Enolate Alkylation
Purpose: create a new C-C bond
A carbonyl can be both an electrophile and a nucleophile
Electrophile - direct addition to carbonyls
Nucleophile - base creates an enolate, and the enolate attacks an electrophile
Enolate Alkylation is a two step reaction
Base removes a hydrogen
Strong base - complete formation of the anion
Add the base first, then add the electrophile in a second step
Although more demanding, the base and electrophile never meet; therefore, compatibility is not an issue.
Weak base - equilibrium lying towards the starting material
Add base and electrophile in one step
This approach is easier however, the electrophile and base must be compatible and unreactive with each other
Sn2 on an alkyl halide
Carbonyl Compounds, Nitriles, and Nitro Alkanes can all be alkylated
Carbolyls - see above
Nitriles
The deprotonated nitrile will not react with other nitriles but will be very reactive with alkyl halides.
This reaction is conducted with a phase change catalysis
More on below
The S character makes it easy to perform Sn2.
Partial deprotonation works
This compound is so nucleophilic that it will work when a quaternary center is being formed.
If two equivalents of base are added, such as NaH, alkylation can happen more than once.
If double alkylation occurs with two equivalents of base and nucleophile in excess quantities, the monoalkylated product can not be isolated and will go straight to the dialkylated product
If there are two nitrile groups, even a neutral amine such as Et3N will deprotonate
If a nucleophile is in the same molecule and the spacing is appropriate, intermolecular Sn2 will occur.
The base and nucleophile are in solution together, but the reaction is so fast that it does not matter
Nitroalkanes
Protons adjacent to a nitro group are as acidic as one adjacent to two carbonyls thus, deprotonation is fairly easy even with weak bases
This reaction can be done in a single step under phase change conditions to prevent alcohol creation.
This reaction will create quaternary carbons in the product
This reaction can cyclize however, weak bases such as potassium carbonate (K2CO3) must be used
Phase Transfer Catalysis
The reaction is carried out in a two-phase mixture (aqueous + organic)
done to prevent hydroxide and nucleophile from creating an alcohol
To make enough hydroxide enter the organic layer to deprotonate we need a phase transfer catalyst such as BnEt3N+Cl-
Electrophile reactivity for alkylation - Because Sn2 is needed, the more branched halides will prefer the E2 reaction and thus are useless here
Best reactivity
Methyl
Allyl
Benzyl
Decent
Primary alkyls
OK
Secondary alkyls
None
Tertiary alkyls
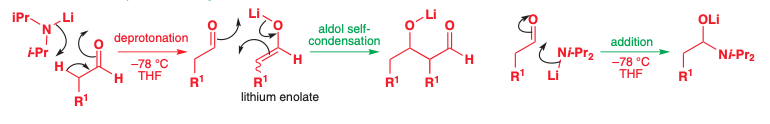
Lithium Enolates of Carbonyl Compounds
very stable and are among the best to use for alkylation reactions
LDA will deprotonate almost all ketones and esters that have an acidic proton
This will occur quickly, cleanly, and irreversibly if done at low temperatures (-78 C)
Deprotonation is a cyclic mechanism where the basic nitrogen Anion takes a hydrogen, the H-C sigma bond becomes a C-C pi bond, and the C-O pi bond becomes an O-Li ionic bond
Alylation of Lithium Enolates
Alkylation of lithium enolates works on both acyclic and cyclic ketones and esters
The reaction is done at low temperatures (-78 C) to minimize self-condensation.
The electrophile is added only after full enolate formation.
Once the electrophile is added, the temperature slowly warms up to speed up the rate of Sn2
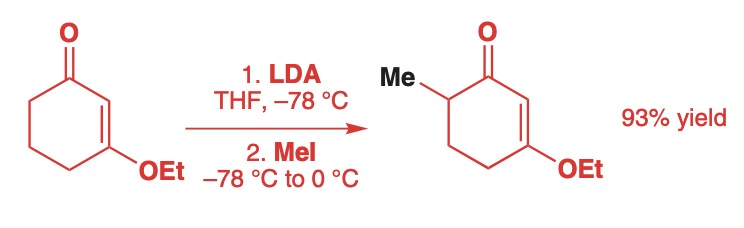
Alkylation of Ketones
Low temperature stabilization makes lithium enolates preferred, but sodium and potassium enolates are also possible.
These enolates are less stable but more reactive.
Very strong sodium and potassium bases are required
Na: NaH, NaNH2, NaHMDS
K: KH, KNH2, KHMDS
This instability of these enolates means that the electrophile and base must be added in one step.
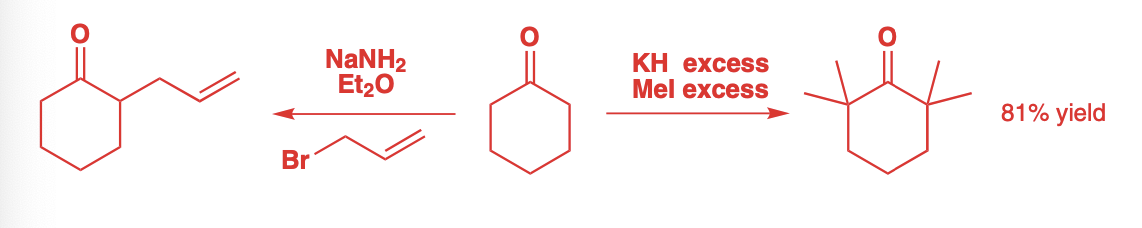
Alkylation of Esters
Claisen condensation - see next chapter
To avoid the Claisen condensation
Add the ester to a solution of LDA
Make the R group as large as possible
t-butyl esters are particularly good
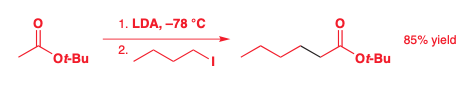
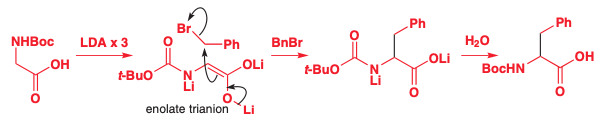
Alkylation of Carboxylic Acids
Lithium enolates can be created from carboxylic acids if there are two equivalents of acid
The first deprotonates the alcohol
The second deprotonates the carbon
The first base can be any, but the second must be strong, so often it’s easiest to just use two equivalents of LDA.
Butyl lithium (BuLi) can be used due to the lithium carboxylate is much less electrophilic than an aldehyde or ketone
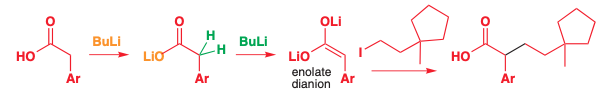
When there are multiple acidic hydrogens, such as OH or N,H then the equivalents of acid must equal # of acidic hydrogens + 1
Alkylation will always occur at the least stable anion thus keeping the two more stable charges
Why do enolates alkylate C
Carbon has a greater HOMO and is softer
Oxygen has a greater total charge and is harder
Alkyl halides are soft and will react at the carbon
TLDR:
Hard = O
Alkyl sulfates, sulfonates
Soft = C
Alkyl halides (I > Br > Cl)
Polar Protic Solvents = O alkylation
DMSO, DMF
Etheral solvents = C alkylation
THF, DME
Larger Alkali metals (Cs > K > Na > Li) react at O
Alkylations of aldehydes
Avoid LDA
Not fast enough to outpace aldol reactions
Direct addition of base to carbonyl group is also a problem.
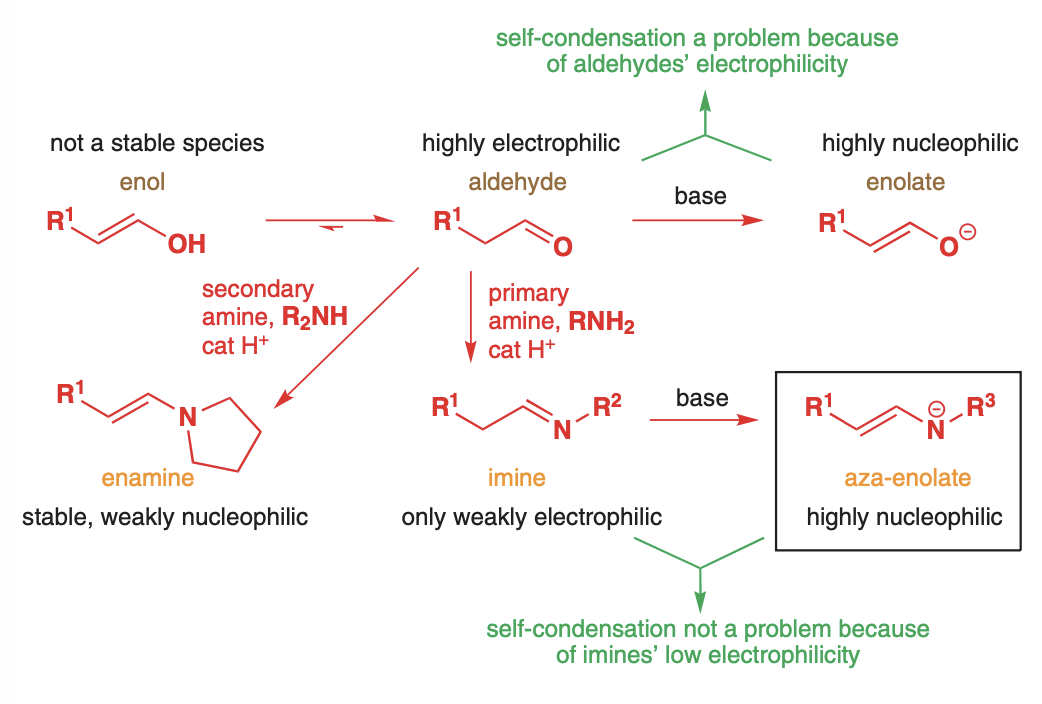
3 main ways to make an aldehyde or ketone enol
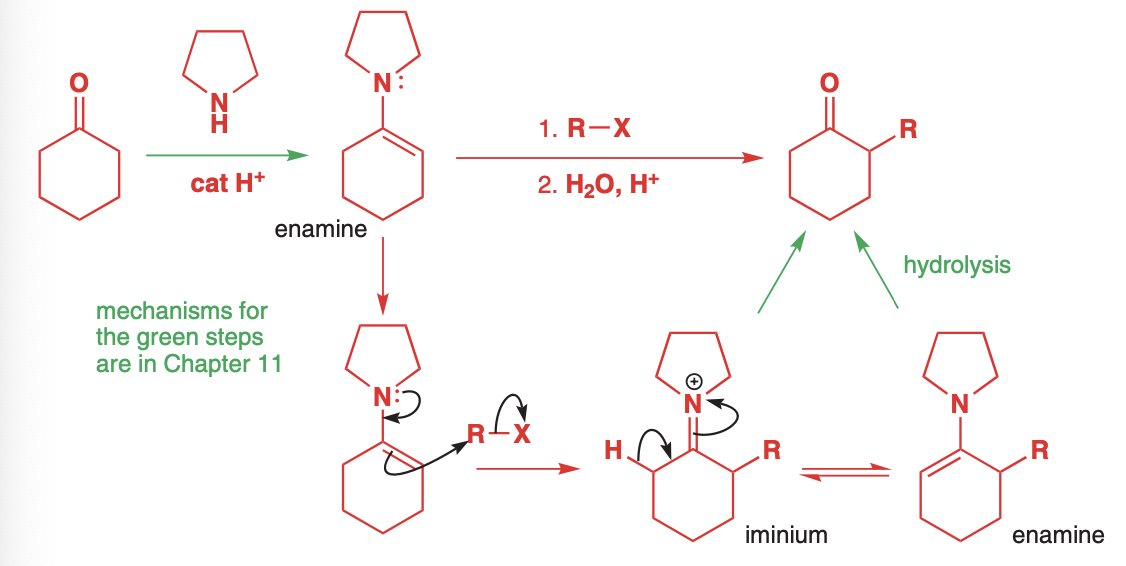
enamines
Aldehydes/ketones react with secondary amines
The overall process is an enolate alkylation; however includes no strong bases or enolates therefore there is no self-condensation.
The lower reactivity, however, dictates that the reaction be done for a long time and at high temperature
The choice of secondary amine is not arbitrary
Cyclic amines such as pyrrolidine, piperidine, and morpholine are common as they increase nucleophilicity and “hold back” the alkyl groups. They also have a higher boiling point.
Alpha-bromocarbonyl compounds work well for Sn2 and will react well with the weak enamines
When forming enamines, the less substituted enamine is most likely
Due to thermodynamic control, the less sterically hindered enamine is more favored; however, less stable
What alkylating agents one uses is also very important.
Simple alkyl halides like MeI will create an ammonium salt which will return the original reactants.
Reactive alkylating agents, allylic halides, benzyl halides, and alpha-halo carbonyl compounds are much more likely to C-alkylate vs N-alkylate.
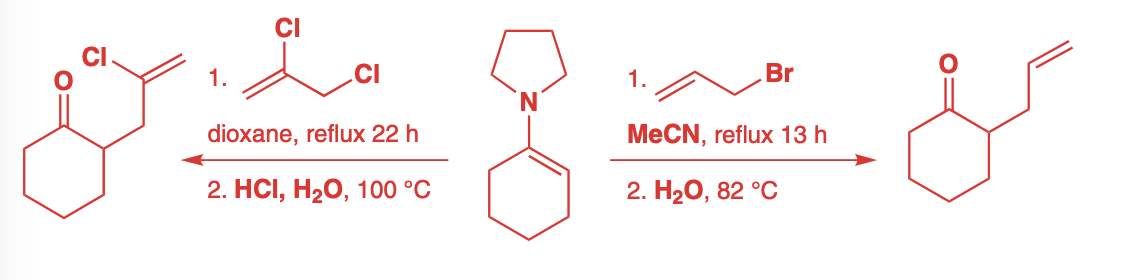
silyl enol ethers
Due to the basicity of aza-enolates, Sn1 reactions will not work
If one wanted to add something like a tertiary alkyl halide, silyl enol ethers must be used.
The silyl enol ethers decrease reactivity therefore, they must be reacted with a carbocation
Tertiary alkyl halides are best as lewis acids such as TiCl4 or SnCl4 can remove the halide to create a tertiary carbocation
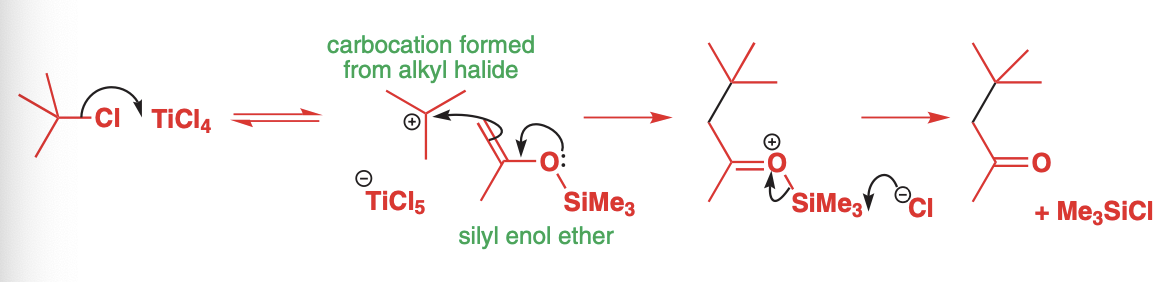
aza-enolates derived from imines
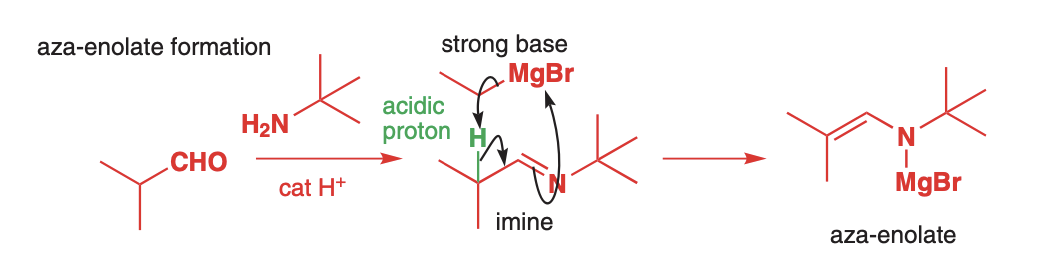
Attach a primary amine to an aldehyde or ketone to create an imine and then treat with base to create the aza-enolate
There is no worry about condensation due to the weak electrophilicity of imines
The aldehyde is alkylated with a bulky primary amine such as t-butyl or cyclohexylamine to discourage further nucleophilic attack at the imine carbon. Then LDA or a grignard is used to deprotnate and give an aza-enolate
Aza-enolates react the same as ketone enolates with Sn2 alkylating agents
This process works so well that it has been extended to ketones
TLDR:
Lithium enolates can be used for Sn2 electrophiles but do not work with aldehydes
Aza-enolates can be used with the same Sn2 electrophiles and can work with aldehydes
Enamines of aldehydes or ketones can be used with allylic, benzylic, or alpha-halocarbonyl compounds
Silyl enol ettheres of aldehydes or ketones can be used with Sn1 electrophiles such as allylic, benzylic, or tertiary alkyl halides.
Alkylation of beta-dicarbonyl compounds
The presence of a second or even third electron-withdrawing group will make the remaining proton(s) so acidic that even weak bases such as alkoxides can deprotonate
A diketone can be enolized by something like potassium carbonate and will react with MeI in high yield.
K2CO3 is so weak as a nucleophile that the base and electrophile can be added in one step
Need to know two important beta-dicarbonyls
diethyl (or dimethyl) malonate and ethyl acetoacetate
The choice of base is important
Choose the alkoxide identical with the alkoxide of the ester i.e. ethoxide for diethyl malonate
There are multiple electron withdrawing groups that can be used with success, not just carbonyls