1.1 Atomic Theory
%%atom:%% the smallest particle having properties of an element.
%%element:%% substance that cannot be broken down into simpler substances.
The Greeks (400 B.C.)
- Aristotle: deduced that all matter was made of 4 elements
- @@ex: fire, air, water, and earth@@
Democritus (350 B.C.)
- Greek philosopher
- matter is not infinite
- “atomos”= indivisible
- atoms move in empty space
- no experiments to support the idea
Alchemy
- believed they could turn any cheap metals to gold
- almost like magicians
Contributions of Alchemists
- ==mercury, sulfur, and antimony==
- properties of some other elements
- ==production of gunpowder==
John Dalton
- beginning of modern atomic theory
- ^^billiard ball model^^
Theories
- all matter is composed of atoms
- all atoms of a pure element are identical
- different atoms combine in whole number ratios to form compounds
- ==atoms are separated, combined, rearranged in chemical reactions==
Size of an atom
- nucleus is tiny compared to an atom
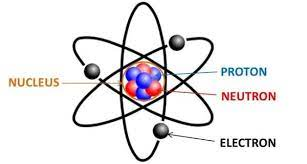
Basic Structure
- atoms can be divided into 3 subatomic particles (electron, proton, and neutron)
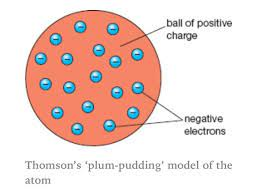
John Thomson (1904)
- ^^Plum pudding model^^ (electrons embedded in positively charged nucleus)
- ==electrons, isotopes, mass spectrometer==
Earnest Rutherford
- ^^nuclear model,^^ almost all of the mass was in the nucleus, positively charged
credited with discovery of: ==protons and dense nucleus==
99.97% of atom’s mass is in nucleus, 0.0001% in volume
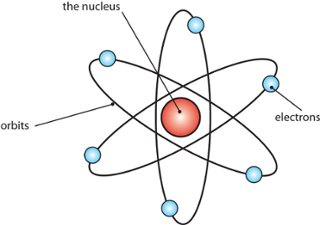
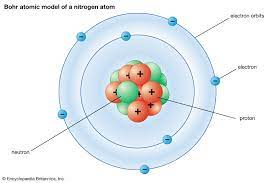
Bohr
- ^^planetary model^^, electrons travel in fixed orbits
- orbit determined by electrons’ energy
Schrodinger
- math to determine where the electrons are
Chadwick
- confirmed existence of neutrons