Chemistry of the atmosphere
Evolution of the atmosphere
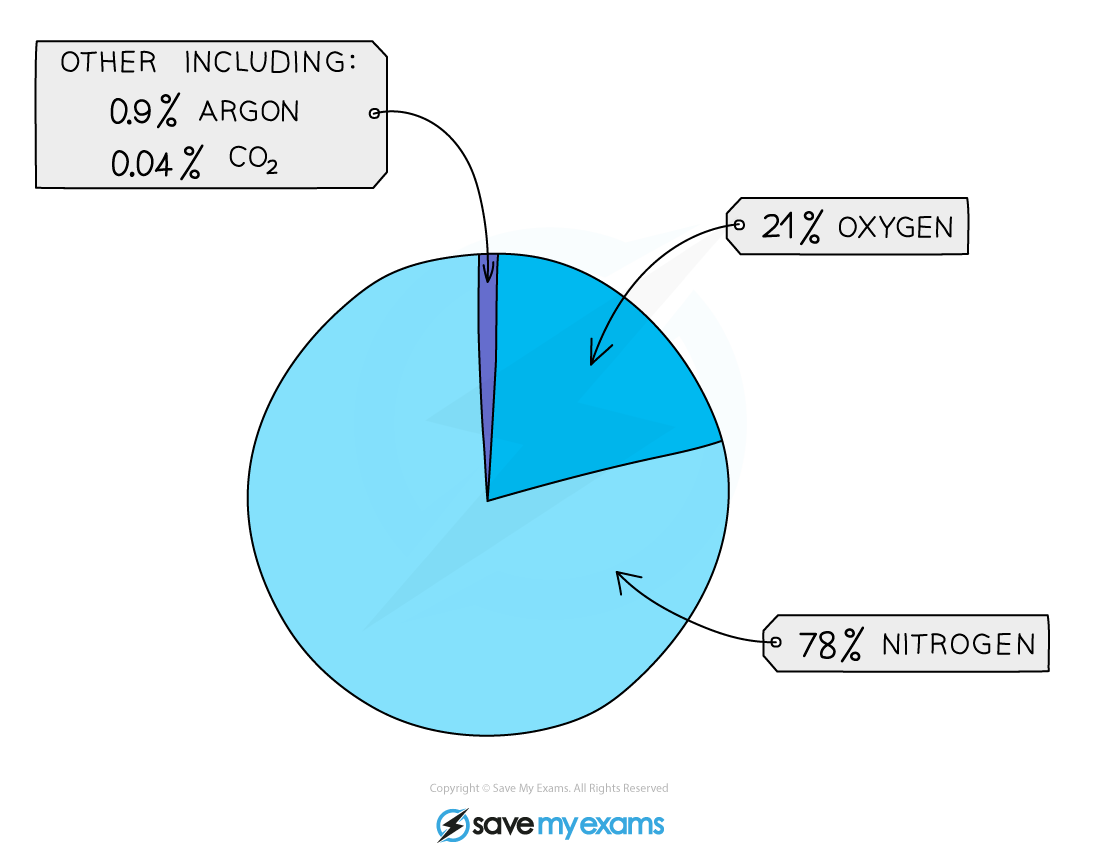
In the first billion years, the earth was very dry and had intense volcanic activity
This created lots of carbon dioxide, water vapour and nitrogen and smaller amounts of methane and ammonia
The early atmosphere was mainly carbon dioixde
Water vapour then condensed to form oceans, in which lots of carbon dissolved
This formed carbon precipitates and eventually sediments
2.7 billion years ago algae began to appear slowly, and then evolved to create lots of green plants rapidly
These plants could photosynthesise - CO2 + H2O → C6H12O6 + O2
CO2 levels decreased as it was taken in
O2 levels increased as it was released (levels built up)
From this oxygen, more complex life forms evolved - animals
Plants can only hold small amounts of carbon in their bodies
When they died, they settled in the seabed and became buried by layers of sediment
Over millions of years, it compressed to form sedimentary rock, oil and gas with trapped carbon
This prevented the release of carbon into the atmosphere
The substance that was formed from dead matter depends on what organism was buried originally
Crude oil and gas - mainly from dead plankton
Coal - a sedimentary rock, mainly from dead plant material
Limestone - a sedimentary rock, from calcium carbonate (in shells and skeletons)
Greenhouse gases and climate change
Around the earth, there is a trapped layer of gases that form the atmosphere
These gases act as an insulating layer (a greenhouse)
It traps the perfect amount of the sun’s thermal energy so earth is a warm and stable temperature
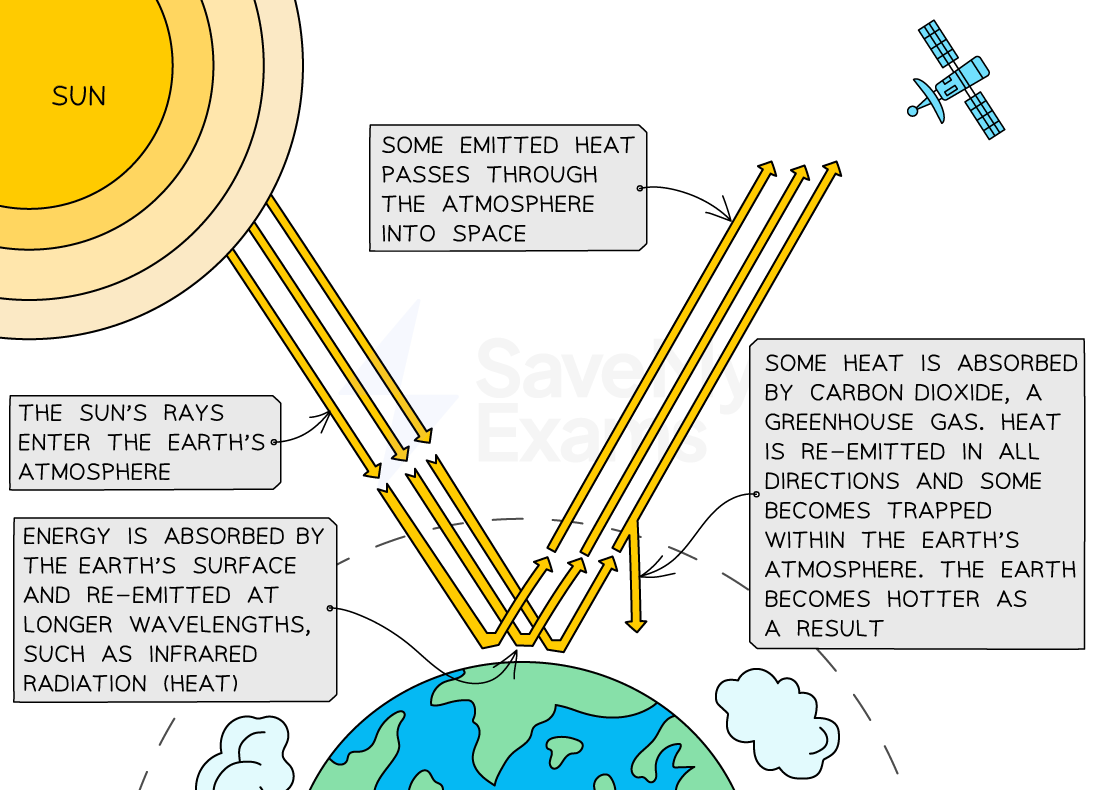
Short wave length radiation is emitted from the sun, part of which is absorbed into the earth, but most is re-emitted/reflected back to space
This energy is re-emitted as long wave length radiation, some of which makes it back to space but most is absorbed by particles of gas in our atmosphere
After a short delay, energy is re-emitted again from the particles, either towards space or the earth
Most of this energy is absorbed by gas particles, and then re-emitted - this cycle repeats
This means the heat energy remains close to earth - maintains a warm and stable temperature
Carbon dioxide, methane and water vapour are all greenhouse gases
The warming they cause is called the greenhouse effect
Because humans are producing lots of these gases, their concentration is increasing
Increases the greenhouse effect, which leads to global warming
Carbon dioxide increase is caused by:
Deforestation - less photosynthesis to remove CO2
Burning fossil fuels - releases stored carbon
Methane increase is caused by:
Farm animal’s digestion
Huge amounts of waste - releases methane as it decomposes
There are lots of variables that add to global warming, so its difficult to predict what will happen
The impact it causes is called climate change
Climate change is the effect of global warming on long term weather patterns
Rare weather events are becoming more common and severe
Droughts, floods, hurricanes
Sea levels are rising due to ice melting into oceans
Water temperature is increasing which causes seasonal flooding and potential submersion of places
General effect on biodiversity as species might not adapt to new conditions fast enough
Some species may adapt or migrate, but its not common
Carbon footprints
A carbon footprint is the total amount of carbon dioxide and other greenhouse gases emitted over something’s entire lifecycle
A product, service or event
The benefits to calculating carbon footprints are:
Can calculate if something is something is worth the cost of how environmentally damaging it is
Can identify the most polluting activities
The issues with calculating carbon footprints are:
It is hard to measure carbon footprint accurately
Have to take materials used and where they were sourced, the manufacturing process and it’s end disposal, as well as transport, etc.
It is only a rough estimate, but we can get an idea on what is best/worst
Reducing greenhouse gas emissions
Using renewable energy sources
More efficient manufacturing processes
Less energy use
Less waste (can decompose and release methane)
Governments
Can make new laws
Can tax companies for greenhouse gas emission
Carbon capture technology
Captures carbon dioxide from burning fossil fuels and it stores it underground
Currently, renewable energy is more expensive and everything is built on fossil fuels (cars, machinery, etc.)
Governments worry if they prioritise the environment that the economy may suffer and international agreements become difficult
Air pollution
Burning fossil fuels produce carbon dioxide and water, as well as releasing lots of energy
This only happens when pure hydrocarbons are burnt with plenty of oxygen - allows for complete combustion
This doesn’t always happen, and instead pollutants are produced (carbon monoxide, soot, sulphur dioxide, nitrogen oxides)
Hydrocarbon + excess oxygen → water = carbon dioxide
Complete combustion
When three isn’t enough oxygen present, incomplete combustion occurs
Carbon monoxide and particulates (solid carbon particles - soot) are formed
Particulate issues
If inhaled, it can damage lungs and cause respiratory problems
It can form dark clouds (smog), so sunlight is reflected back to space - less light on earth
This is called global dimming
Carbon monoxide issues
It is very dangerous
If it is breathed in, it diffuses into the blood stream and binds to haemoglobin
This means less oxygen can be carried, so less is transported around the body
It can lead to fainting, comas and death
It is colourless and odourless, so hard to detect
Some hydrocarbons that are burnt have impurities
For example, sulphur which gets oxidised when burnt to form sulphur dioxide (SO2)
When the oxygen formed reacts with nitrogen in the air, nitrogen oxide is formed
Only happens at very high temperatures, so unusual
Can happen in internal combustion engines (cars)
When sulphur dioxide and nitrate oxides are formed, they cause lots of issues
When breathed in, they can damage lungs and cause respiratory problems
As the gases mix with cloud, they dissolve to form dilute sulphuric acid or dilute nitric acid
This falls as acid rain
Acid rain issues
It corrodes metals
Damages buildings and statues
Damages plants - either direct contact or through soil becoming acidic (difficult to grow)
If in water (oceans, rivers), fish can ingest it and become ill
DONE!!!