AP chem unit 4: chemical reactions
4.1: introduction for reactions
physical change: a physical property of a substance changes without changing the composition
ex) freezing/melting, seperations of mixures, making solutions, etc.
atoms are not rearranged, physical properties(shape, color, density, mass) are
chemical change: a property of a substance changes(breaking of bonds and formation of new ones)
what to look for:t
precipitate forming
color change
light
temp. change
sound
gas production
new substance formed
difficult to reverse
composition(synthesis) | decomposition | single ionic replacement | double ionic replacement | combustion |
A + B → AB | AB → A + B | A + BC → AC + B | AB + CD → AD + CB | CxHy + O2 → CO2 + H2O |
Na + Cl2 → NaCl | KClO3 → KCl + O2 | Mg + HCl → MgCl2 + H2 | LiCl + AgNO3 → LiNO3 + AgCl | C3H8 + O2 → CO2 + H20 |
double ionic replacement includes acid/base neutralization and precipitation reactions
can be considered RedOx reactions(except for double replacement)
4.2: net ionic equations
physical and chemical reactions can be represented by balanced equations
must be balanced!!!
diatomic elements should always be in diatomic form(H2, N2, O2, F2, Cl2, Br2, I2)
overall ionic equations show how ionic compounds dissociate into their ions when dissolved in water
only changes substances that are ionic and aqueous
net ionic equations shows the particles that change in the reaction
cancels out spectator ions
4.3: representation of reactions
chemists use diagrams as a way to represent chemical reactions
solid particles should be close together, liquid particles showing a bit of disorder, and gas particles spread out far
aqueous solutions sometimes only shows the solvent particles
ions should be dissociated & include charge
solvent particles should orient dipoles in the correct way
4.4: physical and chemical changes
a physical process occurs when a substance undergoes a change in properties but not a change in composition(phase change, mixture separation)
ex) H2O(s) → H2O(l)
when the water melts the chemical properties do not change, but the physical properties do
ex) mixture separation(chromatography, distillation, filtration)
a chemical process occurs when a substance is transformed into a substance with a different composition
ex) Na + Cl → NaCl
ex) synthesis, decomposition
4.5: stoichiometry
stoichiometry: conversions that involve mole ratios from a balanced chemical equation
information about >1 reactants signals that you will have a limiting reagent(the reactant that runs out first)
theoretical yield: predicted/calculated mass
actual yield: experiment determined mass
percent yield = (actual yield/theoretical yield)*100
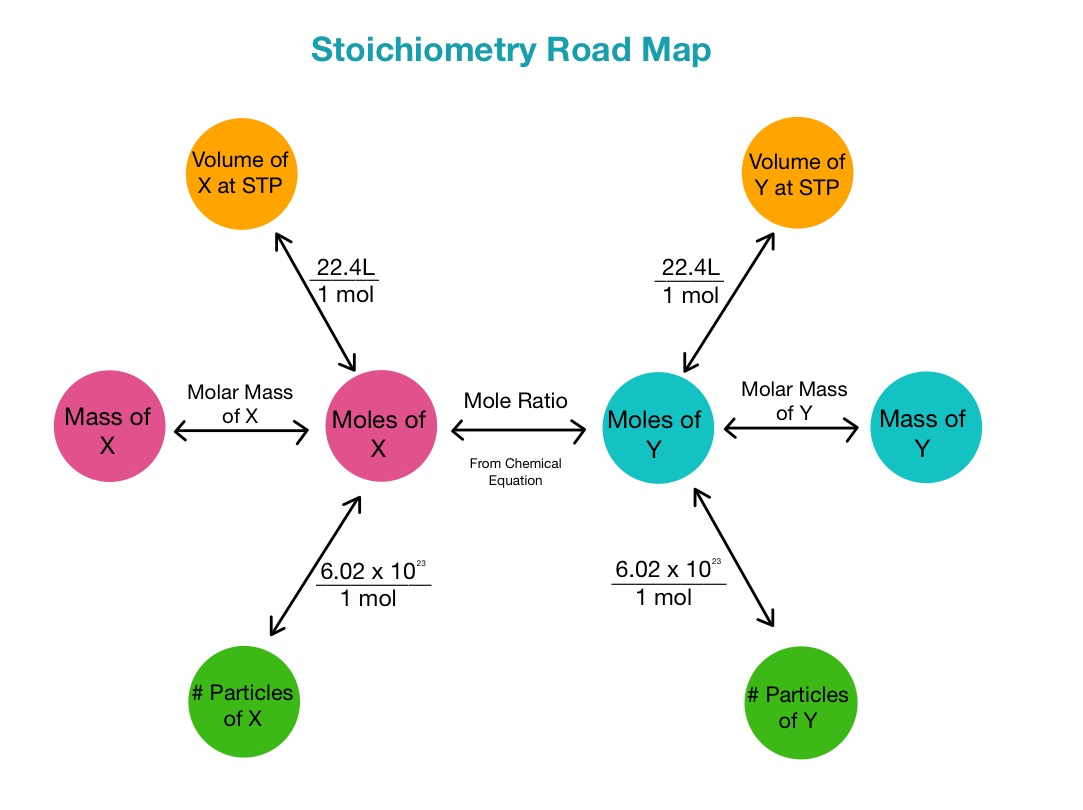
4.6: introduction to reactions
titration: lab technique where a volume and molarity of a titrant is dispensed from a buret to a flask containing a measured quantity of the analyte
moles of titrant can be determined at the equivalence point(when the moles of titrant and analyte are the stoichiometrically the same)
usually indicated by color change
types of common titrations:
acid base titrations: either acid or base can be the titrant
acid base indicator used for end point
redox reactions: color change when oxidation state of a metal ion changes
precipitation titrations: precipitate formed or a color change
when titrant reacts with ions in the analyte
4.7: types of chemical reactions
acid base reactions involve transfer of 1 or more protons between substances
acids are proton donors, bases are proton acceptors
protons are hydrogen ions
hydronium(H3O+) ions form when a hydrogen reacts with water
conjugate acid: a base that has gained one proton
conjugate base: an acid that has lost one proton
acids and bases are described as strong or weak
strong means that the substance will completely separate into its ions
strong acids: H2So4, HI, HBr, HNO3, HCl, HClO3, HClO4(So I Brought No Clean Clothes)
strong bases: LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
oxidation reduction reactions involve transfer of one or more electrons between substances
oxidation: loss of electrons
reduction: gain of electrons
oxidation numbers!!!
hydrogen is always +1
except for hydride, -1
oxygen is always -2
except for peroxide, -1
4.8: introduction to acid base reactions
an acid base reaction is a reaction where a proton(H+) is transferred from an acid to a base
an acid becomes a conjugate base when it transfers a proton to a base
make a conjugate acid base pair(that always differs by one proton)
water can be either a base or an acid
ex) HA + H2O → H3O+ + A-
acid base CA CB
a strong acid/base will ionize completely and is represented by a single arrow(→), a weaker acid/base will not ionize completely is represented by a double arrow(<->)
a stronger acid will form a weaker conjugate base, and vise versa
4.9: oxidation reduction(redox) reactions
redox is the transfer of electrons from one element to another
oxidation reaction cannot occur without reduction
all chemical reactions(except double replacement(precipitation reactions) and neutralization reactions) are redox