ap chem final
rate laws:
determine the rate law experimentally
determine k
determine the rate determining step from proposed mechanisms
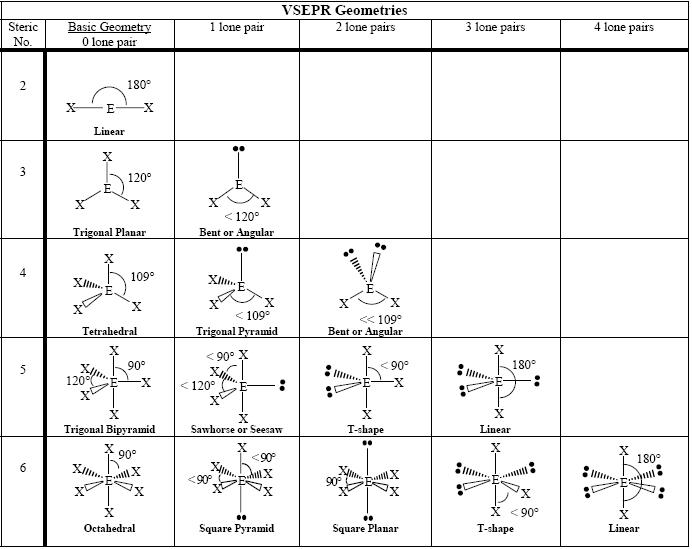
lewis structures:
bond angles
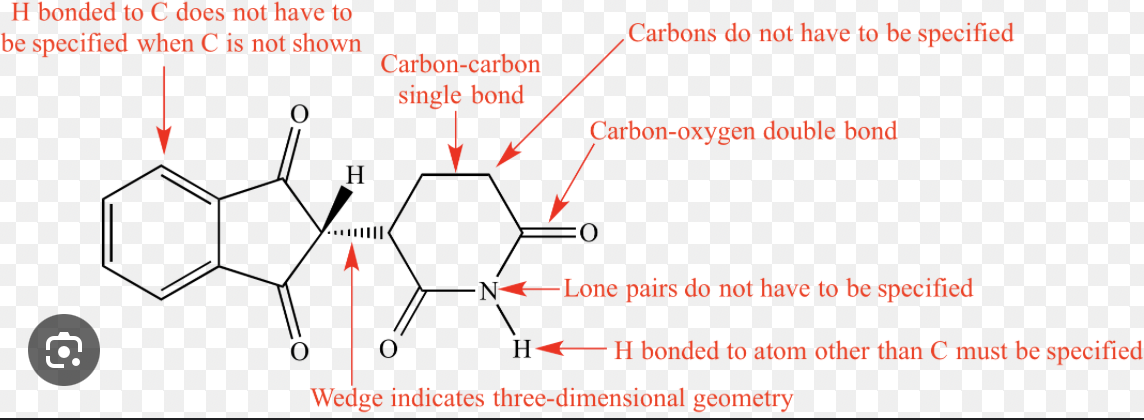
isomers
molecules with the same formula but different arrangements of atoms
structural formula from lewis structure
pi and sigma bonds in lewis structure
Sigma bonds(σ) are always the first bond between two atoms - a single bond
Pi bonds(π) are second and third bonds resulting from the overlap of p orbitals
single bond
1 sigma bond
double bond
1 sigma, 1 pi bond
triple bond
1 sigma, 2 pi bonds
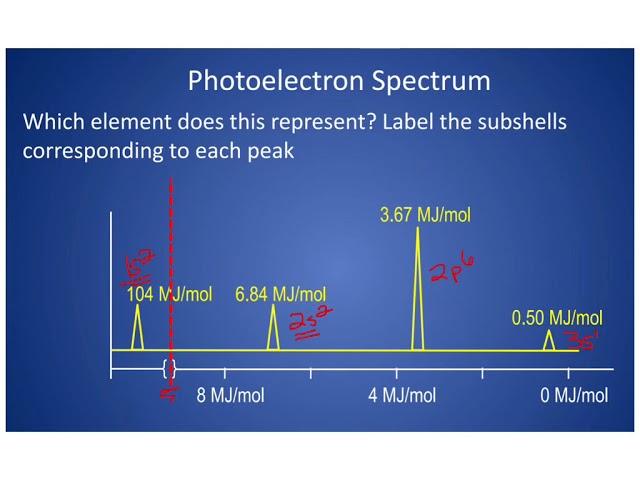
photoelectron spectroscopy:
binding energy graphs
formal charges:
which structure minimizes charge