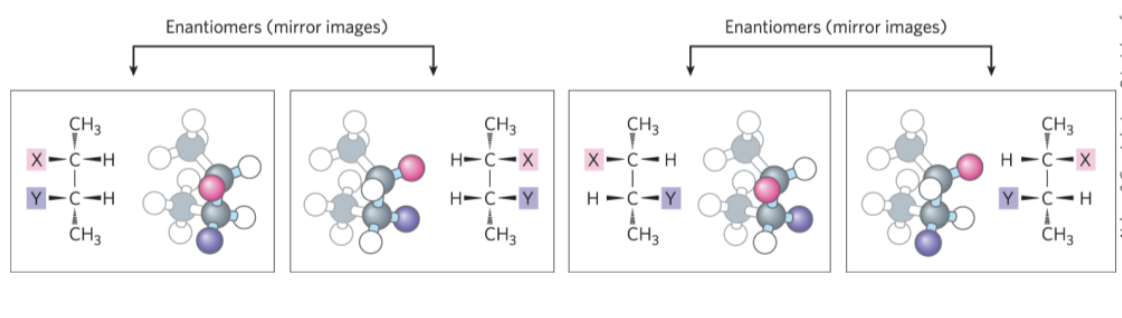
Optical Activity of Enantiomers
enantiomers have nearly identical chemical reactivities, but differ in optical activity
a racemic mixture (equimolar solution of two enantiomers) show no optical rotation
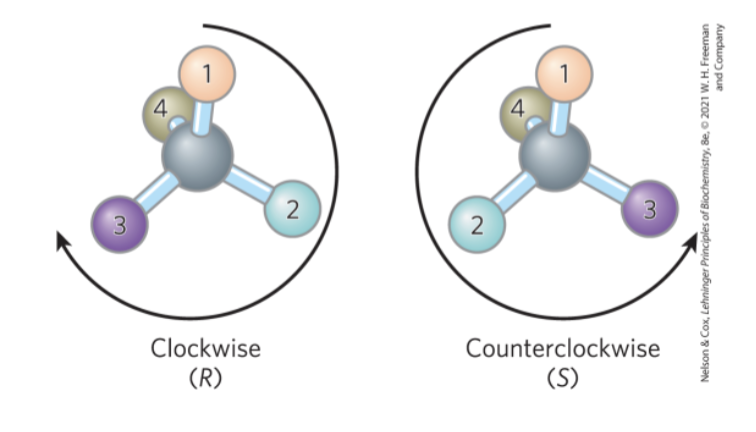
Naming Stereoisomers Using the RS System
each group attached to a chiral carbon is assigned a priority, where:
-OCH3 > -OH . -NH2 > -COOH > -CHO > -CH2OH > -CH3 > -H
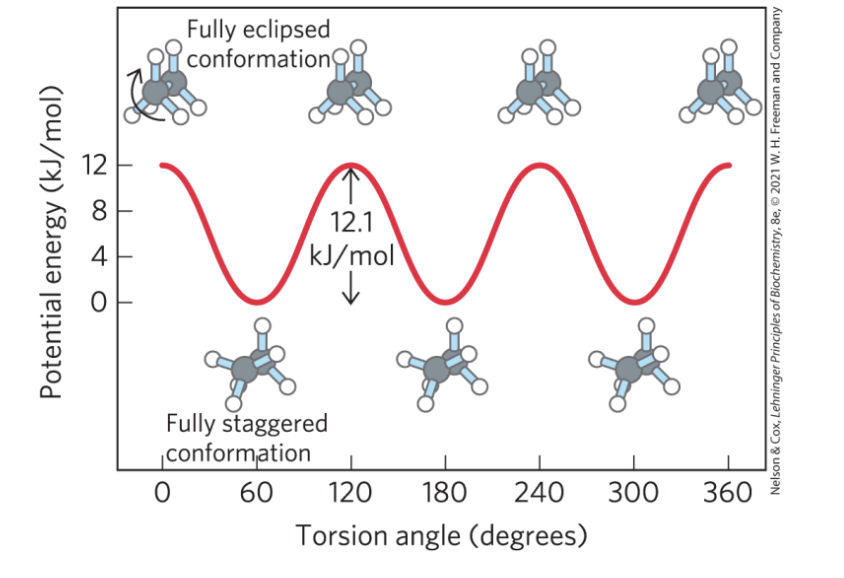
Molecular Conformation
conformation = the spatial arrangement of substituent groups that are free to assume different positions in space
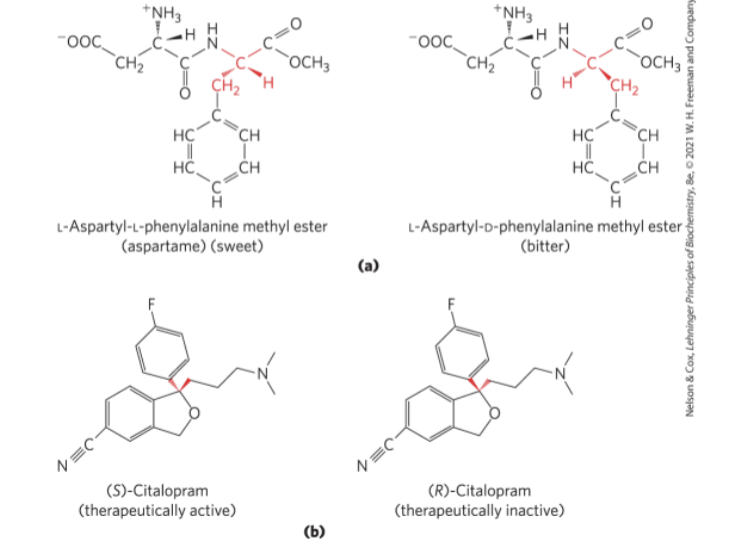
Interactions between Biomolecules are Stereospecific
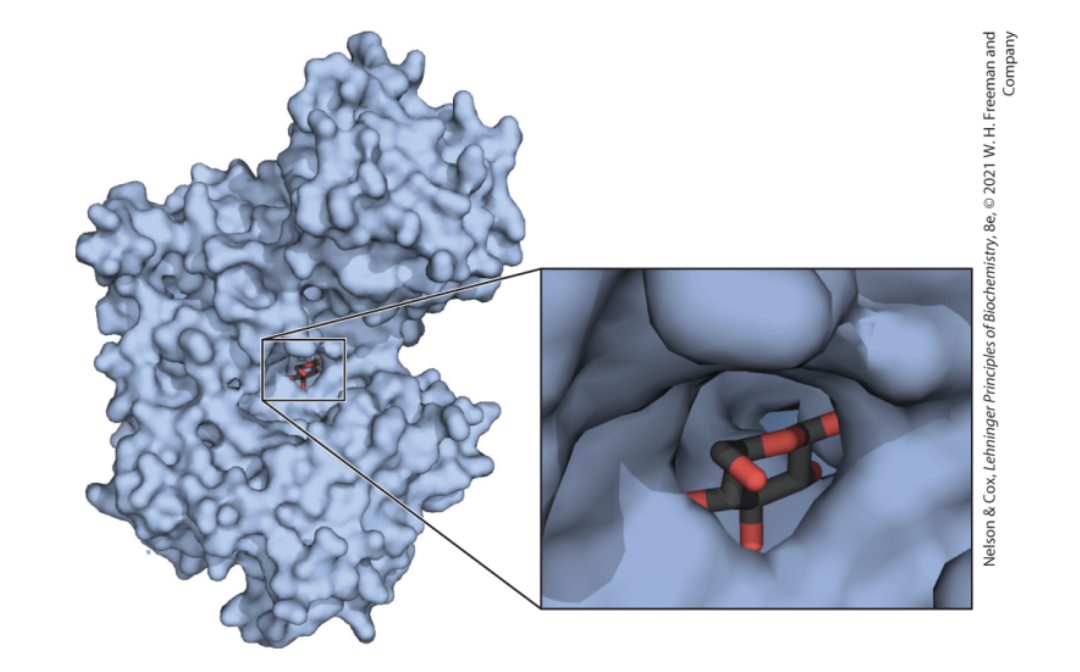
Biological Systems can Distinguish Stereoisomers
stereospecificity = the ability to distinguish between stereoisomers
1.3 Physical Foundations
Living Organisms Exist in a Dynamic Steady State, Never at Equilibrium with Their Surroundings
small molecules, macromolecules, and supramolecular complexes are continuously synthesized and broken down
living cells maintain themselves in a dynamic steady state distant from equilibrium
maintaining steady state requires the constant investment of energy
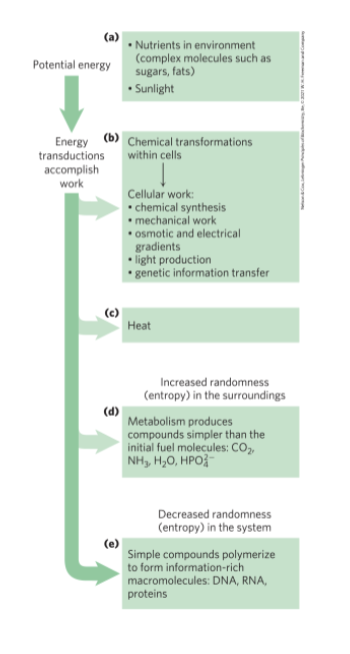
Organisms Transform Energy and Matter from Their Surroundings
system = all the constituent reactants and products, the solvent that contains them, and the immediate atmosphere
universe = system and its surroundings
types of systems:
isolated = system exchanges neither matter nor energy with its surroundings
closed system = system exchanges energy but not matter with its surroundings
open system = system exchanges both energy and matter with its surroundings
Energy Transformation in Living Organisms
first law of thermodynamics: in any physical or chemical change, the total amount of energy in the universe remains constant, although the form of the energy may change
Extracting Energy from the Surroundings
photoautotrophs:
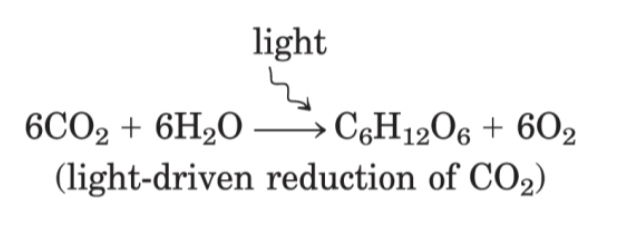
chemotrophs
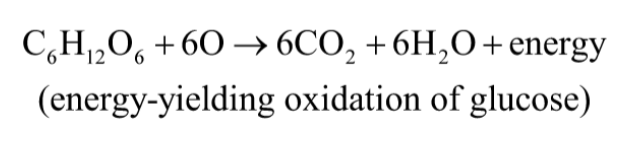
Oxidation-Reduction Reactions
autotrophs and heterotrophs participate in global cycles of O2 and CO2, driven by sunlight, making these 2 groups interdependent
oxidation-reduction reactions = one reactant is oxidized (loses electrons) as another is reduced (gains electrons)
describes reactions involved in electron flow
Creating and Maintaining Order Requires Work and Energy
second law of thermodynamics: randomness in the universe is constantly increasing
entropy, S = represents the randomness or disorder of the components of a chemical system
Free Energy, G
enthalpy, H = heat content, roughly reflecting the number and kinds of bonds
free energy, G, of a closed system = H - TS, where H represents enthalpy, T represents absolute temperature, and S represents entropy
Free-Energy Change, △G
△G = △H - T△S
where △H is negative for a reaction that releases heat, and △S is positive for a reaction that increases the system’s randomness
spontaneous reactions occur when △G is negative
Coupling Reactions
energy-requiring (endergonic) reactions are often coupled to reactions that release free energy (exergonic)
the breakage of phosphoanhydride bonds in ATP is highly exergonic
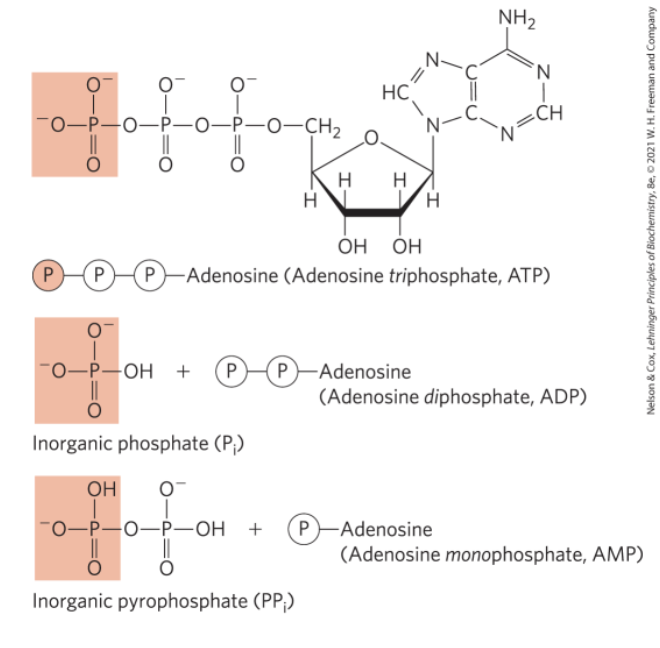
Energy Coupling Links Reactions in Biology
free-energy change, △G = amount of energy available to do work
always less than the theoretical amount of energy released
in closed systems, chemical reactions proceed spontaneously until equilibrium is reached
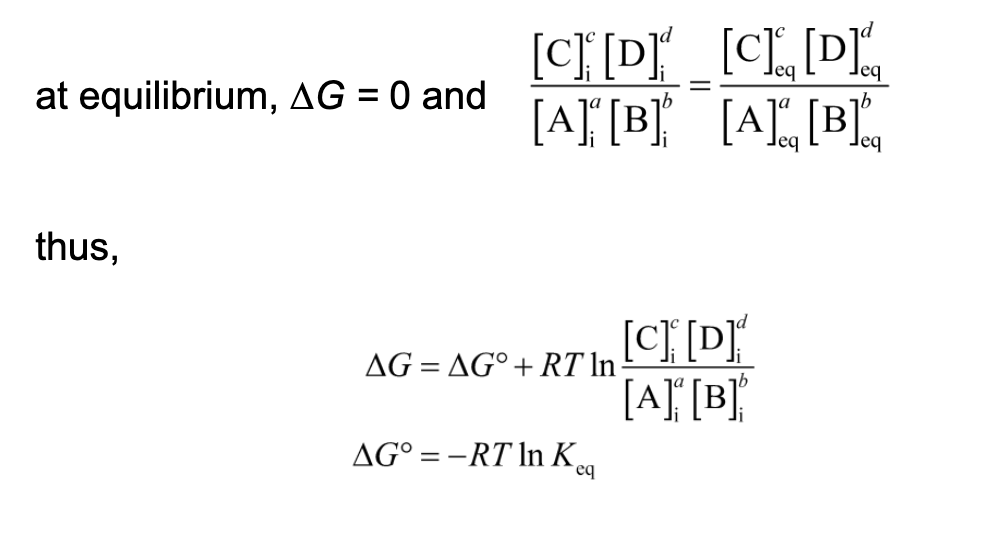
Keq and △Gº are Measures of a Reaction’s Tendency to Proceed Spontaneously
for the reaction,

the equilibrium constant, Keq is given by,
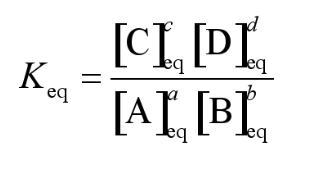

Mass-Action Ratio, Q
mass-action ratio, Q = ratio of product concentrations to reactant concentrations at a given time
can be calculated to determine how far the reaction is from equilibrium
Standard Free-Energy Change, △Gº
△G (the actual free-energy change) for any chemical reaction is a function of the standard free-energy change, △Gº
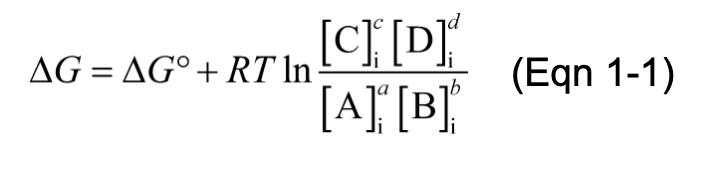

Reactions Can Do No Work at Equilibrium
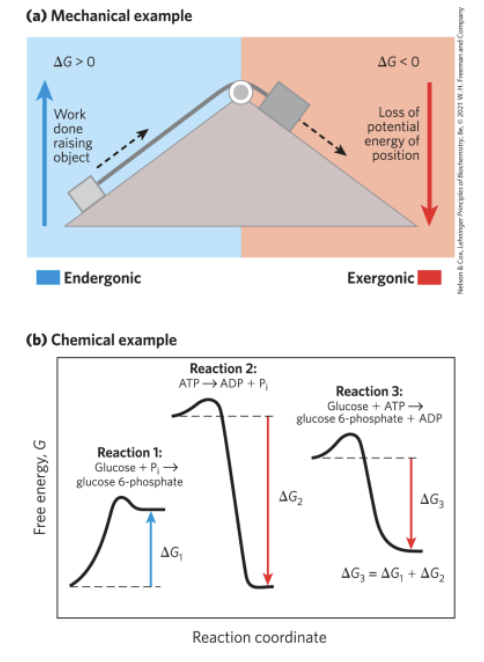
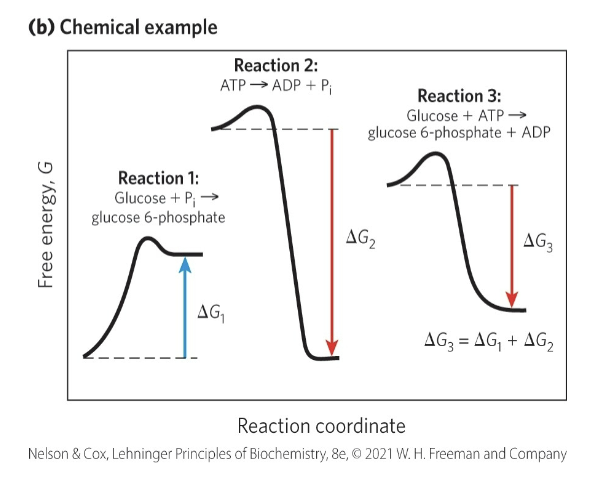
Reaction Coordinate Diagrams
reaction coordinate diagrams = illustrates how exergonic reactions can be coupled to endergonic reactions
reaction 1: endergonic; △G1 is positive
reaction 2: exergonic; △G2 is negative
reaction 3: △G3 is negative
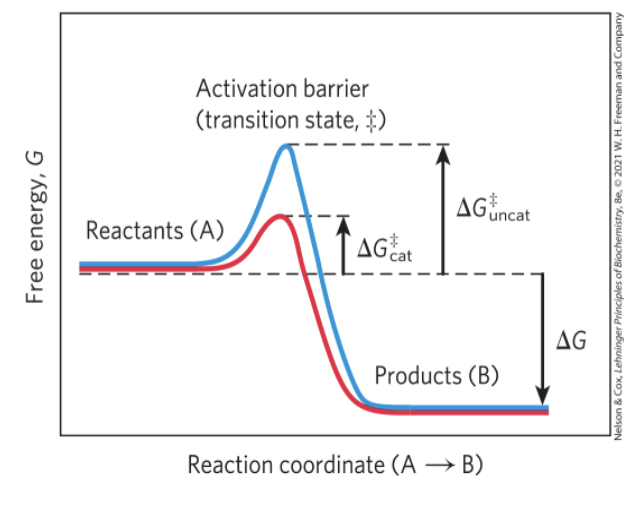
Enzymes Promote Sequences of Chemical Reactions
enzymes = greatly enhance reaction rates of specific chemical reactions without being consumed in the process
transition state = higher free energy than reactant or product
activation energy, △G = difference in energy between the reactant in its ground state and its transition state
Catabolism and Anabolism
pathways = sequences of consecutive reactions in which the prodicy of one reaction becomes the reactant in the next
catabolism = degradative, free-energy-yielding reactions
drives ATP synthesis
produces the reduced electron carriers NAD(P)H
anabolism = synthetic pathways
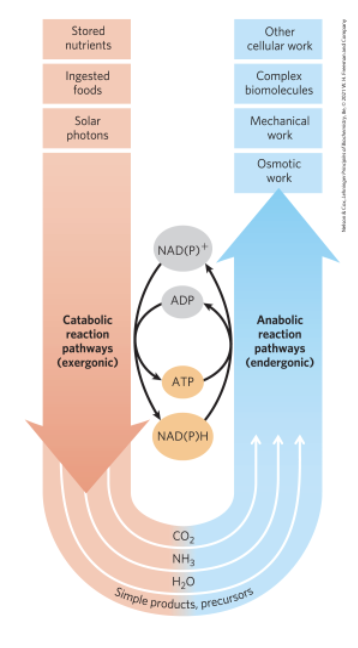
Metabolism
overall network of enzyme-catalyzed pathways, both catabolic and anabolic
unity of life = pathways of enzyme-catalyzed reactions that act on the main constituents of cells- proteins, fats, sugars, and nucleic acods- are nearly identical in all living organisms
Metabolism is Regulated to Achieve Balance and Economy
feedback inhibition = keeps the production and utilization of each metabolic intermediate in balance
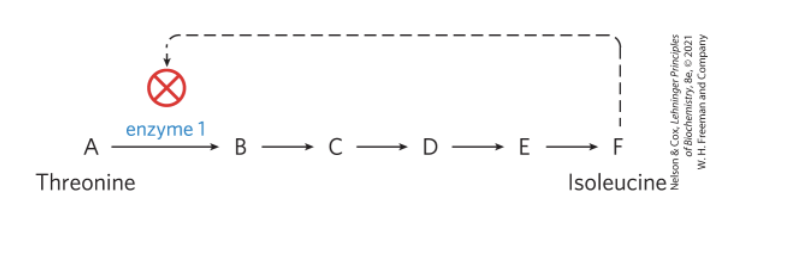
systems biology = tasked with understanding complex interactions among intermediates and pathways in quantitative terms
1.4 Genetic Foundations
Genetic Information is Encoded in DNA
deoxyribonucleic acid, DNA = sequence of the monomeric subunits (deoxyribonucleotides)
encode the instructions for forming all other cellular components
provide a template to produce identical DNA molecules
Genetic Continuity is Vested in Single DNA Molecules
DNA of an E. coli cell is a single molecule containing 4.64 million nucleotide pairs
must be replicated perfectly to give rise to identical progeny by cell division
The Structure of DNA Allows Its Replication and Repair with Near-Perfect Fidelity
deoxyribonucleotides = monomeric subunit that make up the DNA polymer
each deoxyribonucleotide in one strand pairs specifically with a complementary deoxyribonucleotide in the opposite strand
strands are held together by hydrogen bonds
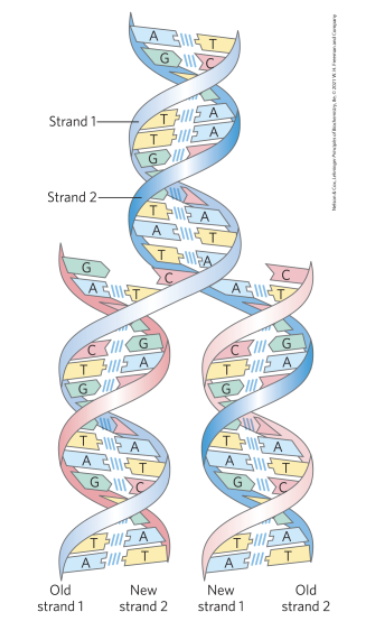
The Linear Sequence in DNA Encodes Proteins with Three-Dimensional Structures
native conformation = precise 3D structure of a protein
crucial to protein function
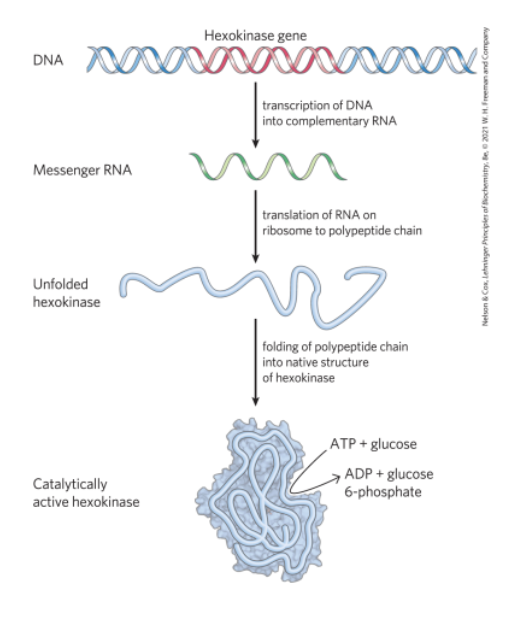
1.5 Evolutionary Foundations
Changes in the Hereditary Instructions Allow Evolution
mutation = changes in the nucleotide sequence of DNA
changes the instructions for a cellular component
can be beneficial
wild type = unmutated cells
Biomolecules First Arose by Chemical Evolution
Miller and Urey experiments found that biomolecules may have been produced near hydrothermal vents at the bottom of the sea or by the action of lightning and high temperature on gaseous mixtures
The Role of RNA in Prebiotic Evolution
RNA (ribonucleic acid) = can act as catalysts in biologically significant reactions
likely played a crucial role in prebiotic evolution, both as catalyst and as information repository
RNA or Related Precursors May Have Been the First Genes and Catalysts
RNA or similar molecule may have been the first gene and the first catalyst
alternatively, simple metabolic pathways may have evolved first, perhaps at the hot vents in the ocean floor
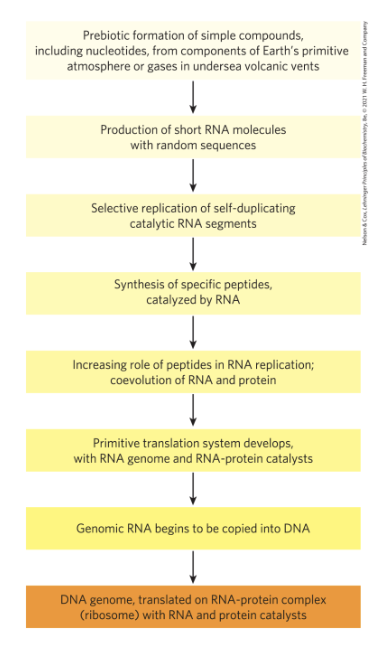
Biological Evolution Began More than Three and a Half Billion Years Ago
lipid vesicles containing organic compounds and self-replicating RNA gave rise to protocells
protocells with the greatest capacity for self-replication became more numerous

The First Cell Probably Used Inorganic Fuels
earliest cells probably obtained energy from inorganic fuels, such as ferrous sulfide and ferrous carbonate
photosynthetic processes:
arose from evolution
pigments capture energy of light from the sun and reduce CO2 to organic compounds
atmosphere became richer in O2 with the rise of O2- producing photosynthetic bacteria
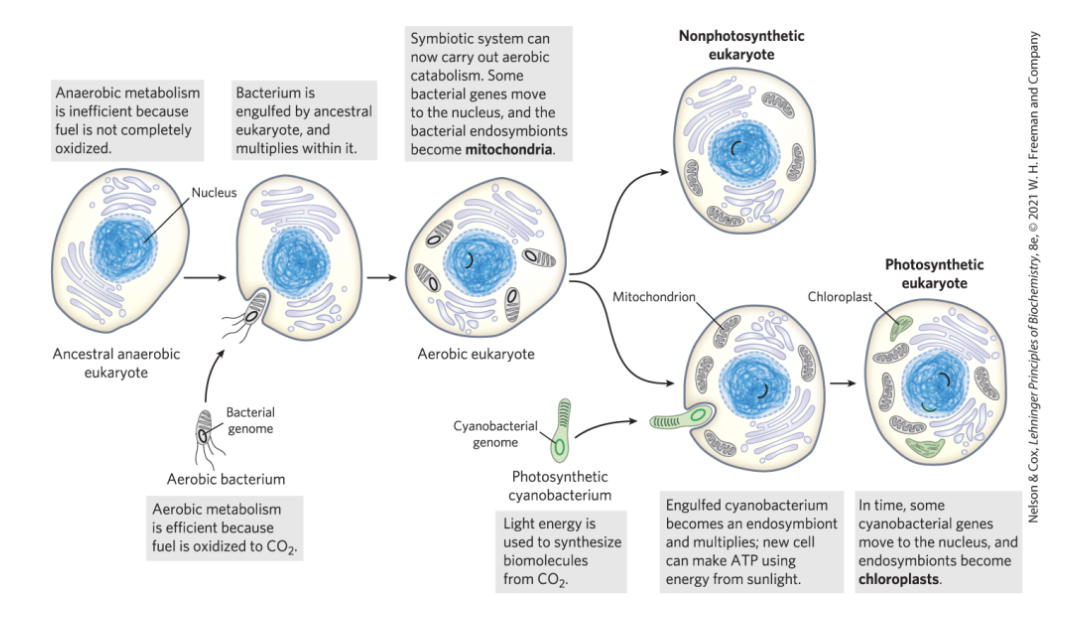
Eukaryotic Cells Evolved from Simpler Precursors in Several Stages
3 major changes led to the evolution of eukaryotes:
evolution of the chromosome
evolution of the nucleus
formation of endosymbiotic associations between early eukaryotic cells and aerobic or photosynthetic bacteria
in multicellular organisms, differentiated cell types specialize in functions essential to the organism’s survival
Evolution of Eukaryotes through Endosymbiosis
Molecular Anatomy Reveals Evolutionary Relationships
homologs = proteins encoded by genes that share ready detectable sequence similarities
gene or protein sequence similarities between organisms can determine phylogenetic relationships
Functional Genomics Shows the Allocations of Genes to Specific Cellular Processes
genes can be grouped according to the specific process in which they function
can approximate the proportion of the genome dedicated to a specific process
genes involved in regulation of cellular processes tend to increase with organism complexity
housekeeping genes = expressed under all conditions, not subject to much regulation
Genomic Comparisons Have Increasing Importance in Medicine
large-scale sequencing studies have identified many genes in which mutations correlate with a medical condition
the proteins these genes encode might become the target for drugs to treat a given condition