Cell Junctional Communication
Overview:
Cell junctions are specialised structures that physically and functionally connect cells to one another and to the extracellular matrix (ECM).
Understanding them is essential to explain how tissues:
Maintain structure and integrity.
Communicate signals.
Coordinate during development, repair, and morphogenesis.
Cells are not isolated. In multicellular organisms, cells continuously interact:
Cell–cell interactions (via junctions between adjacent cells).
Cell–ECM interactions (via integrins and focal adhesions).
These interactions regulate:
Tissue organisation.
Cellular polarity.
Signalling and mechanotransduction (conversion of mechanical forces to biochemical signals).
Cellular Context and the Extracellular Matrix (ECM):
ECM = a complex meshwork of secreted proteins (e.g., collagen, elastin, laminin, fibronectin) and polysaccharides (e.g., proteoglycans).
Provides structural support, anchorage, and biochemical cues.
Cells interact with ECM through cell–matrix junctions, while cell–cell interactions occur via junctional complexes.
Together, these determine tissue architecture and dynamic response to the environment
Evolutionary Origin and Functional Significance:
Multicellularity (~600 million years ago) required mechanisms to:
Distinguish inside vs outside environments.
Keep cells together in defined tissue structures.
Cell junctions evolved to:
Maintain tissue cohesion.
Allow communication between cells.
Support morphogenesis (formation of structures like epithelial tubes).
Junctions are not static “glues”; they are dynamic and responsive to cellular and mechanical cues.
Categories of Cell Junctions:
There are two primary categories based on what cells attach to:
Cell–cell junctions – connect adjacent cells directly.
Cell–matrix junctions – link cells to ECM components.
Within cell–cell junctions, three functional types exist:

Molecular Basis of Cell Junctions:
All junctions rely on transmembrane proteins that:
Span the lipid bilayer.
Have extracellular domains that bind to other cells or ECM.
Have intracellular domains linked to adaptor proteins and cytoskeletal elements.
Adaptor proteins connect these membrane proteins to:
Actin filaments.
Intermediate filaments.
Signalling molecules (e.g., kinases, GTPases).
Binding Types Between Cells:
Homophilic binding: The same type of transmembrane protein on each cell interacts (e.g., E-cadherin–E-cadherin).
Heterophilic binding: Different transmembrane proteins or carbohydrate motifs interact (common in cell–ECM junctions).
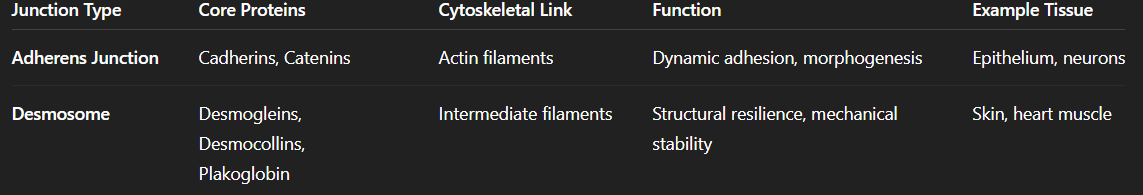
Adherens Junctions (Zonula Adherens):
Overview:
Prominent in epithelial tissues, forming a continuous adhesion belt (zonula) near the apical surface.
Anchor actin filaments from one cell to those of another.
Provides mechanical cohesion and allows tension transmission across tissues.
Key Components:
Transmembrane protein: Cadherins (especially classical cadherins such as E-cadherin).
Intracellular adaptor proteins:
β-catenin and p120-catenin bind directly to cadherins.
α-catenin links the complex to actin filaments.
Additional adaptors (e.g., vinculin) regulate actin recruitment.
Structural Principles:
Extracellular domain:
Contains Ca²⁺-binding sites → calcium stabilises the cadherin structure.
Without Ca²⁺, cadherins are floppy and non-functional.
Intercellular interaction:
Cadherins from neighbouring cells bind via their extracellular domains.
Single interactions are weak; strength arises from arrays (clusters) forming Velcro-like adhesion.
Intracellular domain:
Links to actin cytoskeleton through adaptor complexes → forms contractile actin belts.
Regulation and Dynamics:
Expression levels: Altered cadherin expression modulates adhesion strength (important in development and metastasis).
Mechanotransduction:
Mechanical tension can unfold α-catenin, exposing binding sites for vinculin → recruits additional actin filaments.
This translates mechanical forces into biochemical responses.
Allows one cell’s tension to alter the cytoskeletal state of its neighbour without gene expression changes.
Functional Examples:
Epithelial tube formation (morphogenesis):
Localised increase in actomyosin contractility at adherens junctions causes epithelial invagination.
Example: formation of neural tube or glandular structures.
Neuronal synapse development:
Similar principles used in axon–dendrite recognition.
Nectin-1 (axonal) and Nectin-3 (dendritic) provide specificity.
Cadherins then reinforce the connection once the correct pairing is established.
Desmosomes (Macula Adherens):
Overview:
Another type of anchoring junction, providing mechanical resilience.
Especially abundant in tissues under high mechanical stress (e.g., skin, cardiac muscle).
Link intermediate filaments (e.g., keratin, desmin) between cells to form a supracellular scaffold.
Structural Organisation:
Transmembrane proteins: Desmogleins and desmocollins (non-classical cadherins).
Adaptor (plaque) proteins: Plakoglobin, plakophilin, and desmoplakin.
Cytoskeletal link: Intermediate filaments (rather than actin).
Creates dense plaques seen in electron micrographs as thickened regions of the plasma membrane.
Functional Significance:
Provides 3D structural integrity across cell layers.
Acts like spot welds connecting intermediate filament networks between cells.
Allows tissues to absorb mechanical stress without tearing.
Essential in maintaining tissue cohesion in the epidermis and myocardium.
Regulation:
Although more stable than adherens junctions, desmosomes are not static:
Protein kinase C (PKC) can phosphorylate desmosomal components, modulating adhesion strength.
The number and distribution of desmosomal units adjust in response to tissue stress or remodelling.
Mutations in desmosomal proteins → disease associations:
Pemphigus vulgaris: Autoantibodies against desmogleins cause epidermal blistering.
Arrhythmogenic right ventricular cardiomyopathy (ARVC): Defective desmosomal proteins weaken cardiac tissue.
Integrating Cytoskeletal and Signalling Networks:
Both adherens junctions and desmosomes form part of a larger junctional complex that integrates:
Cytoskeletal architecture.
Intracellular signalling pathways (e.g., β-catenin in Wnt signalling).
Junctions act as mechanosensors and signal transducers.
Converts changes in cell adhesion or mechanical stress into gene expression or cytoskeletal responses.
Zonula Adherens vs Macula Adherens:
Tight and Gap Junctions:
Tight Junctions: Structure and Function:
Role in Epithelial Organisation:
Found predominantly in epithelial tissues, which create functional compartments in the body.
Maintain a permeability barrier between the apical (outer/luminal) and basal (inner) domains.
Enable tissues to be polarised, meaning:
The apical surface faces the lumen (e.g., intestinal cavity).
The basal surface anchors to the basement membrane.
Function: allows controlled exchange — not completely impermeable, but selectively permeable to ions and small solutes.
Morphological Analogy:
Think of epithelial junctions as:
Adherens junctions – “belts” for structural cohesion.
Desmosomes – “spot welds” for tensile strength.
Tight junctions – “zippers” that seal the gaps between cells and regulate passage of molecules.
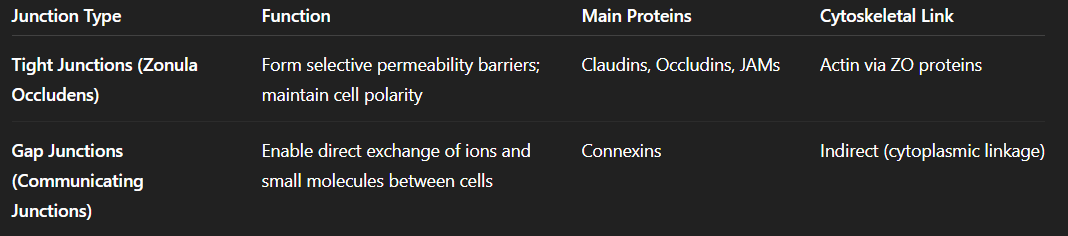
Molecular Composition of Tight Junctions:
Transmembrane Proteins:
Claudins and Occludins are the primary components.
Forms continuous strands that encircle each cell near the apical surface.
Different combinations of claudins create tissue-specific permeability profiles.
Cytoplasmic Scaffold:
ZO proteins (Zonula Occludens-1, -2, -3):
Act as scaffold proteins connecting transmembrane proteins to actin.
Contain multiple PDZ domains for protein–protein interactions.
Bind occludins/claudins, each other, and regulatory enzymes.
Sites of phosphorylation – crucial for dynamic regulation of tight junction assembly and disassembly.
Regulatory Mechanisms:
Phosphorylation by kinases (e.g., PKC, AMPK) alters permeability and protein localisation.
Dynamic modulation allows adjustment of junction tightness in response to:
Hormonal signals.
Metabolic state (e.g., during exercise or stress).
Inflammatory cytokines.
Tight Junction Functions: “Gate” and “Fence”:
Gate Function:
Controls paracellular permeability – passage of solutes between adjacent cells.
Selectivity depends on size and charge of molecules:
Small solutes (e.g., 4 kDa dextrans) can pass.
Larger molecules (e.g., 10 kDa dextrans) are excluded.
Determines tissue-specific barrier tightness:
Tight epithelia (kidney collecting duct, blood-brain barrier) → minimal paracellular transport.
Leaky epithelia (intestinal mucosa, renal proximal tubule) → greater paracellular permeability.
Fence Function:
Maintains cell polarity by restricting diffusion of membrane proteins and lipids between apical and basolateral domains.
Ensures:
Apical and basal membranes have distinct compositions.
Polarity of transporters and receptors is preserved.
Loss of fence function = loss of polarity, a hallmark of epithelial-to-mesenchymal transition (EMT) and tumour progression.
Functional Example: Glucose Transport Across the Intestinal Epithelium:
Context:
Tight junctions underpin directional transport in epithelial cells.
The intestinal epithelium must move glucose from lumen (low concentration) → bloodstream (low concentration), against its gradient.
Mechanism:
Apical membrane:
Contains sodium–glucose symporter (SGLT1) – active transport using Na⁺ gradient to move glucose into cell.
Basolateral membrane:
Contains GLUT2 – passive glucose transporter allowing diffusion into blood.
Tight junctions’ role:
Fence function prevents GLUT2 from diffusing into the apical domain.
Gate function prevents paracellular glucose leakage.
Physiological and Clinical Relevance:
Regulation by kinases (e.g., AMPK during exercise) adjusts epithelial permeability and glucose absorption.
Pathological disruption (inflammation, infection) → “leaky gut” and nutrient malabsorption.
Gap Junctions: Direct Intercellular Communication:
Gap junctions permit direct transfer of ions and small signalling molecules (<1 kDa) between cytoplasms of adjacent cells.
Allows rapid electrical and metabolic coupling.
Found in almost all tissues, especially cardiac muscle, smooth muscle, and neurons.
Molecular Structure of Gap Junctions:
Connexins:
Family of ~21 proteins in vertebrates.
Each connexin:
4 transmembrane domains.
Cytoplasmic N- and C-termini.
Assemble as hexamers called connexons (or hemichannels).
Connexon Assembly:
1 connexon = 6 connexin subunits.
Two connexons (one from each cell) align to form a complete gap junction channel.
Channels can be:
Homomeric: identical connexins.
Heteromeric: mixed connexins.
Homotypic: same connexons from each cell.
Heterotypic: different connexons on each side.
Combinatorial diversity confers specificity and selectivity for ions or metabolites.
Dynamic Regulation of Gap Junctions:
Junctions are not static:
Connexins turn over rapidly (half-life ≈ 1–5 hours).
New connexons inserted into membrane; old ones internalised and degraded.
Channels exhibit open and closed conformations, influenced by:
Intracellular Ca²⁺ concentration.
pH changes.
Membrane potential.
Post-translational modifications (phosphorylation, nitrosylation).
Different connexin compositions → variable permeability and conductance.
Experimental Demonstration of Dynamic Exchange:
Fluorescent tagging shows rapid replacement of connexins:
Green-labelled connexins replaced by red-labelled ones within hours.
Demonstrates continuous turnover and remodelling.
Indicates that cells can rapidly alter the composition — and thus properties — of their gap junctions.
Functional Roles of Gap Junctions:
Electrical and Metabolic Coupling:
Enable spread of:
Ions (Na⁺, K⁺, Ca²⁺)
Second messengers (cAMP, IP₃)
Metabolites (ATP, glucose derivatives)
Synchronise activity in tissues:
Cardiac muscle: ensure coordinated contraction.
Smooth muscle: mediate peristalsis and vascular tone.
Neurons: facilitate electrical synapses.
Electrical Synapses vs Chemical Synapses:

Functional Advantages:
Electrical synapses:
Synchronise neuronal firing and maintain homeostatic ionic balance across networks.
Allow for current shunting – distributing electrical potential to regulate excitability.
Chemical synapses:
Permit signal amplification and complex integration.
Both coexist to provide flexibility and control in nervous system communication.
Broader Biological and Clinical Importance:
Gap junctions coordinate cell populations (e.g., heart rhythm, embryonic development).
Mutations in connexins cause disorders such as:
Charcot–Marie–Tooth disease (Cx32).
Deafness (Cx26 mutations).
Cardiac arrhythmias (Cx43 dysfunction).
Regulation of gap junctions crucial for:
Wound healing.
Synchronised hormone release.
Tissue homeostasis.
Extracellular Matrix (ECM) Structure and Cell–Matrix Interactions:
The ECM is a dynamic, multifunctional, and tissue-specific microenvironment composed of macromolecules secreted by resident cells.
It provides both:
Biomechanical support (tensile strength, elasticity, and compressive resistance).
Biochemical regulation (signal presentation, adhesion cues, migration pathways).
ECM composition varies greatly across tissues, producing a highly context-dependent landscape rather than a uniform “blanket” around cells.
Major ECM Components:
Glycosaminoglycans (GAGs):
Long, unbranched, and highly sulphated polysaccharides built from repeating disaccharide units.
Their dense negative charge attracts cations (e.g., Na⁺), generating a large hydration shell that produces powerful osmotic swelling pressure.
Produce gel-like matrices capable of resisting compression — essential in structures such as articular cartilage and the intervertebral discs.
Adopt extended conformations due to steric hindrance and charge repulsion, preventing globular folding and increasing their effective volume.
Proteoglycans:
Formed when GAG chains are covalently linked to a core protein, typically via a tetrasaccharide linker added in the ER–Golgi.
Exhibit enormous structural diversity because:
Multiple GAG types can attach to one core protein.
Core proteins vary between tissues.
Aggrecan is a key example:
Contains >100 GAG attachment sites.
Forms enormous aggregates via interactions with hyaluronan, reaching sizes comparable to small bacteria.
Critical in cartilage for absorbing and redistributing compressive load
— — — — —
Collagens:
The principal tensile components of the ECM.
Built from three α-chains wound into a triple helix, forming fibrils and fibres.
Humans express ~42 α-chain genes, enabling tissue-specific mechanical properties through different combinations (e.g., collagen I in tendon, collagen II in cartilage).
Collagen fibres are hierarchically organised into bundles that resist stretch in multiple directions.
Secreted largely by fibroblasts and remodelled continuously during tissue homeostasis.
— — — — —
Elastin:
A highly hydrophobic protein forming elastic fibres that enable recoil in tissues such as arteries, lungs, and skin.
Crosslinked through desmosine and isodesmosine, allowing dramatic reversible stretching.
Essential in tissues requiring cyclical deformation (e.g., blood pressure wave propagation).
— — — — —
ECM Glycoproteins:
Provide specificity, organisation, and adhesion cues.
Act as “molecular signposts” that guide migrating cells or stabilise cellular architecture.
Key examples include:
Fibronectin – dimeric, highly modular, with ~50 exons allowing extensive alternative splicing; central to cell adhesion, migration, and wound healing.
Tenascin, thrombospondin, CYR61 – regulate adhesion, growth, and mechanotransduction.
Their modular domains generate precise binding patterns, giving tissues the ability to fine-tune cell behaviour spatially and temporally.
Functional Properties of the ECM
Biomechanical Roles:
Resistance to Compressive Forces:
Achieved by GAG-rich proteoglycan networks, especially in cartilage.
Example of mechanical demand:
Humans take ~7000 steps/day → ~200 million over a lifetime.
Each step generates ~1.5× body weight on the knee joint.
The meniscus and articular cartilage must dissipate these forces continuously without failing — a level of performance still unmatched by synthetic biomaterials.
Resistance to Tensile Forces:
Achieved through collagen fibres, particularly in skin, tendons, ligaments, and fascia.
Fibres are arranged in rope-like hierarchical bundles that tolerate stretch with minimal deformation.
— — — — —
ECM Remodelling and Regulation:
Matrix metalloproteinases (MMPs) degrade ECM components, enabling dynamic remodelling.
Their activity is tightly controlled by tissue inhibitors of metalloproteinases (TIMPs).
This spatial regulation enables:
Branching morphogenesis (e.g., blood vessel sprouting).
Selective tissue expansion during development.
Wound healing, fibrosis, and regeneration.
Local ECM degradation can create permissive pathways for cell migration while stiff regions provide directional constraint.
— — — — —
ECM as a Guidance and Signalling Architecture:
Migration Barriers and Tracks:
Variations in ECM stiffness and composition create topographical cues that guide migrating cells.
Cells follow adhesive tracks defined by local enrichment of ECM glycoproteins.
Signal Reservoirs:
Due to their charge and binding specificity, many ECM components act as ligand traps, concentrating growth factors, chemokines, and morphogens.
This enables high local ligand concentration, allowing even low-affinity receptors to be activated effectively.
Essential in tissues with tight spatial regulation (e.g., embryonic patterning, wound healing).
Cell–Matrix Junctions:
General Features:
Cells interpret ECM signals using transmembrane receptors that bind ECM components externally and link to the cytoskeleton internally.
Unlike cell–cell junctions, these are heterophilic interactions (cell receptor ↔ ECM molecule).
Allows cells to:
Sense mechanical stiffness (mechanotransduction).
Anchor themselves for traction.
Initiate signalling pathways.
— — — — —
Integrins:
Structure and Composition
Integrins are α/β heterodimers with no intrinsic enzymatic activity.
Functions by linking the ECM to the intracellular cytoskeleton, acting as mechanical and biochemical integrators.
Activation States
Exist in inactive (bent) and active (extended) conformations.
Activation is regulated by inside–out signalling, enabling the cell to decide when it should become adhesive.
Example:
Thrombin activates a GPCR → activates Rap1 → recruits talin → talin binds β-integrin tail → integrin switches to high-affinity state and binds fibronectin.
Ligand Specificity
24 integrin heterodimers exist in humans, allowing recognition of many ECM proteins:
Fibronectin
Laminin
Collagen
Fibrinogen
Requires divalent cations (Ca²⁺, Mg²⁺) for proper ligand binding.
— — — — —
Focal Adhesions:
Activated integrins cluster to form focal adhesions, large multiprotein complexes containing:
Talin, kindlin, vinculin (linking to actin)
Focal adhesion kinase (FAK)
Integrin-linked kinase (ILK)
Functions:
Mechanical coupling to the cytoskeleton.
Signalling hubs that regulate survival, proliferation, polarity, and migration.
Provide cells with a means to convert mechanical inputs into biochemical outputs (mechanotransduction).
ECM and Cell Behaviour: Developmental and Disease Contexts:
Axon Guidance in the Nervous System:
Growing axons extend growth cones enriched in actin-rich filopodia.
Growth cones express high levels of integrins during development.
Filopodia “sample” the ECM; integrin–fibronectin binding generates traction forces as actin polymerisation pushes the membrane forward.
Local ECM patterns therefore create axon trajectories, establishing neural wiring.
Cancer and ECM Dysregulation:
Tumour progression involves profound changes to the ECM:
Fibroblast activation (“cancer-associated fibroblasts”).
Excessive collagen deposition (fibrosis).
Aberrant MMP activity, enabling invasion.
Loss of epithelial polarity plus ECM remodelling enables malignant cells to proliferate, migrate, and recruit vasculature (angiogenesis).
ECM composition and stiffness are now recognised as major determinants of tumour behaviour.