Lecture Notes - Carbohydrates and Energy
What We Learned Last Lecture
Energy is required by all cells to maintain cell viability and to build new biological macromolecules for reproduction.
Cells bring energy from the external environment and convert it to a usable form; adenosine triphosphate (ATP) is a major molecule for energy storage.
Heterotrophs obtain energy from imported organic molecules.
Catabolism breaks down molecules to regenerate ATP and provide materials for anabolism (building new macromolecules).
Cells are thermodynamically open systems.
Enthalpy (H) is the total energy in the cell and its surroundings.
Gibb’s free energy (G) is the usable energy from catabolic reactions.
Free energy change () can be released (exergonic, ) or required (endergonic, ).
Entropy (S) is unusable energy; a positive means less ordered products and is increased by temperature (-T).
The overall free energy equation is:
Most ATP regeneration occurs during oxidative phosphorylation.
Catabolic oxidation of inorganic (lithotrophic) or organic (heterotrophic) molecules provides electrons to power oxidative phosphorylation.
In eukaryotic cells, 24 electrons are harvested from one glucose molecule via glycolysis, pyruvate oxidation, and the citric acid cycle.
These electrons are carried by NAD+ (20 electrons) and FAD (4 electrons), with each molecule carrying 2 electrons.
Electrons from NADH and FADH2 feed into electron transport proteins in the plasma membrane (prokaryotes) or inner mitochondrial membrane (eukaryotes).
The free energy released () as electrons move through these proteins drives ions (protons) across the membrane against their gradient.
In aerobic cells, two electrons are placed onto oxygen atoms to generate , which combines with protons to form water.
The removal of electrons allows more electrons to enter the chain, reforming the ion gradient.
The proton gradient's free energy drives the ATP synthase complex rotation.
This rotation directly provides the energy to form a covalent bond between adenosine diphosphate (ADP) and inorganic phosphate (Pi), forming ATP.
Oxidative phosphorylation generates 34 of the 38 ATP molecules from complete glucose oxidation.
The four macromolecule groups in cells are proteins, lipids, carbohydrates, and nucleic acids.
We have studied the structures and functions of lipids and proteins.
We have not yet studied carbohydrates and nucleic acids.
Understanding the structure and function of carbohydrates, especially glucose, is important.
Understanding catabolic pathways for glucose oxidation and ATP regeneration is important.
Enzymes are essential proteins that increase the rate of most reactions in cells.
Carbohydrates: Monosaccharide Structure
Carbohydrates contain carbon, hydrogen, and oxygen.
Monosaccharides (simple sugars) are the monomeric units of all carbohydrates.
Most monosaccharides have a carbon:hydrogen:oxygen ratio of 1:2:1 (CnH{2n}On)).
Disaccharides form by covalently bonding two monosaccharides.
Polysaccharides are chains of covalently attached monosaccharides.
Basic Structure of Monosaccharides
Monosaccharides must have at least 3 carbon atoms.
All monosaccharides have a single carbonyl and two or more hydroxyl functional groups on different carbon atoms.
Sugars are defined by:
Number of carbons: trioses (3), pentoses (5), hexoses (6).
Position of the carbonyl group: terminal (aldose) or internal (ketose).
Orientation of the hydroxyl group furthest from the carbonyl group.
Nomenclature
Defined by:
Number of carbon atoms [trioses (3), pentoses (5) and hexoses (6)]
Carbonyl group position: terminal (aldose) or internal (ketose).
Carbon #1 is the terminal carbon with a carbonyl or the closest to the carbonyl.
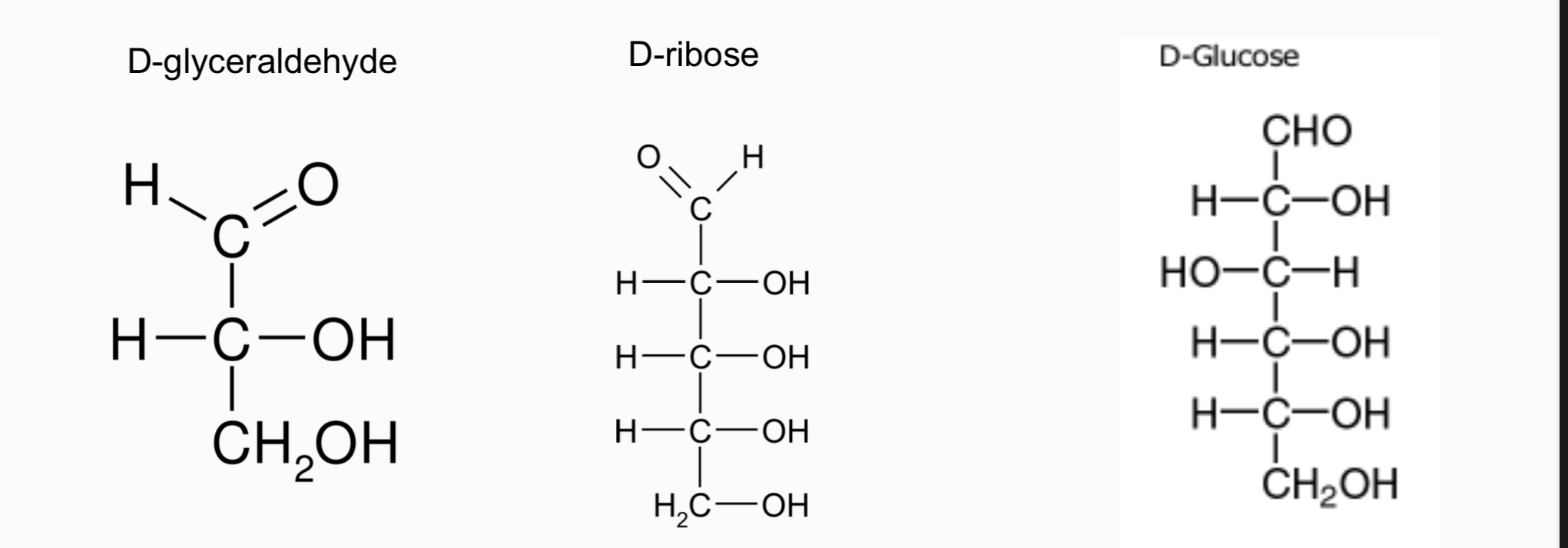
In D-sugars, the hydroxyl on the chiral carbon furthest from the carbonyl is oriented as in D-glyceraldehyde.
Examples include:
D-glyceraldehyde: Intermediate product of the glycolysis and photosynthesis
D-ribose-Required for ATP synthesis, DNA, and RNA.
D-glucose: Main circulating energy source in mammals, stored as polymers in muscle and liver; important intermediate in glycolysis and photosynthesis.
D-glyceraldehyde: triose
D-ribose: pentose
D-glucose: hexose
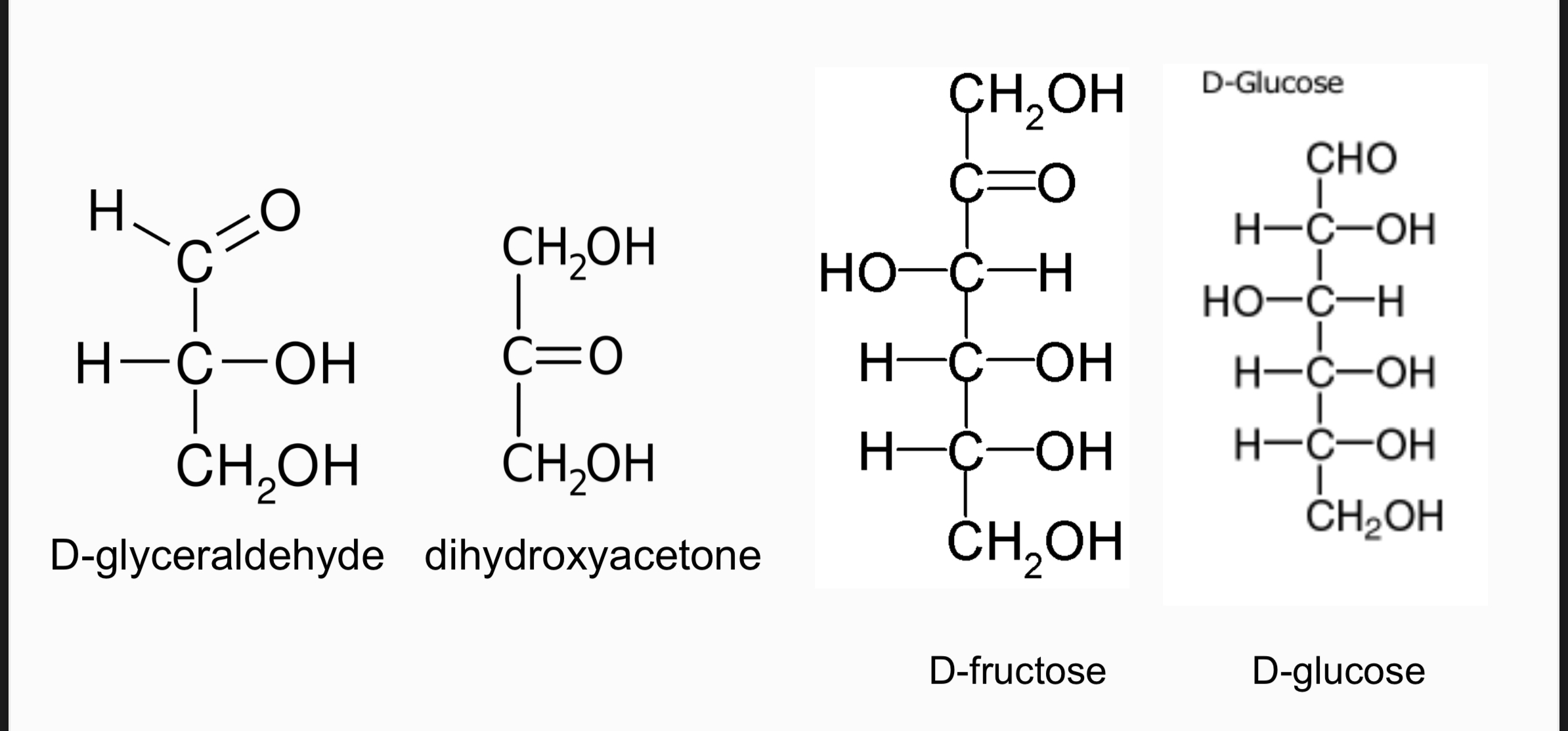
D-glyceraldehyde: triose aldose (C3H6O3)
dihydroxyacetone: triose ketose (C3H6O3)
D-glucose: hexose aldose (C6H{12}O6)
D-fructose: hexose ketose (
C6H{12}O6)
Numbering carbons:
In D-glyceraldehyde and D-glucose, the terminal carbonyl is carbon #1; subsequent carbons are numbered sequentially.
Phosphate bonded to the hydroxyl oxygen on the carbon atom furthest from the carbonyl group in glucose would be labelled as glucose 6-phosphate.
In D-fructose, carbon #1 is the terminal carbon closest to the carbonyl since it is a ketose sugar.
Dihydroxyacetone carbons are unnumbered because the carbonyl group is equidistant from both terminal carbons.
Ketoses: Dihydroxyacetone, Ribulose.
Aldoses: Fructose, Glyceraldehyde, Ribose, Glucose, Galactose.
Trioses (C3H6O3)
Pentoses (C5H{10}O5)
Hexoses (C6H{12}O6)
Structral isonomers are compounds with the same molecular formula but different structural arrangements.
Optical isomers where the structures are non-superimposable mirror images of each other .
Glycoysis: Glucose—- glucose 6-phosphate—-frutose 6-phosphate— frutose 1-6bisphonphate
Chirality
Most simple sugars have one or more chiral carbons.
A chiral carbon is bonded to four different groups.
A carbonyl carbon is not chiral because it is attached to only 3 groups.
The carbon in is not chiral because it is attached to 2 separate hydrogen atoms.
Sugars with group position differences at a chiral carbon have stereoisomers (enantiomers or optical isomers).
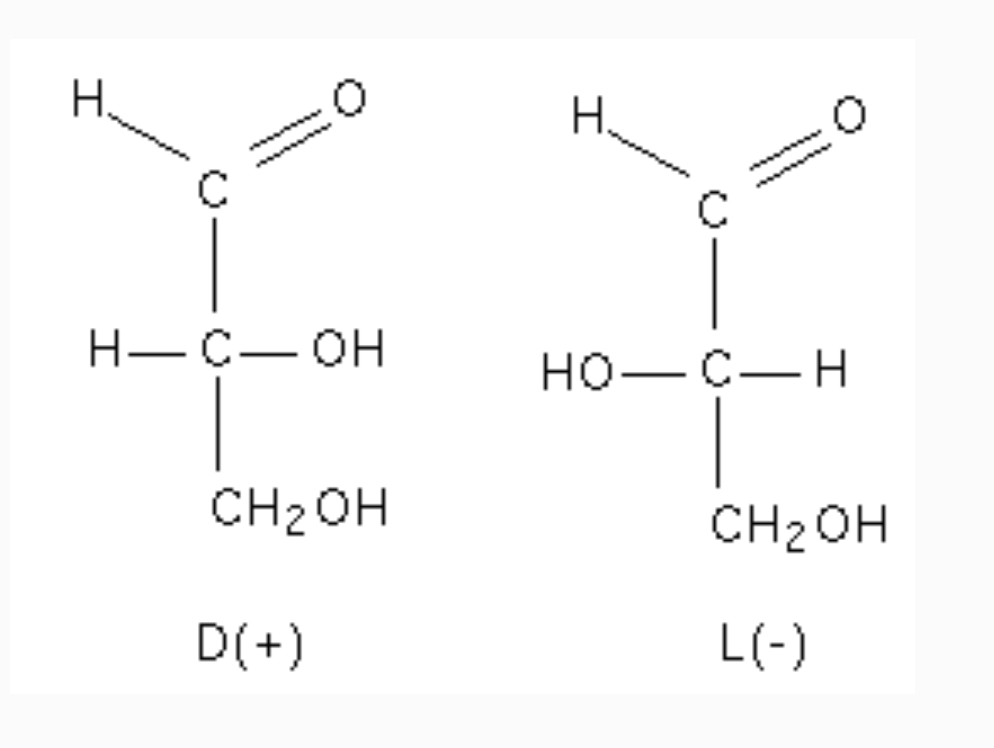
D- vs. L- designates enantiomers at a specific carbon position in the simple sugar.
Chiral Carbons
Chiral carbons have four different covalently bonded groups.
Glyceraldehyde is the simplest example of a monosaccharide with an asymmetric carbon.
If the hydroxyl is to the right, it is D-; if to the left, it is L-.
Most biologically active sugars are in the D-conformation.
Designations
If the hydroxyl is to the left in the Fischer projection, the sugar is L-.
If the hydroxyl is to the right in the Fischer projection, the sugar is D-.
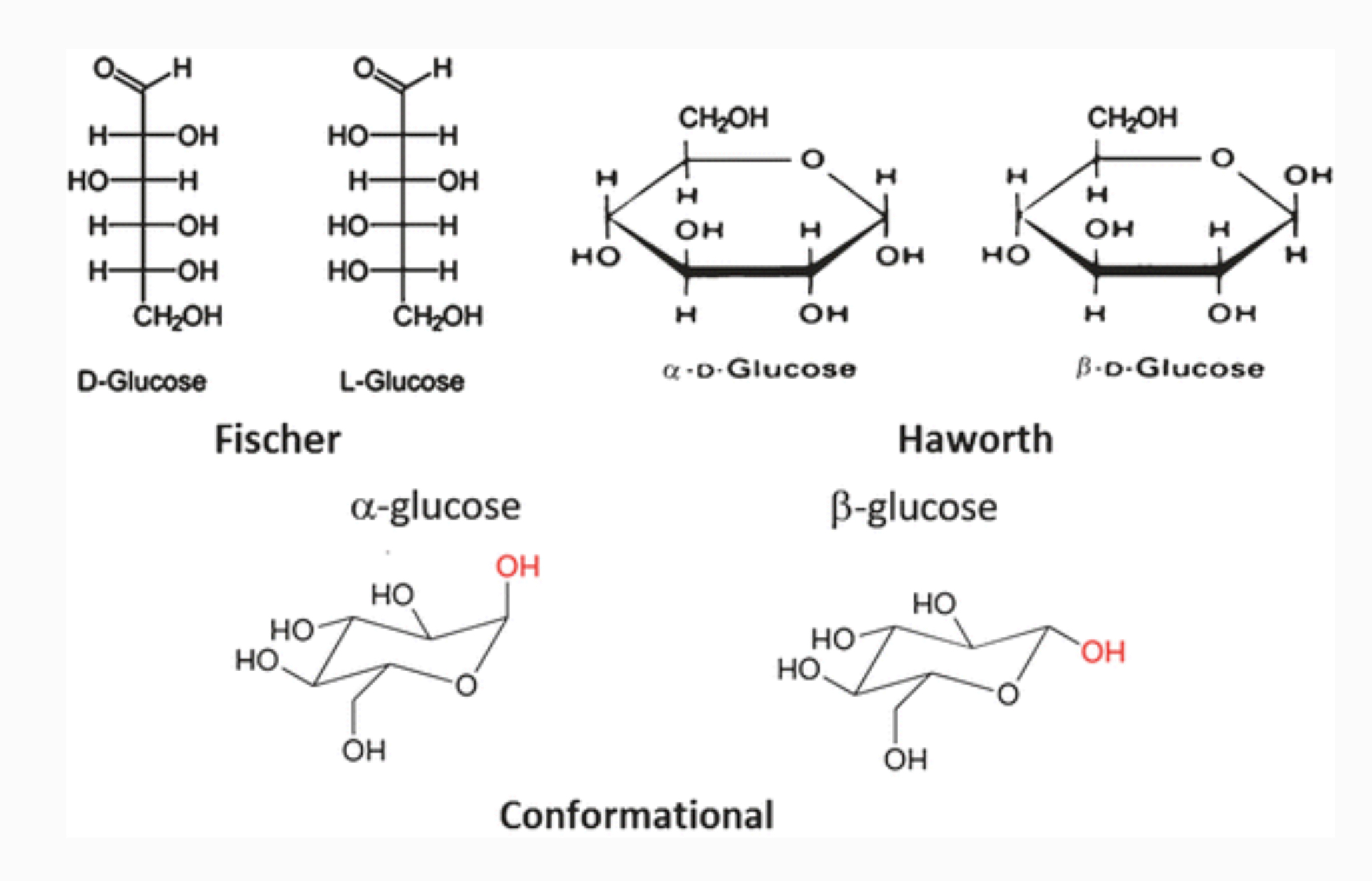
Representations of Glucose
Linear structures are Fischer projections (D- or L-glucose).
Sugars with 5+ carbons cyclize in aqueous solution to form ring structures (Haworth projections, D-glucopyranose and L-glucopyranose).
Ring structures can also be represented as conformational projections.
Drawing Haworth Structure of Glucose
Draw a six-member ring with 5 carbon atoms and 1 oxygen atom.
Label carbons 1’ through 5’ clockwise, starting to the right of oxygen.
For D-glucopyranose, place the group of carbon 6’ in the up position from carbon 5’.
Place the hydroxyl on 4’ down, 3’ up, and 2’ down; carbon 5 has no hydroxyl group in the ring.
Atoms on carbons without 4 bonds are hydrogen.
For -D-glucopyranose, the hydroxyl on carbon 1 points below the ring; for -D-glucopyranose, it points above.
From Haworth to Fischer Structure
Draw six covalently connected carbon atoms in a linear arrangement.
Since glucose is an aldose, place a carbonyl and hydrogen atom on carbon 1 and on carbon 6.
If the hydroxyl is down in the Haworth projection, place it to the right in the Fischer projection; if it is up, place it to the left.
Since this is D-glucose, the hydroxyl on carbon 5 is to the right.
The remaining positions on the carbons are hydrogen atoms.
Ring Formation
The most stable conformation of simple sugars with 5+ carbons in aqueous solution is as closed rings.
In glucose, the carbonyl carbon forms a bond with the oxygen on the hydroxyl group of carbon 5 to form a hemiacetal; the oxygen at carbon 5 incorporates into the ring.
In glucose, when the ring closes, the carbonyl oxygen at carbon 1 converts to a hydroxyl group.
In the alpha () position, the hydroxyl is below the ring in the Haworth projection.
In the beta () position, it is above the ring in the Haworth projection.