AP chem unit 3 part 2: Properties of Substances and Mixtures
3.7: Solutions and mixtures
solutions can be in solid, liquid, or gas form
Molarity= moles solute/liters total solution
properties of liquid solutions:
the components cannot be separated by using filtering
there are no components large enough to scatter visible light(tyndall effect)
the components can be separated using processes that are a result of the intermolecular attractions between and among the components(distillation, chromatography…)
ionic compounds dissociate into ions in solutions
concentration of solution may not be the same as original
ex) nacl will dissolve into 1 mole na+ and 1 mole cl-, but cacl2 will dissolve into one ca+2 ion and 2 cl- ions
3.8: Representations of solutions
IMFs exist between solute particles, solvent particles, and between solute and solvent particles
may promote or prevent the formation of a solution
the tendency to mix is usually strong, but if the solvent-solvent and solute-solute attractions are strong compared to the solvent-solute interactions, a solution will not form
miscibility: the ability of two substances to mix without separating
similar materials are miscible(like dissolves like)
polar solvents dissolve polar solutes, nonpolar dissolves nonpolar
a solutions will form if the solvent-solute interactions is greater or relatively equal to the solvent-solvent and solute-solute interactions
particle drawings
concentration can be represented by the # of particles
diff in concentration showed by the ratio of particles

3.9: separation of solutions and mixtures chromatography
chromatography provides a method of separating a mixture of solutions based upon polarity differences
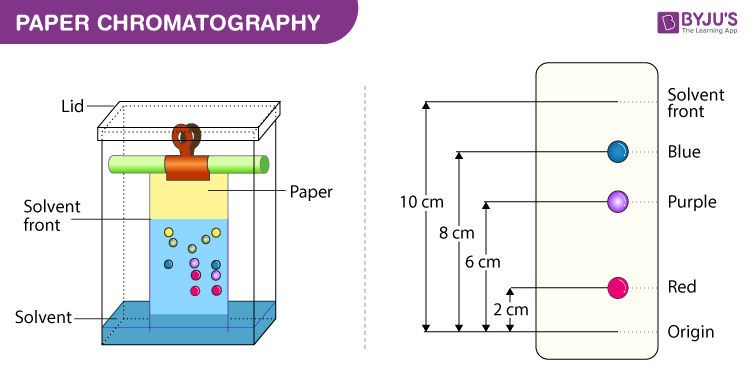
paper chromatography
thin layer chromatography
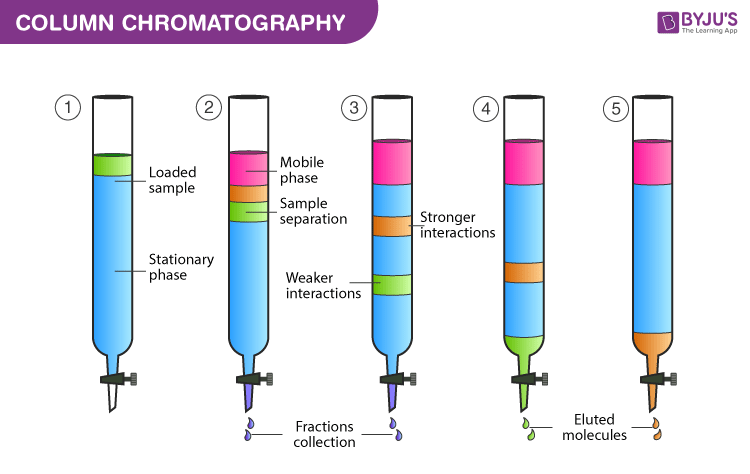
column chromatography
Rf(retardation factor): the ratio of the solute’s distance traveled to the solvent’s distance traveled
paper chromatography(most common)
a line is drawn near the end of a strip of paper
a drop of the sample to be separated is place on the line
the paper is place in a sealed container with a shallow layer of solvetn
solvent and dyes will separate over time
remove the paper before the solvent reaches the top of the paper
measure the height of the leading edge of the solvent and measure the leading edge of each dye
the same compound will move at the same rate relative to the same solvent
different compounds will have an at least slightly different polarity to other compounds
so the Rf traveled will differ between compounds
the more similar in polarity the sample is to the solvent the further it will travel
identification of a sample is based on the Rf value
NOT COMPARING DISTANCES
the stationary phase is the paper and mobile phase is the solvent
thin layer chromatography
very similar to paper chromatography
separation occurs on a thin layer on a sheet of plastic
can be used to separate invisible samples
nonpolar travels further
column chromatography
separates a mixture in solution based on their polarity
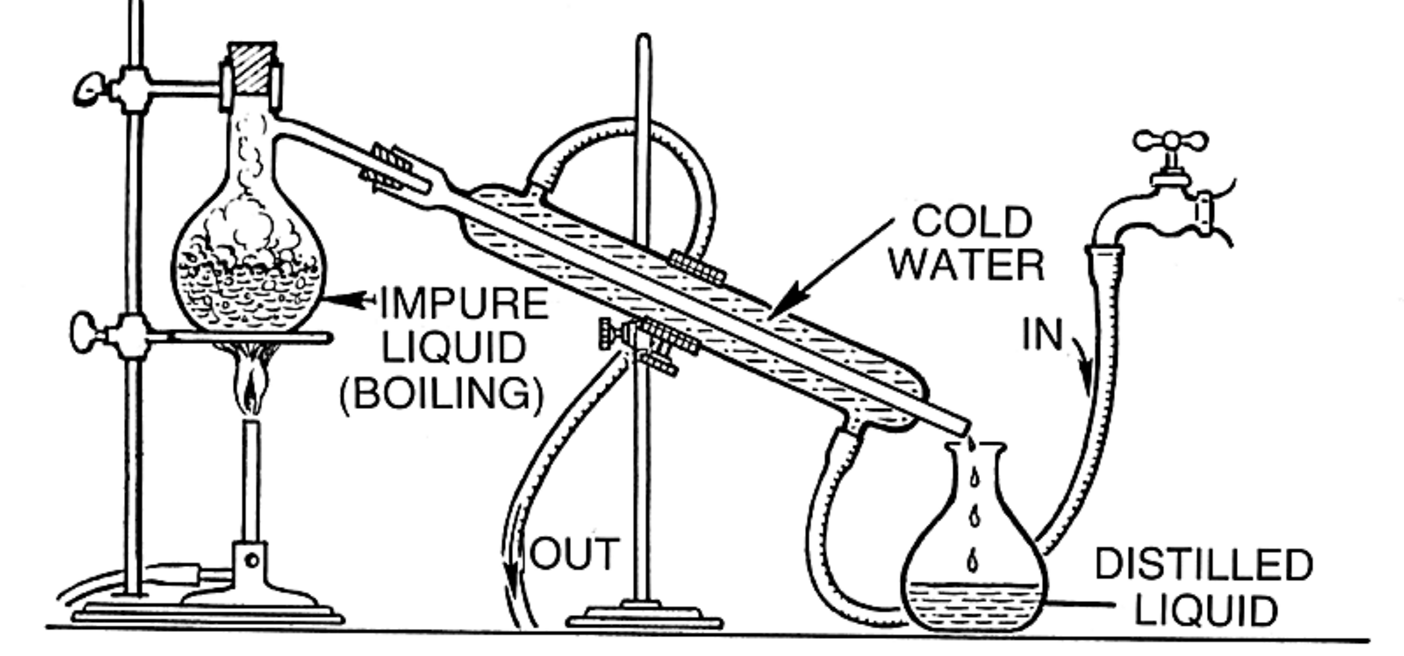
distillation
physically separates layers based on boiling point and imfs
3.10: solubility
solubility: the extent to which a solute will dissolve into a solvent to form a solution
If a lot of the solute can dissolve it is highly soluble, if little dissolves it is slightly soluble, if none dissolves its insoluble
a solution is aqueous when it has water
water is a common solvent because it can dissolve many different substances
good solvent because it is highly polar
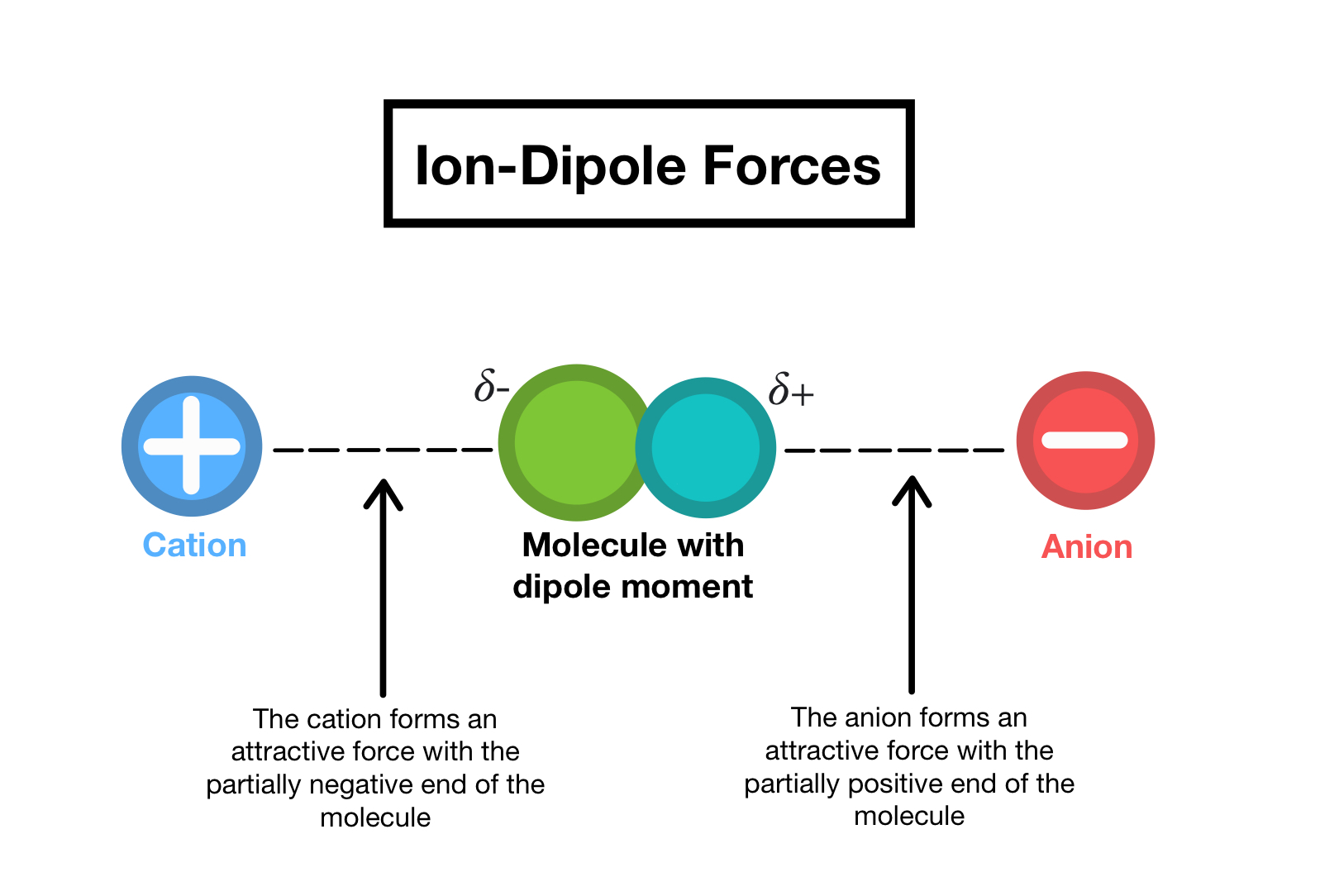
hydration: the process where the water molecules surround the ions to dissolve them
when water molecules surround the ions they orient the positive ends of the water molecule are close to the negative ions and negative ends are toward positive
referred to as a ion-dipole
“like dissolves like”
substances with similar IMFs will dissolve
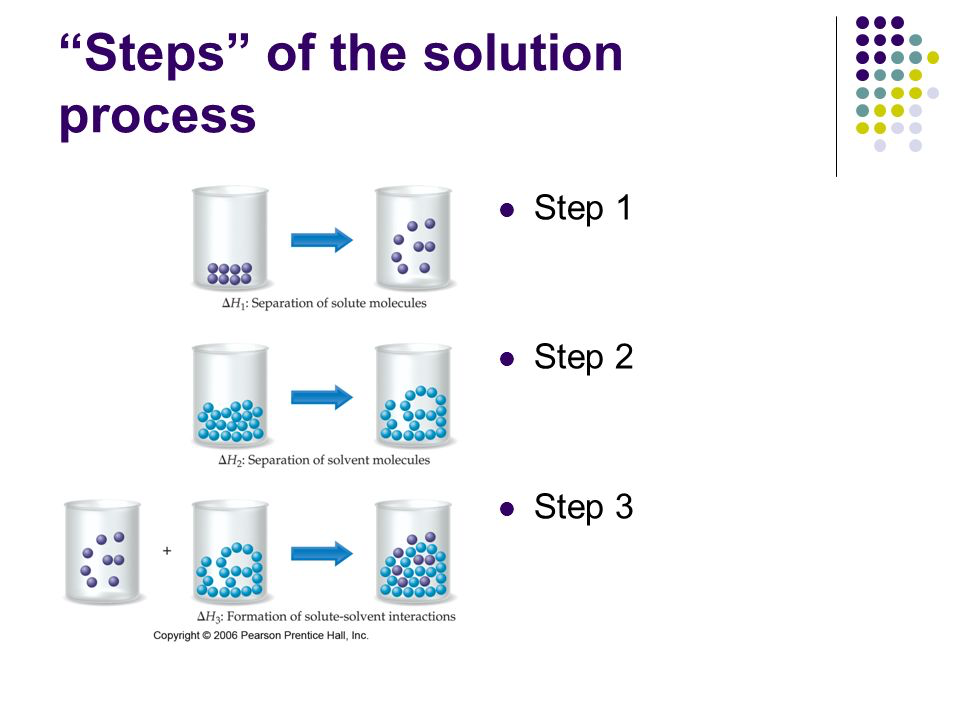
steps for a solution to form
the solute particles must separate(energy required)
the solvent particles must separate(energy required)
the solute and solvent particles must come back together(energy released)
polar and non-polar substances don’t dissolve in each other because there is not enough energy released in the last step for the first 2 steps
amount of energy needed for each step depends on the IMFs of the materials
the stronger the IMFs, the more energy needed
miscible: can mix together
immiscible: cannot mix together
3.11: spectroscopy and the electromagnetic spectrum
spectroscopy: the study of how matter interacts with electromagnetic radiation
electromagnetic radiation is classified into different types based on the wavelength of the radiation

molecules can moved in different ways
translational
movement from one place to another
rotational
spinning in place
vibrational
moving in place
bending, stretching

microwave rotational spectroscopy
microwave radiation is lower in energy than visible light
when microwave radiation strikes the molecule it causes them to rotate
due to the interaction of the dipole of the molecules interacting with the electromagnetic field of the microwave photons
infrared vibrational spectroscopy
higher in energy than microwave but lower than visible
measures the vibration of atoms and determines them
lighter atoms and stronger bonds tend to vibrate at higher frequencies
ultraviolet visible light spectroscopy
part of electromagnetic spectrum that we can see
the interaction of these wavelengths of electromagnetic spectrum and molecules or atoms can allow the determination of the concentration of the molecules in the solution
the photons of colored light are absorbed by a compound causing electrons to move from their ground state to a higher energy excited state
spectrophotometer
measuring the amount of light emitted from a sample and comparing it to the amount of light that entered we can find the amount of light that was absorbed by the sample
can determine the concentration of a colored substance in a solution
3.12: the photoelectric effect
photoelectric effect: when light shines on the surface of metal, electrons can be ejected from the surface
based on light having wave like and particle like properties

C = Vλ
E = energy(J)
h = planck’s constant(6.626×10^-34Js)
v = frequency(Hz, s^-1)
1×10^-9 m = 1 nm
by measuring the amount of energy needed to remove the electrons we can determine how tightly the electrons are being held in the atom(binding energy)
more energy = electrons that are closest to the nucleus or the nucleus has a higher charge
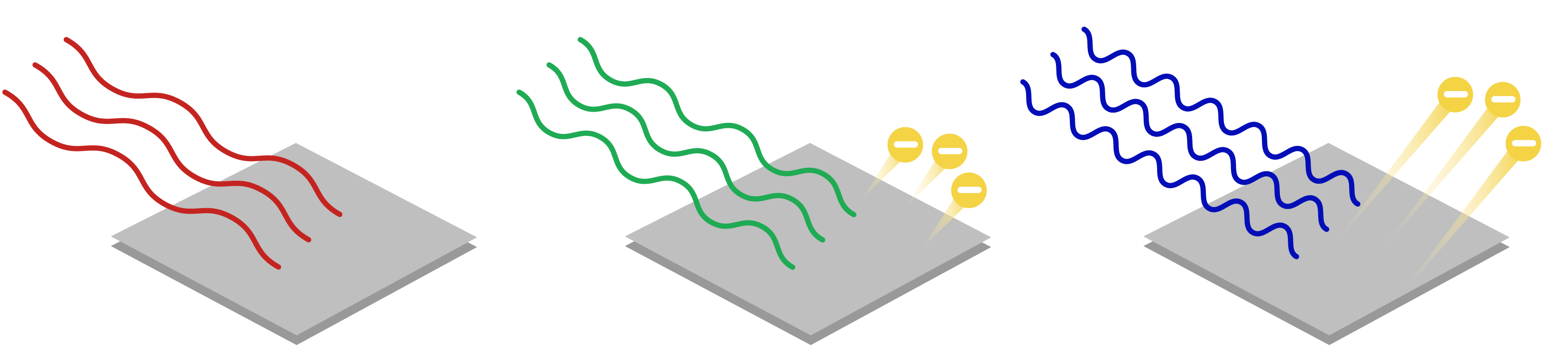
not all light causes electrons to be ejected
red light doesn’t have enough energy to remove electrons
threshold frequency(V0): amount of energy needed to remove an electron
if a frequency higher than the threshold frequency is used the electrons will eject with more kinetic energy
amount of energy needed to eject an electron can be thought as longest wavelength needed in order to eject an electron(threshold wavelength(λmax))
the total energy of the light, the photon(Ephoton) is equal to the energy of the ejected electron,(KEelectron)+binding energy
the energy of the photons must meet the minimum energy
the rate of emission of the photoelectron is known as the photoelectric current, which is proportional to the intensity of the light
if the minimum energy is met to eject electrons, the kinetic energy of the photoelectron is dependent on the energy of the light used to eject the electrons
3.13: beer lambert law
a spectrophotometer or colorimeter can be used to measure the absorbance of a substance
the measure of the light that is stopped by or absorbed by a solution
the more concentrated the solution is, the less light can pass through a solution
solute: the substance that is dissolved
solvent: the substance that is dissolving
different colors of solutions absorb different colors of light
if we are using a red solution, we use a green light to measure it
to determine the best wavelength to use for your sample, place the sample into a spectrophotometer and generate a graph of absorbance vs wavelength
the best choice for a wavelength is one where the absorbance is close to 1
then you create a calibration graph with solution concentration and their absorbance value
graph should be linear
A = EBC
A = absorbance
E = molar absorptivity constant(slope of the graph)
B = path length(usually 1 cm)
c = concentration(M)
you can measure the absorbance for an unknown sample and determine the concentration from the line