From TB ( 1 )Alkenes & Electrophilic addition reaction
What is Alkenes ?
→ General Formula Cn H2n + 2
→ At least one C=C double covalent bond
→ Unsaturated : they can make more bonds with an extra atoms in addition reactions
→ Reactive + high electron density : there’s two pairs of electrons in the C = C bonds
With cyclic Alkene : there’s 2 few H atom than an open chain, in a C = C can make one less bond with hydrogen.
Electrophilic Addition Reaction
→ The double bond in an Alkene opens up and atoms are added to the carbon atoms
→ the double bond has plenty of electrons, and is easily attacked by electrophilies
( electron lone pair acceptors ) < short of electron, so attracted to ares with high electrons.
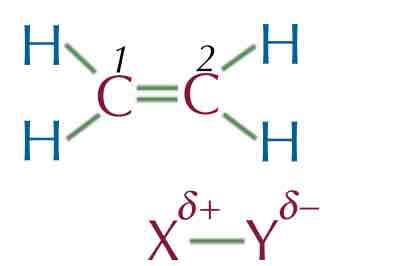
Example 1 : Ethene with an electrophile X-Y
CH2 CH2 + X-Y → CH2 X CH2 Y
The double C=C repels the electron in X-Y, which polarises the X-Y bond
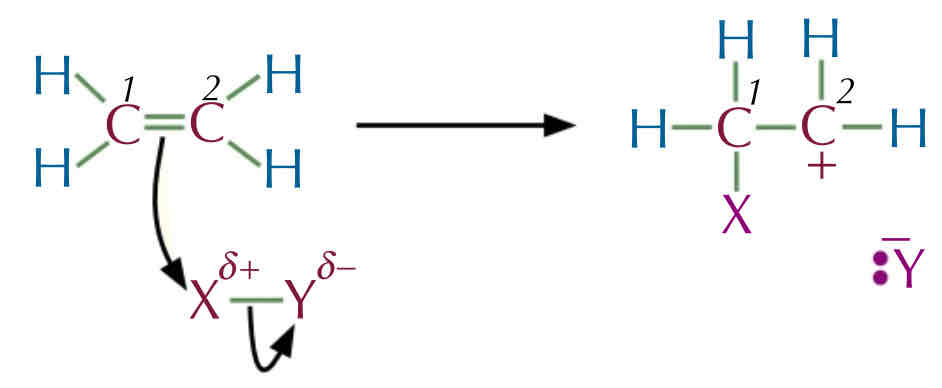
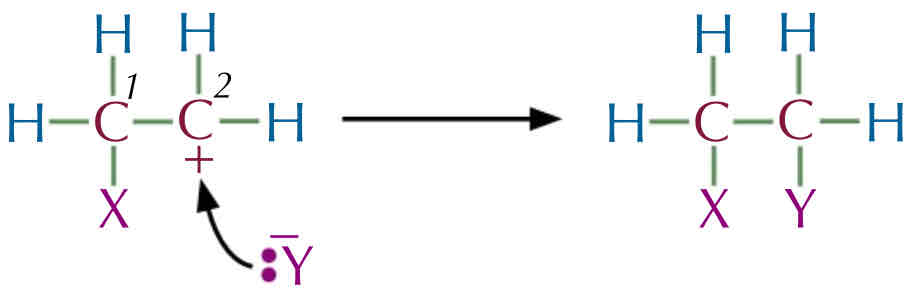
Carbocation Intermediate :
The 2 electron from the C = C attacks the s+ X atom → to form new bond between carbon 1 and the X atom.
The X—Y bond breaks and the electron from the bond are taken by the Y atom to form a negative ion with a lone pair of electron.
Carbon 2 is left with a positive charge , since the double bond is broke carbon 1 and took the electron to form a bond with X atom.
The Y- ion acts as a nucleophile, attacking the positively charge carbocation, donating its lone pair of electron → forming a new bond with carbon .
The X-Y molecule has been added to the Alkene across the double bond to form a saturated compound. Alkane *
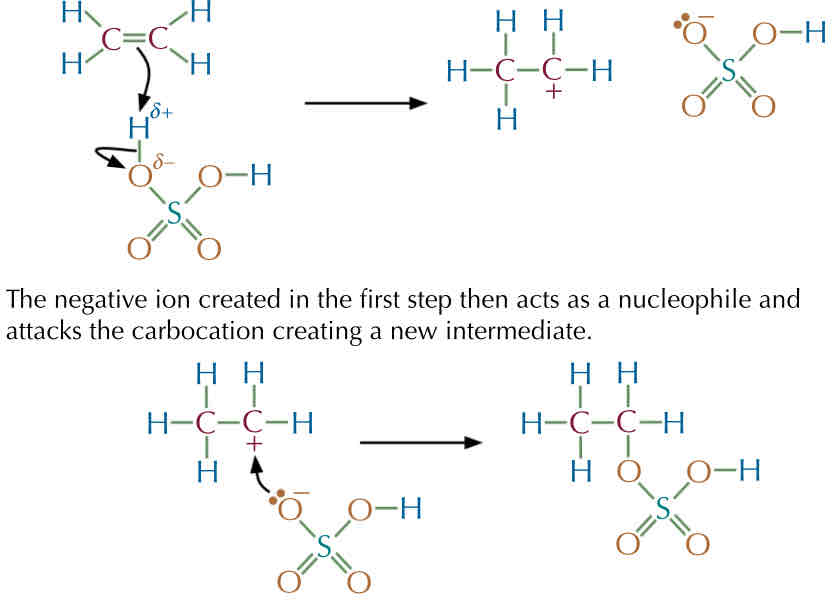
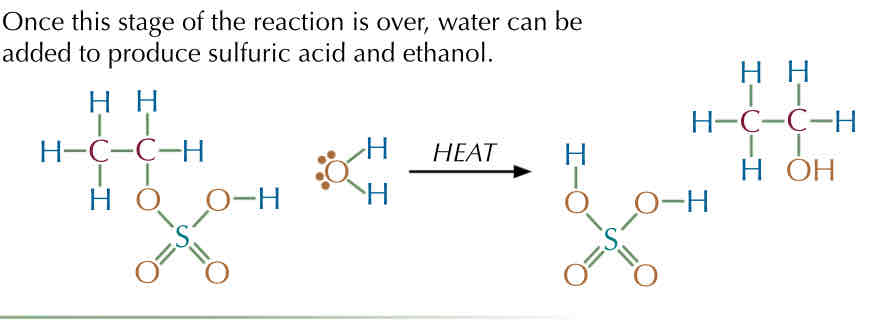
Example 2 : Mechanism with ethene with water → H2 SO4 —> Ethanol
The C =C bond attacks a hydrogen s- atom on the H2 SO4 molecule → new bond is form between one of the carbon and hydrogen, and the electron from the O -H bond are taken by the oxygen atom ( lone pair )
The 2nd carbon is let with a positive charge : it has lost the electron from the double bond.