Properties of Ionic Compounds
Structure of Ionic Compounds
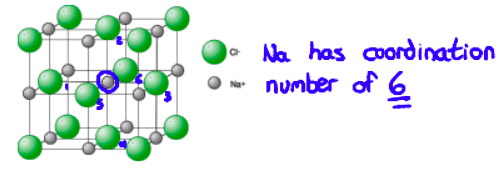
Ionic compounds exist as three-dimensional lattice structures. The structure is based around a repeating unit (the formula unit)
The coordination number is used to express the number of ions that surround a given ion in the lattice.
 Lattice enthalpy is a measure of the strength of the forces between ions in an ionic solid.
Lattice enthalpy is a measure of the strength of the forces between ions in an ionic solid.
It depends on…
ionic radius (smaller radius, stronger attraction)
charge of ions (higher charger, stronger attraction
The physical properties of ionic compounds reflect their lattice structure
Melting and Boiling Points
Ionic compounds tend to have high melting and boiling points due to the strength of the electrostatic attractions between the ions in the lattice.
The melting and boiling points are generally higher when the charge on the ions is greater.
Volatility
the tendency of a substance to vaporize
Substances with high volatility are more likely to exist as a vapor
Ionic bonds are very strong, require large amounts of energy to overcome
At room temperature, ions do not have enough energy to break away
ionic compounds have no odour
Solubility
the ease with which a solid (the solute) becomes dispersed through a liquid (the solvent) to form a solution.
Dissolving in water involves the attraction of polar water molecules to the oppositely- charged ions in the ionic lattice, and the hydration of the separated ions
Electrical Conductivity
Two things are needed for electrical conductivity
1. Presence of charged particles
2. Charged particles must be free to move
Solid Ionic Lattice
No conduction because no freely moving charged particles, ions are
locked into position