group 7 - the halogens
What are the group 7 elements?
there are elements in group 7 - fluorine, chlorine, bromine, iodine and astatine (we don’t do much about astatine in detail but you still need to know a bit about it)
These elements are non-metals that are poisonous
All halogens have similar reactions as they have seven electrons in their outer shell
Halogens are diatomic, meaning they form molecules made of pairs if atoms sharing electrons (forming a single covalent bond)
When halogens gain an electron during reactions they form -1 ions called halide ions, this is because they now have one more electron than they do protons
Properties of the group 7 elements
Fluorine
yellow gas at room temp
Very reactive, poisonous gas
Colourless in solution
Chlorine
pale yellow - green gas at room temp
Reactive, poisonous and dense gas
Pale green in solution
Bromine
red/brown liquid at room temp
Dense red-brown volatile (will easily become a gas) liquid
Orange in solution
Iodine
grey solid at room temp
Shimmery, crystalline solid that sublimes to form a purple vapour
Dark brown in solution
Tip to remember; going down the group they get more and more solid and dark in solution
Boiling points
The melting and boiling points of the halogens increase as you go down the group
This is due to the increasing intermolecular forces (remember they are diatomic, meaning covalent) as the atoms get large
Meaning more energy is required to over come these forces
Reactivity and displacements reactions of the halogens
the atoms in group 7 get larger as you go down the group because there are more electrons and therefore energy levels
All group 7 metals are reactive since they have 7 electrons in their outer shell and it does not require much energy to gain only one more
However, the smaller atoms are more reactive than the larger ones. This is because in, for example, fluorine the outer shell that the electrons is going to is very close to the positive nucleus making it easier to attract the negative electron.
Whereas in iodine the outer shell is very far from the nucleus making it more difficult to attract the electron.
However, whilst iodine is the least reactive of the halogens it is still a reactive element compared to others.
Displacement reactions
You only need to know about the displacement reactions between chlorine bromine and iodine
A halogen displacement reaction occurs when a more reactive halogen displaces a less reactive halogen from an aqueous solution of its halide. So for example, chlorine could displace bromine but iodine could not.
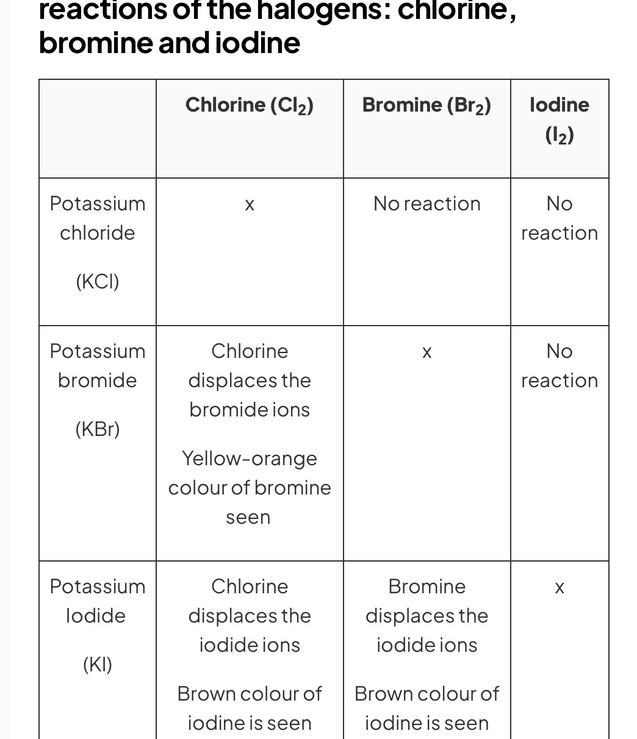
Reactions of the halogens
Metal halides
chlorine, bromine and iodine react with metals and non-metals to form compounds
The halogens react with some metals to form ionic compounds which are metal halide salts
The halide ion carries a -1 charge so the ionic compound formed will have different numbers of halogen atoms, depending on the valency of the metal
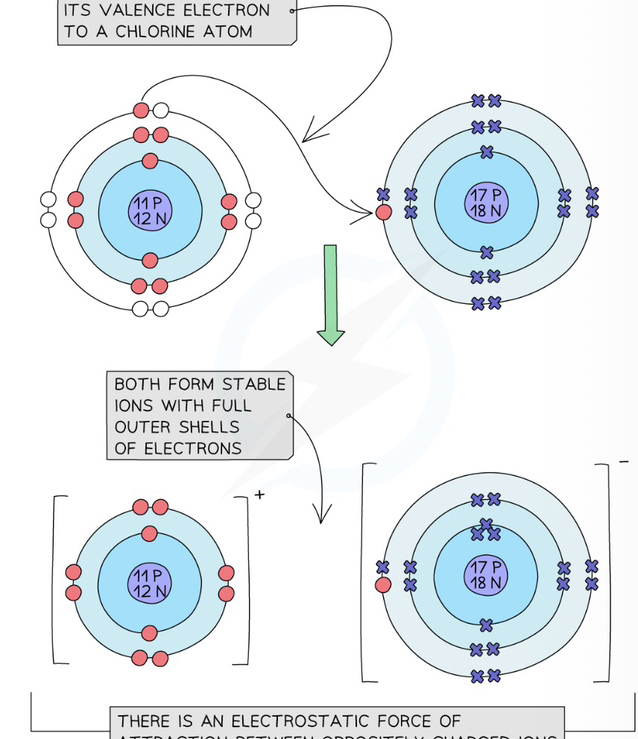
E.g Sodium is a group 1 metal: 2 Na + Cl 2 —> 2 NaCl
Calcium is a group 2 metal: Ca + Br 2 —→ CaBr2
Formation of sodium chloride
Non-metal halides
the halogens react with non-metals to form simple molecular covalent structures
For example, the halogens react with hydrogen to form hydrogen halides (e.g hydrogen chloride)