VSEPR Theory
Valence Shell Electron Pair Repulsion
if there are electron pairs in the valence shell of an electron, they will repel.
the pair of electrons will take up positions as far away from each other as possible causing a bend in the shape of the molecule
5 possible shapes:
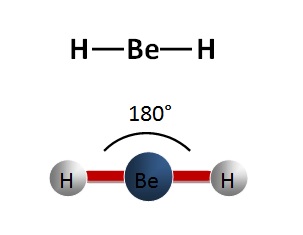
linear
no repulsion, 2 bonding pairs, no lone pairs
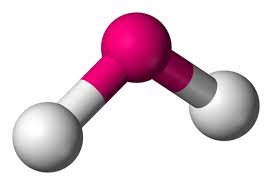
bent
repulsion, 2 bonding, pairs, 2 lone pairs
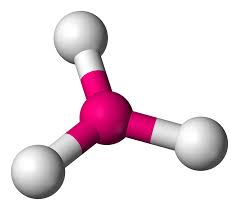
trigonal planar
no repulsion, 3 bonding pairs, no lone pairs
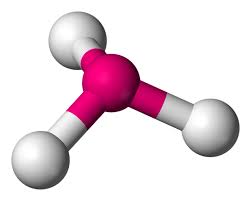
pyramidal
repulsion, 3 bonding pairs, one pairs
tetrahedral
no repulsion, four bonding pairs, no lone pairs
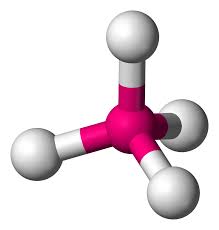
polarity using vsepr theory:
if there is repulsion, it’s polar
when there is an equal sharing of e-, it is non-polar
when the sharing is unequal, there is repulsion, therefore it is always polar when there is repulsion