Lecture 23-24-Carbonyl Chemistry
Core Concepts
Understand the general mechanism for nucleophilic addition to the carbonyl (aldehyde/ketone) group.
Apply nucleophilic addition to the carbonyl group to the formation of hemiacetals/hemiketals and acetals/ketals.
Explain and show how acid-catalysis accelerates acetal/ketal formation.
Apply the concept of equilibria to show acetal/ketal formation is reversible, allowing the hydrolysis of acetals/ketals.
Use curly arrow notation to predict the product of nucleophilic addition of other nucleophiles.
Reaction Mechanisms
Curly Arrows: Used to represent the movement of electrons in reaction mechanisms.
Predicting Reactivity:
Identify polarized bonds due to differences in electronegativity.
Opposites attract: Electrostatic attraction between partially positive and partially negative atoms.
Identify the nucleophile (electron-rich) and electrophile (electron-deficient).
Draw reagents in full, including all atoms and lone pairs.
Steps for Drawing Mechanisms
Draw molecules clearly.
Add lone pairs and hydrogen atoms at active centers.
Identify bonds made and broken.
Identify the most nucleophilic center.
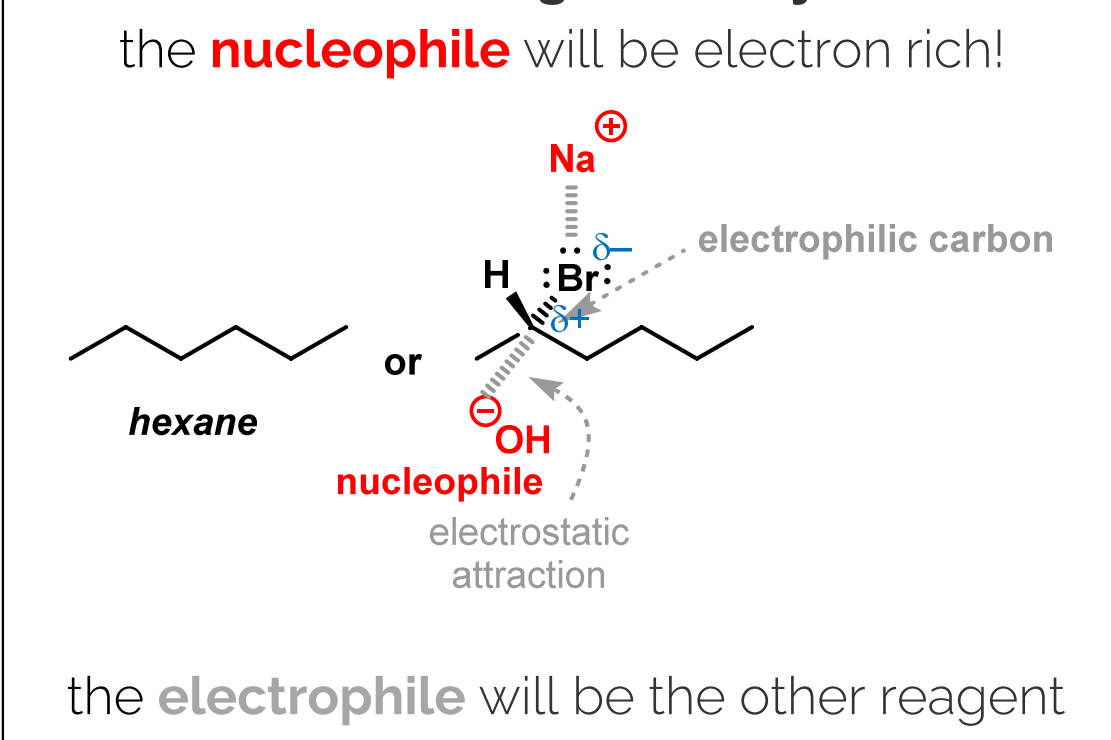
Identify the most electrophilic center.
Draw a curly arrow from the nucleophile to the electrophile, starting with electrons.
If the atom attacked has too many bonds, break a bond using a curly arrow.
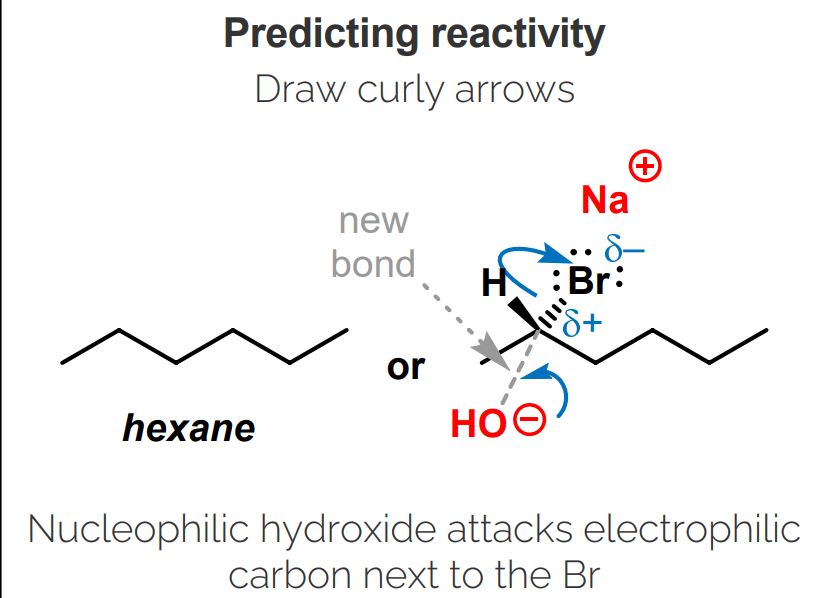
Electrons should flow towards the more electronegative atom.
The polarized bond is broken.
Draw the products.
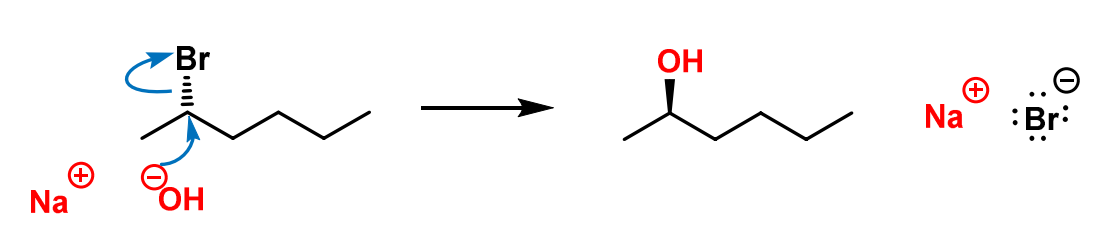
Repeat until a stable product is formed.
Sometimes you form intermediates.
Carbonyl Group (C=O)
Importance: The carbonyl group is prevalent in many biological molecules, such as glucose, peptide bonds, and DNA monomers.
Polarity: The carbonyl group is polarized due to the difference in electronegativity between carbon and oxygen, resulting in a partial positive charge (δ+) on carbon and a partial negative charge (δ-) on oxygen.
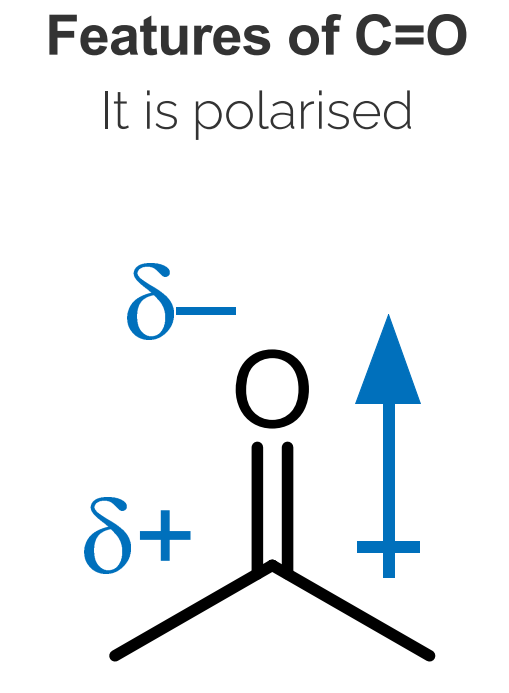
Reactivity:
Reacts with nucleophiles because of the electrophilic carbon.
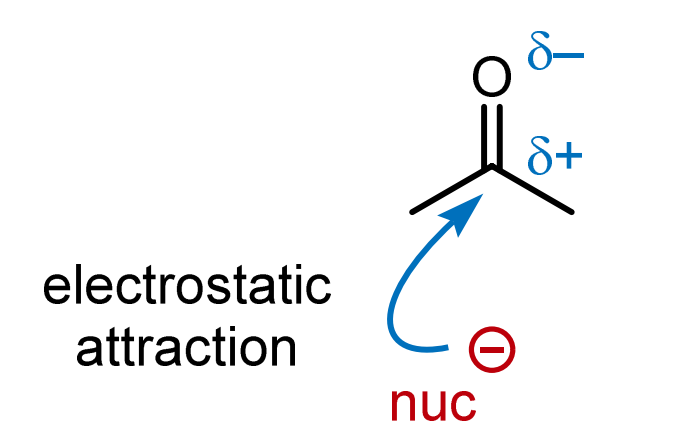
New bond formation exceeds carbon's valence (too many bonds).
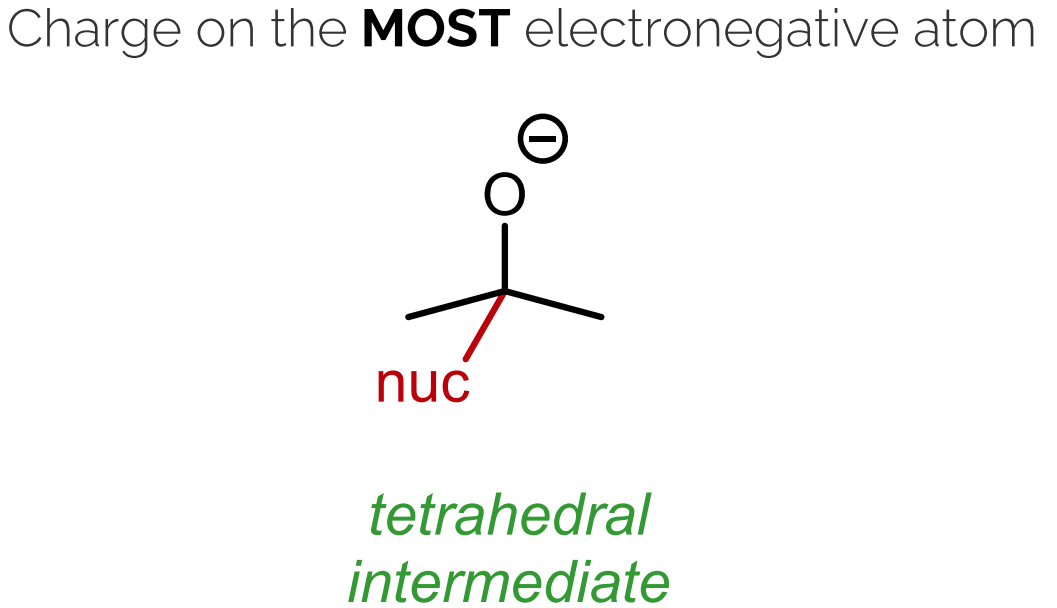
Pi bond broken.
The negative charge ends up on the most electronegative atom (oxygen), forming a tetrahedral intermediate.
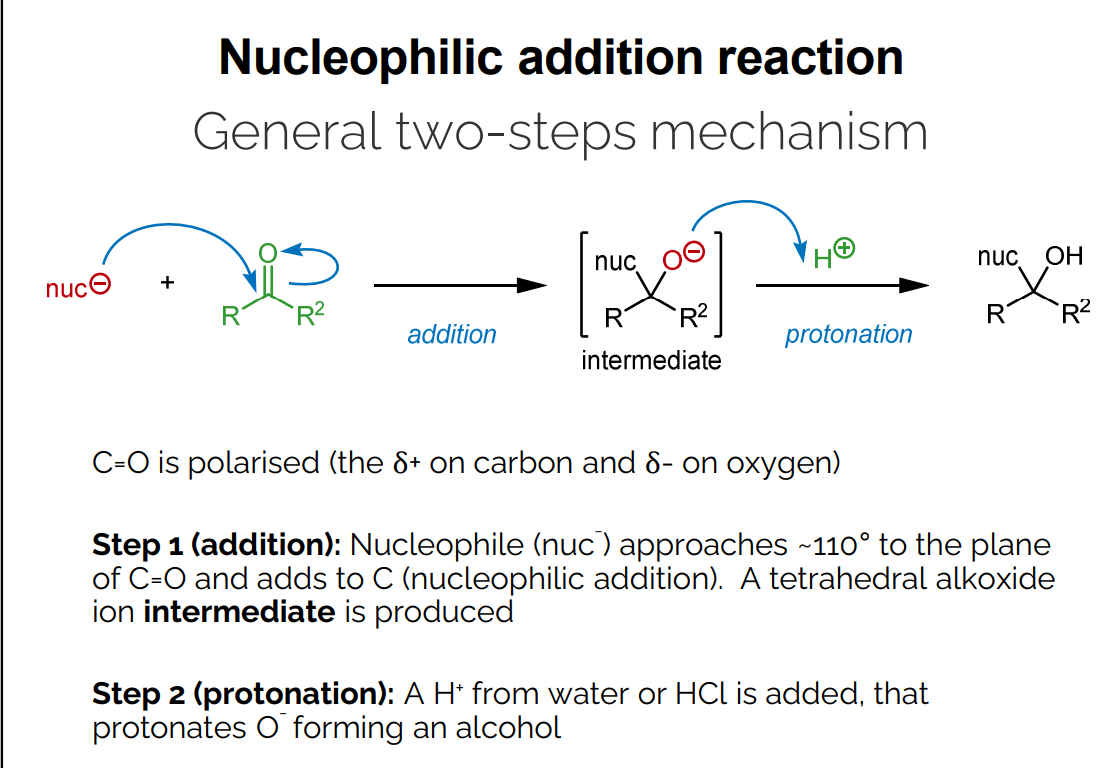
General Two-Step Mechanism: Nucleophilic Addition
Step 1 (Addition):
A nucleophile (nuc-) approaches the carbonyl carbon at approximately 110 degrees.
The nucleophile adds to the carbon, resulting in a tetrahedral alkoxide ion intermediate.
Step 2 (Protonation):
A proton (H+) from water or HCl is added to the oxygen (O-), forming an alcohol.
Aldehydes vs. Ketones
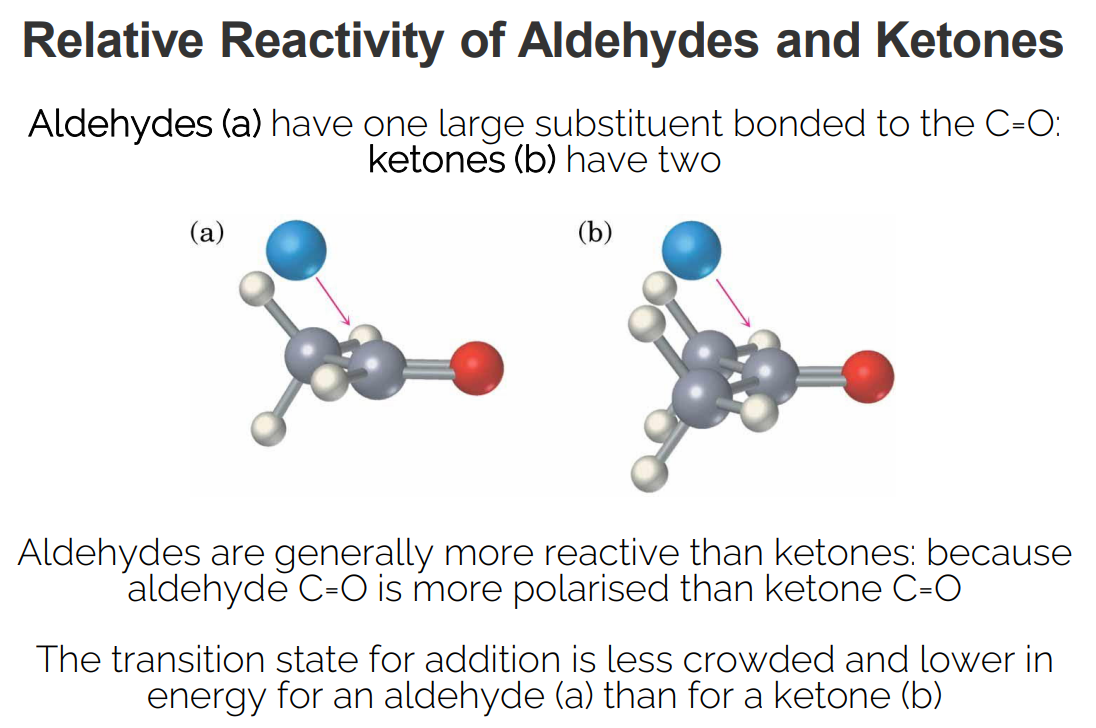
Aldehydes: Have one large substituent bonded to the carbonyl carbon.
Ketones: Have two substituents bonded to the carbonyl carbon.
Reactivity: Aldehydes are generally more reactive than ketones (because they are more polarized).
Polarization: Aldehyde carbonyl groups are more polarized than ketone carbonyl groups.
Steric Hindrance: The transition state for nucleophilic addition is less crowded and lower in energy for aldehydes than for ketones.
Curly Arrows: Revision
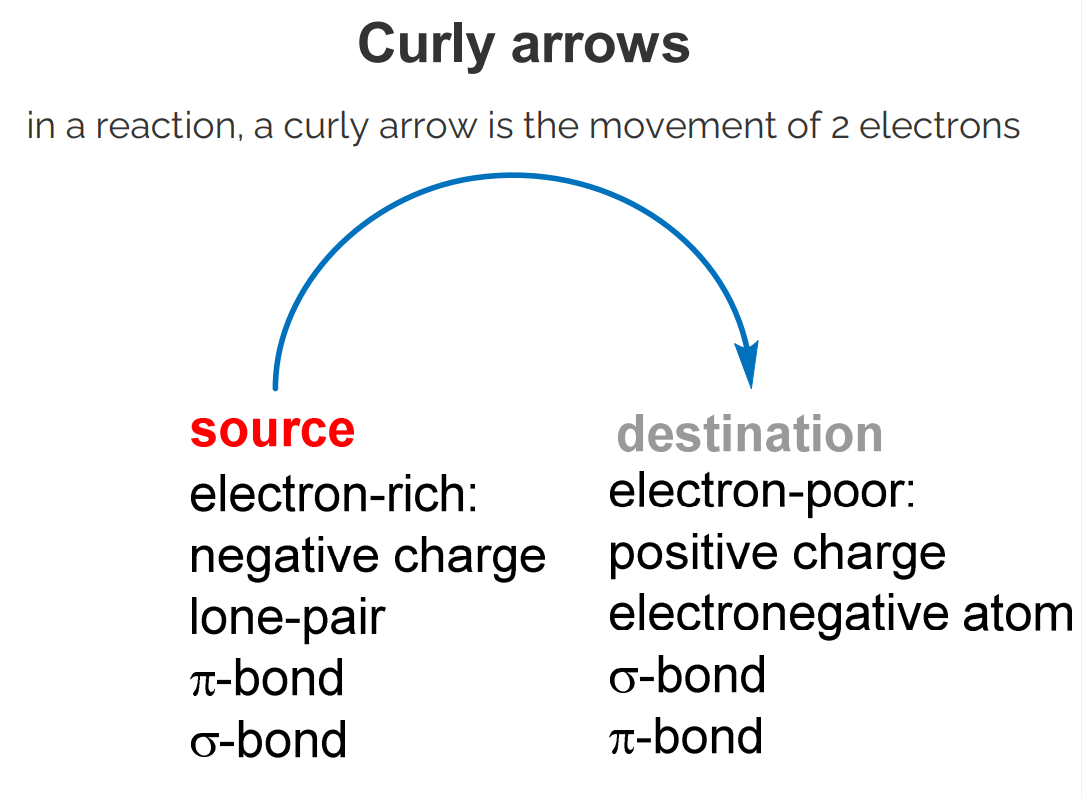
Represent the movement of two electrons.
Electrons flow from electron-rich areas to electron-poor areas.
Three allowable types:
From bond to lone pair.
From lone pair to bond.
From bond to bond.
Do not violate the octet rule.
Never draw curly arrows coming together.
Reaction Mechanisms
A reaction mechanism describes how a reaction occurs.
It details:
Which bonds are broken and which new ones are formed.
The order and relative rates of bond-breaking and bond-forming steps.
The role of the solvent (if in solution).
The role of the catalyst (if there is one).
The position of all atoms and the energy of the system during the reaction.
Developing a Reaction Mechanism
Design experiments to reveal details of the chemical reaction.
Propose a set of steps that might account for the overall transformation.
Establish a mechanism by showing it is consistent with every test devised.
This does not mean that the mechanism is correct, only that it is the best explanation we are able to devise
Why Reaction Mechanisms Are Important
Reaction mechanisms allow chemists to predict the outcomes of reactions (to maximize yield ect.), understand the behavior of different functional groups, and guide the design of new synthetic pathways.
They are the framework within which to organize descriptive chemistry.
They provide an intellectual satisfaction derived from constructing models that accurately reflect the behavior of chemical systems.
They are tools with which to search for new information and new understanding.
I.e. to create a cure for HIV.
Nucleophilic Addition Reactions
Looking at various nucleophiles.
Strong Nucleophiles
Cyanide Ion and Cyanohydrin Formation
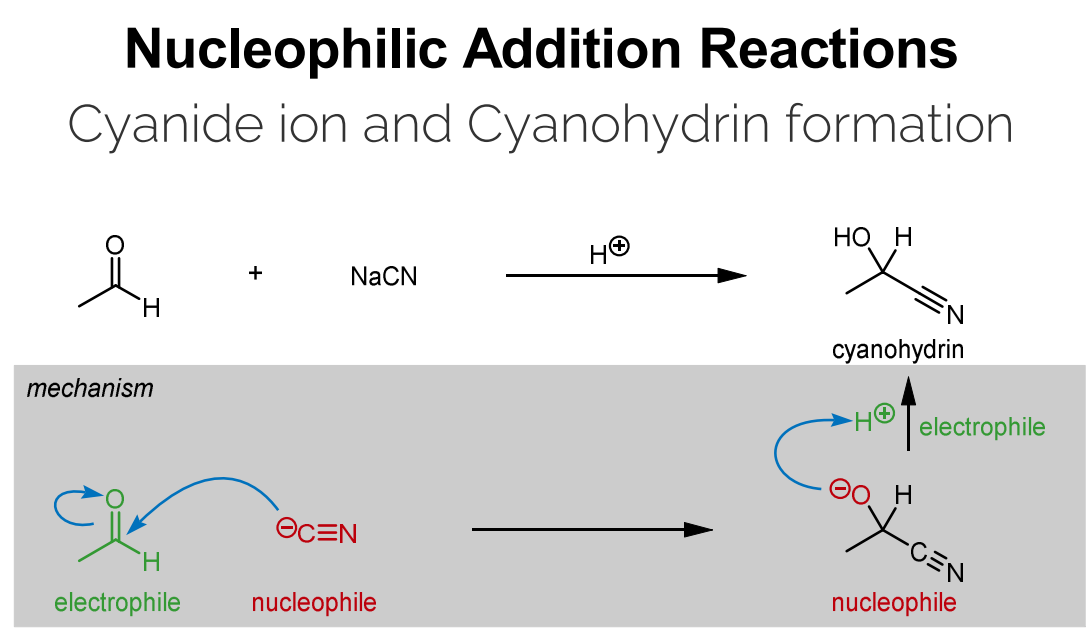
Cyanide ion (CN-) acts as a nucleophile.
The Carbon of the Carbonyl is the electrophile.
Due to the difference in electronegativity between the carbon and the oxygen (polarized bond).
Reacts (nucleophilic attack by cyanide) with aldyhyde/ketone to form Cyanide Ion which is then protonated to form cyanohydrins..
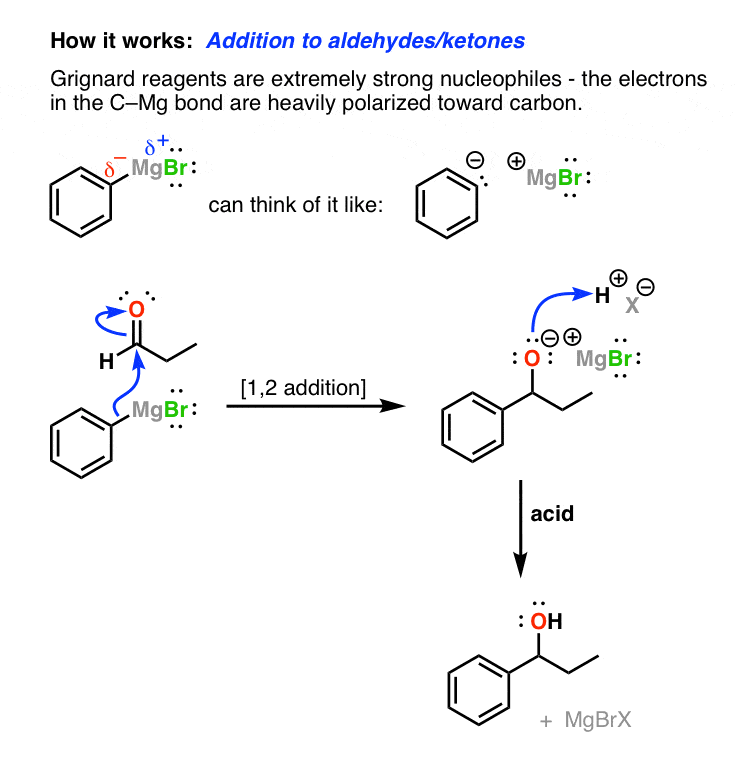
Grignard Reagents
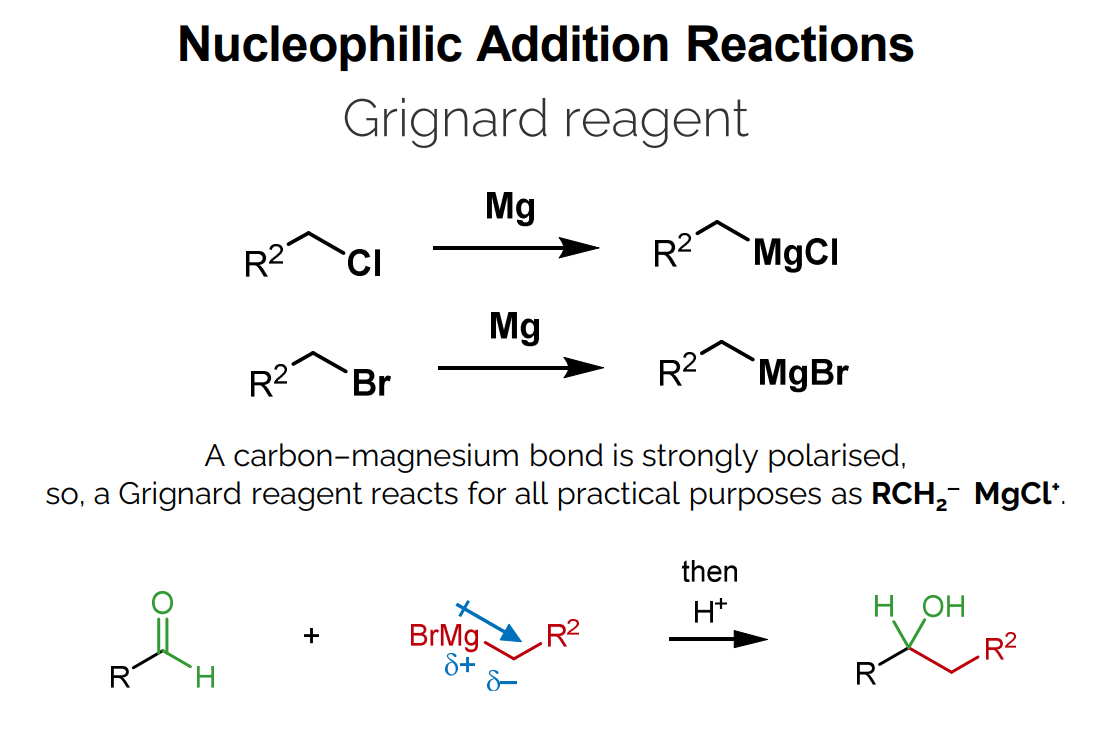
A carbon-magnesium bond is strongly polarized.
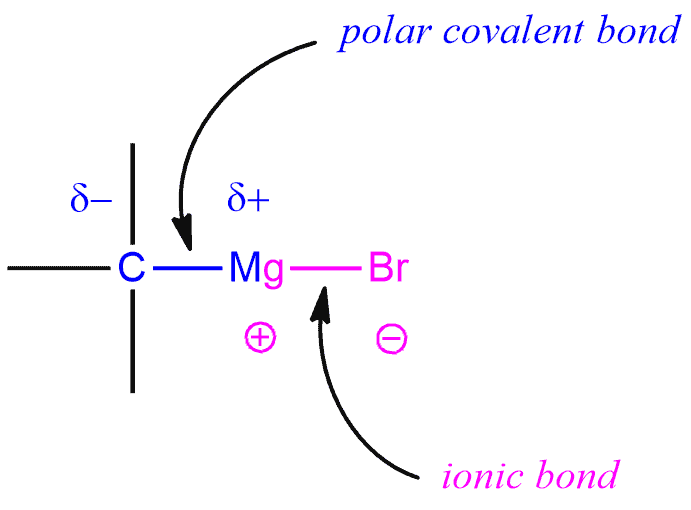
There is negative charge is on the carbon to cancel out the positive charge on the magnesium (Mg is +2 and Cl is -1 so total charge of +1).
RC^{}H_2^{-}-MgCl^{+}
Weak Nucleophiles
Reaction with Alcohols
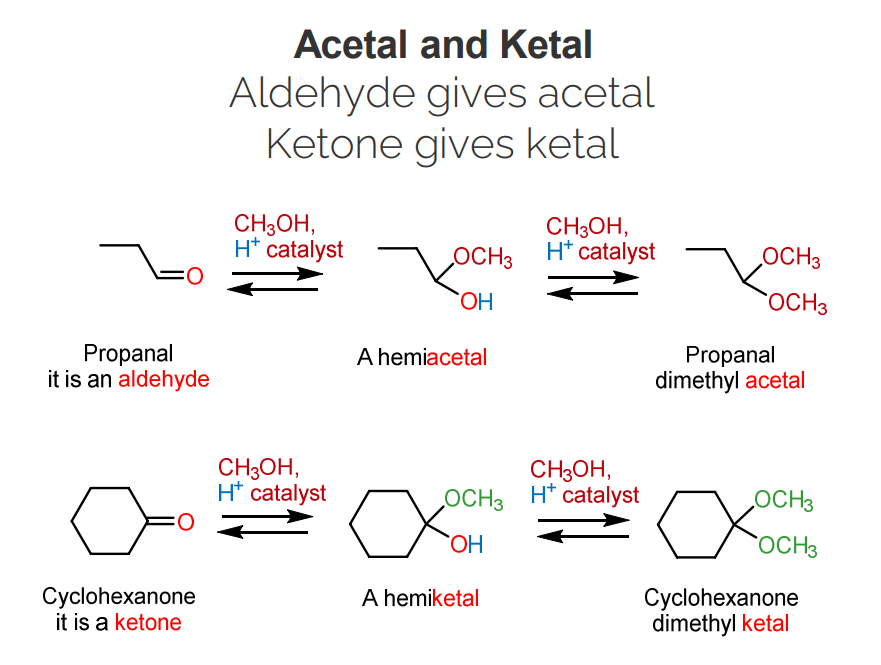
Alcohols react with aldehydes and ketones to form hemiacetals/hemiketals and acetals/ketals.
If you ever see the oxygen-carbon-oxygen, then you are dealing with hemiacetals/hemiketals and acetals/ketals.
Reaction with one alcohol molecule produces hemiacetals/hemiketals.
This with have an O-H.
Reaction with two alcohol molecules (excess alcohol) produces an acetals/ketals, with the removal of water.
This will have a O-R2.
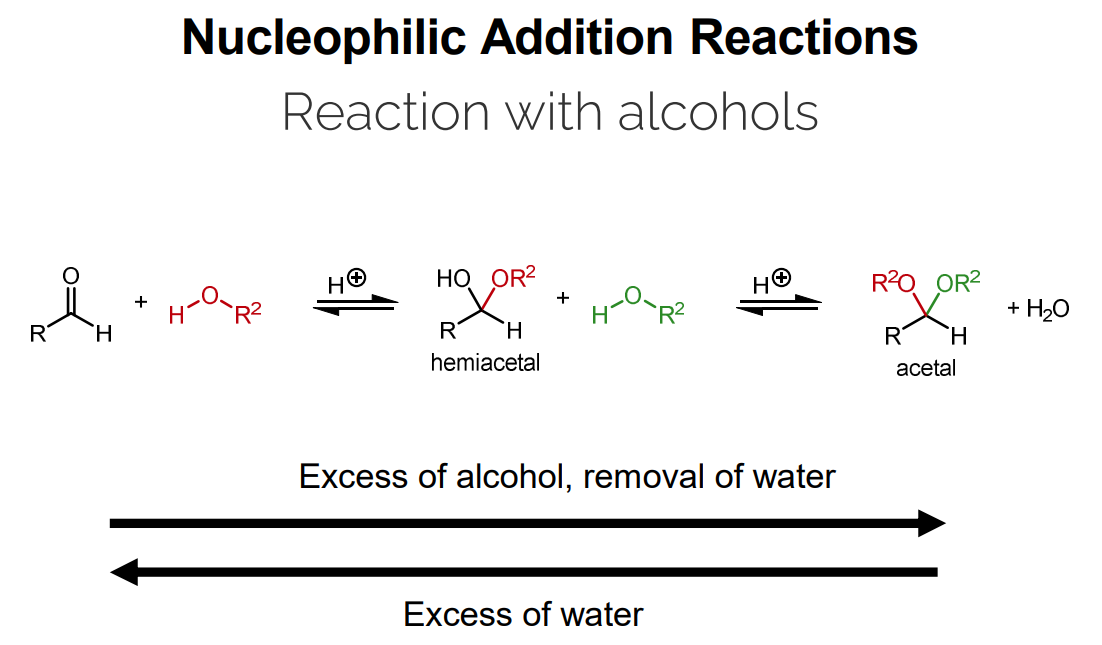
Equilibrium can be shifted by using an excess of alcohol or water.
H^+ + R-OH \rightleftharpoons R^2O-OR^2 + H_2OReaction:
R + RNH2 \rightarrow R2C=NR + H_2O
Mechanism: Addition of Alcohols to C=O
Alcohols and amines are
Step 1: Protonation of the carbonyl oxygen to create a stronger electrophilic center.
Step 2: Addition of alcohol as a nucleophile.
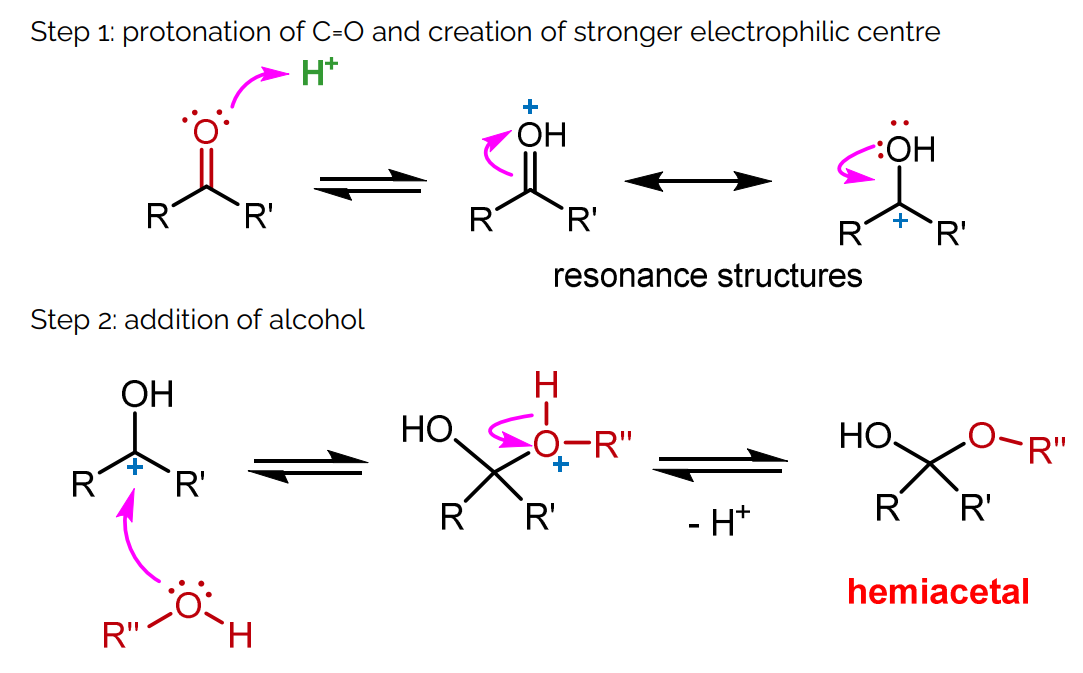
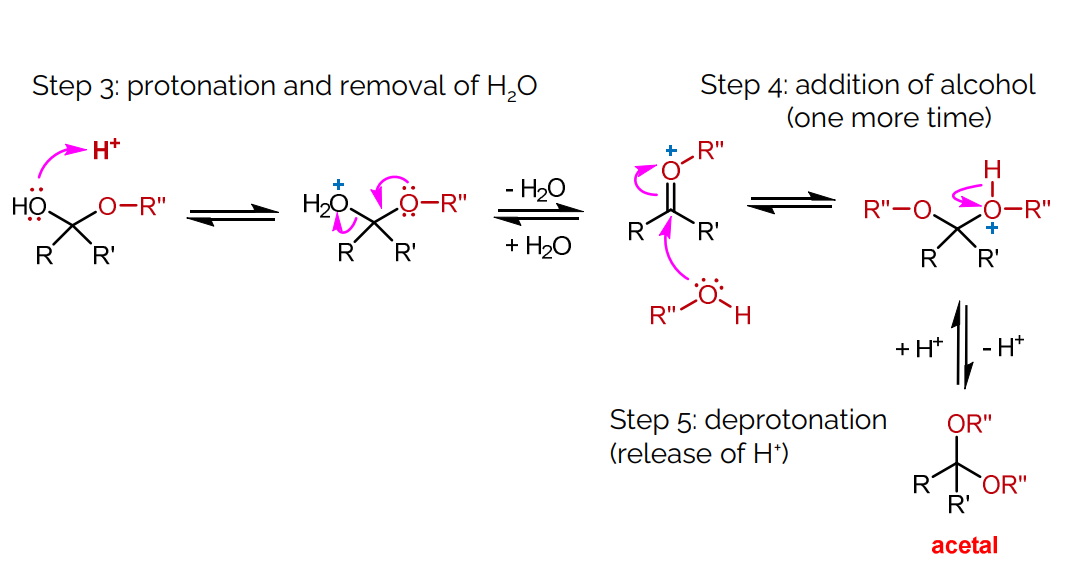
Step 3: Protonation and removal of water.
H+ is a Catalyst (not used up)
Make is a stronger electrophile=> speeding up the reaction.
Step 4: Addition of a second alcohol molecule.
Step 5: Deprotonation to release H+.
H+ is a Catalyst(not used up)
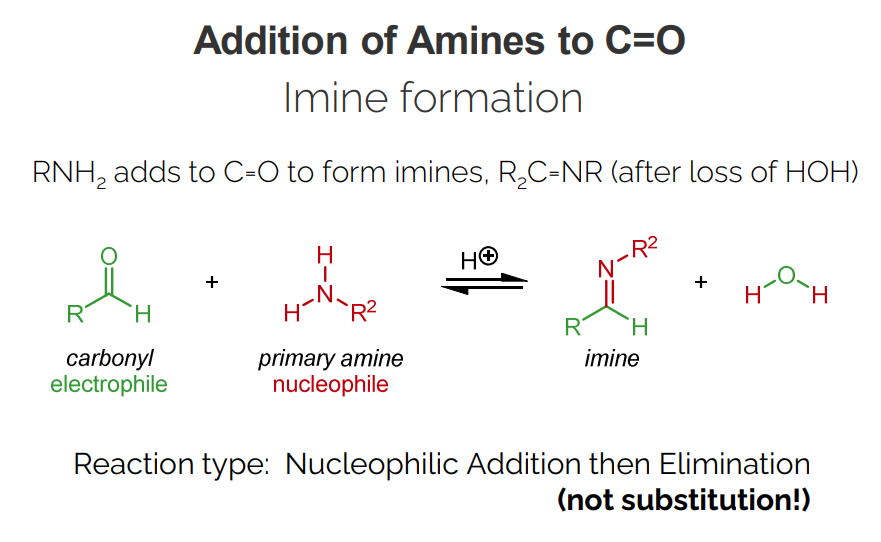
Addition of Amines to C=O
Amines (RNH2) add to carbonyls to form imines (R2C=NR), with the loss of water.
Reaction type: Nucleophilic Addition, then Elimination (not substitution).
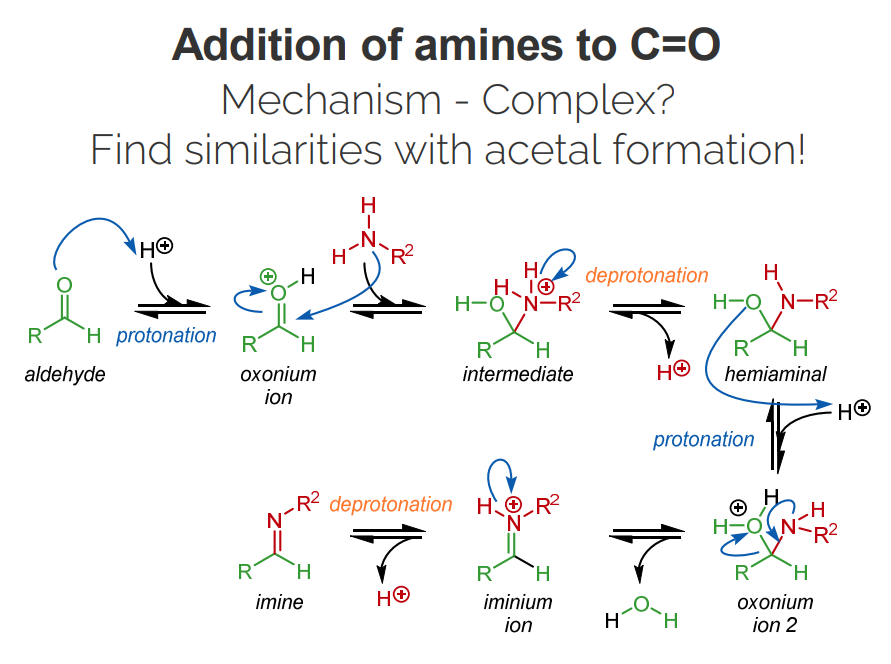
Lots of similarities with acetal formation; initial steps are the same (except obviously replace an O with N).
Imine Derivatives: Brady’s Reagent
2,4-Dinitrophenylhydrazine is used as Brady's reagent to detect the presence of carbonyl groups.
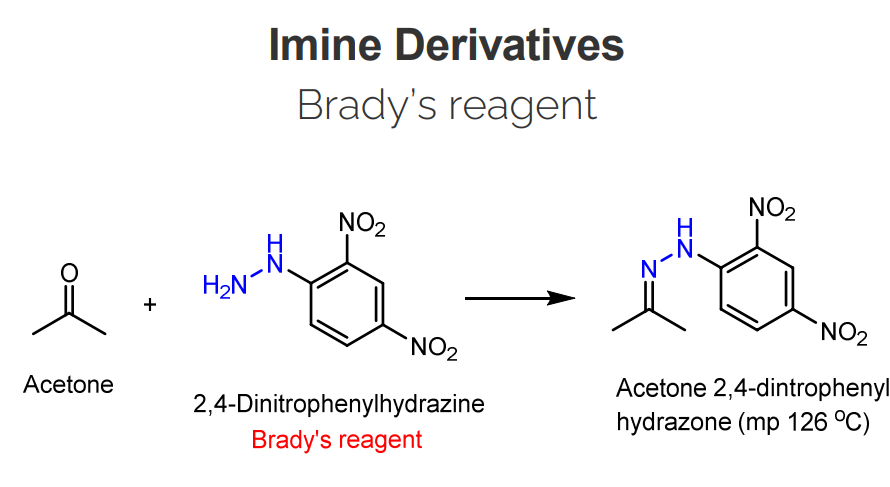
The reaction proceeds via a nucleophilic addition mechanism, where the nucleophilic-NH2 group of Brady's reagent attacks the electrophilic carbonyl carbon.
A color change occurs as the resulting hydrazone product forms with carbonyl compounds; a bright yellow or orange precipitate.