Equilibrium
State of the system:
Described by stating the equilibrium pressure of water vapor at the given temperature
With constant volume and temperature, the partial pressure of each gas should be proportional to the number of moles of that gas
Reactions are reversible - forward and reverse reactions take place at the same rate
Equilibrium Constant:
The relationship between the partial pressures of different gases present at equilibrium
Equilibrium constant expression:
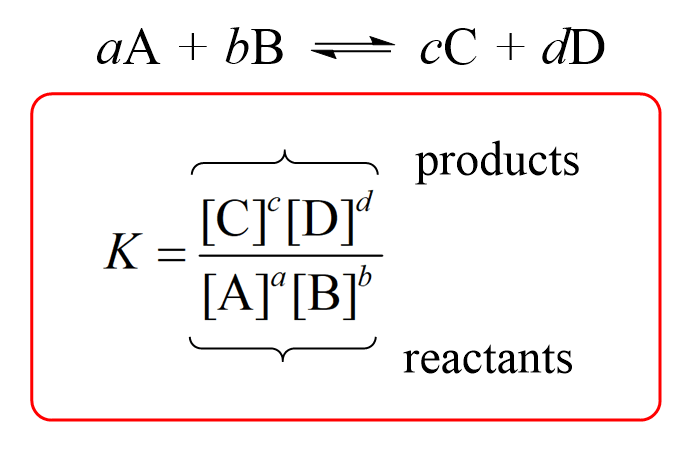
If K>1, that means products are favored
If K<1, that means reactants are favored
Reciprocal Rule: the equilibrium constants for the forward and reverse reactions are the reciprocals of each other (K=1/K)
Rule of multiple equilibria: If the reaction can be expressed as a sum of 2+ reactions, K for the overall reaction if the product of equilibrium constants of individual reactions
Only temperature affects K
We cannot tell reaction rate or enthalpy from K
Reaction Quotient (Q):
Measures the beginning reactants and products, whereas K is measuring R and P after the equilibrium
The form is the same as that for the equilibrium constant, K
If Q was big and K is small, then the reaction would go backward (P—>R)
If Q and K were around the same (both big or both small) then there would be no/little change
If Q was small and K is big, then the reaction would go forward (R—>P)
Equilibrium:
when rates of forward and reverse reactions and equal
Forward and reverse reactions occur at the same rate
The concentration of species remains constant with time and they are independent of the direction from which equilibrium is approached
End Point: