Lecture 12 Non-Covalent Interactions
Learning Outcomes
Identify polar and non-polar molecules
Use electronegativity differences to identify polar bonds
Determine which molecules exhibit different forms of non-covalent interaction.
Identify structural features that allow hydrogen bonding
Identify structural features that allow ion-dipole interactions and dipole-dipole interactions
Determine which molecules can exhibit large temporary or momentary dipole interactions (dispersion forces)
Order the importance of the various non-covalent interactions
Use non-covalent interactions to explain observations of physical properties
Use non-covalent interactions to explain boiling points and solubilities
Non-Covalent Interactions
Non-covalent interactions: these are attractive forces that do not involve the sharing of electrons between atoms (unlike covalent bonds).
They refer to the interactions between molecules or between different parts of larger molecule.
Physical Properties and Non-Covalent Interactions
Non-covalent interactions are crucial for the physical properties of chemicals.
E.g. Boiling Points and solubility.
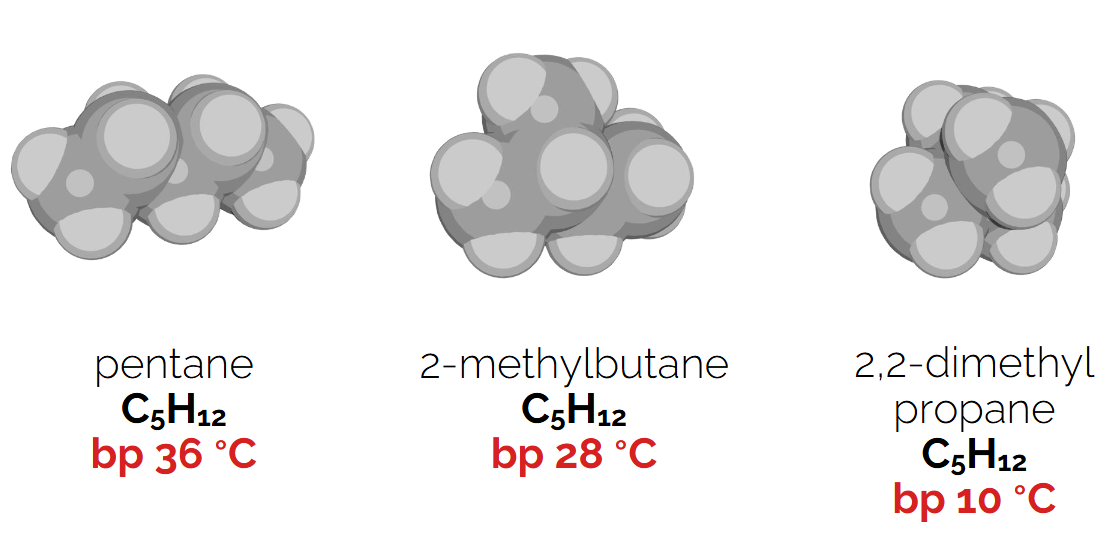
Increasing branching decreases boiling point due to lesser surface area contact and reduced van der Waals interactions..
Electronegativity and Bond Polarity
Electronegativity: The tendency of an atom to attract electrons in a bond.
More electronegative atoms attracts electrons
Bonds are non-polar if there is no electronegativity difference
If one atom is more electronegative it will attract electrons -the bond is polar
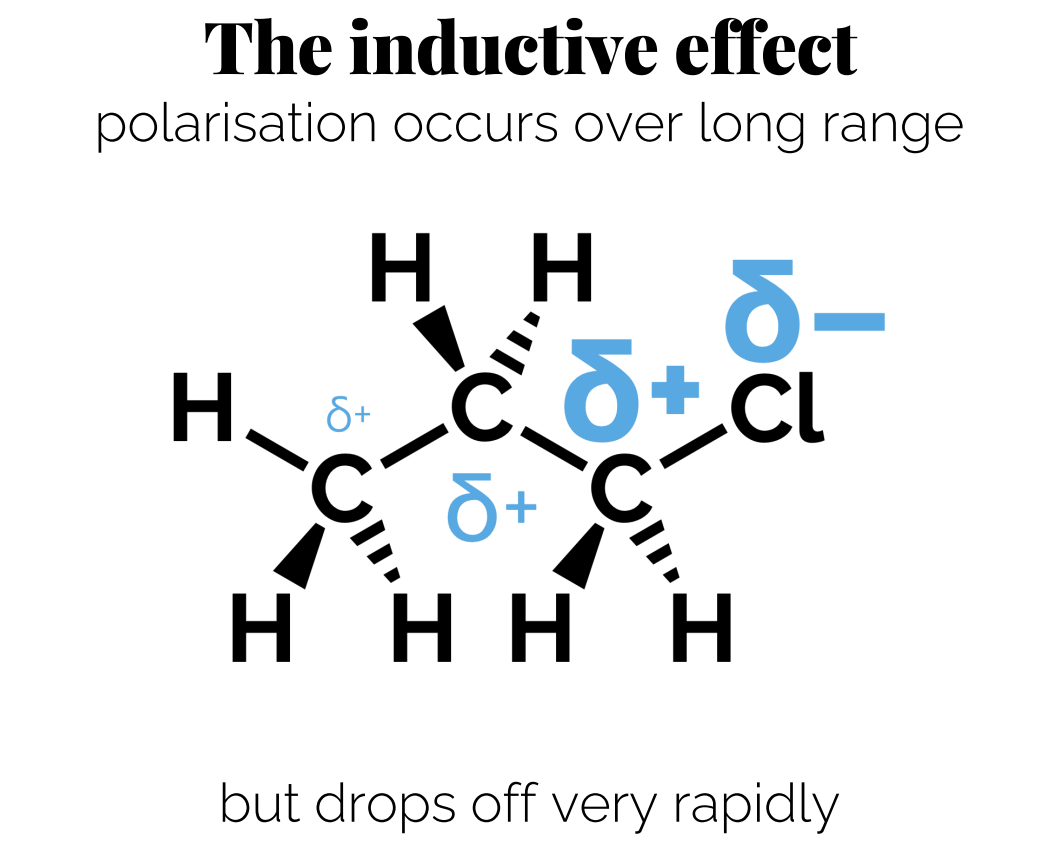
The inductive effect: If the dipole is very strong the polarization can occur over a long range. ( is not significant but is important to be aware of it)
Non-covalent interactions are additive- the sum of many small interactions is greater than just one large interaction.
Bond Dipoles
A dipole is the separation of two partial charges.
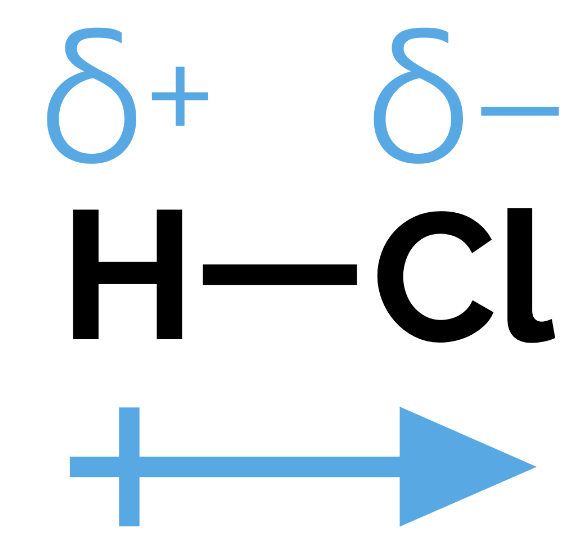
Molecular vs. Bond Dipoles
Molecular dipoles result from adding up all the bond dipoles, taking into account direction, in a molecule.
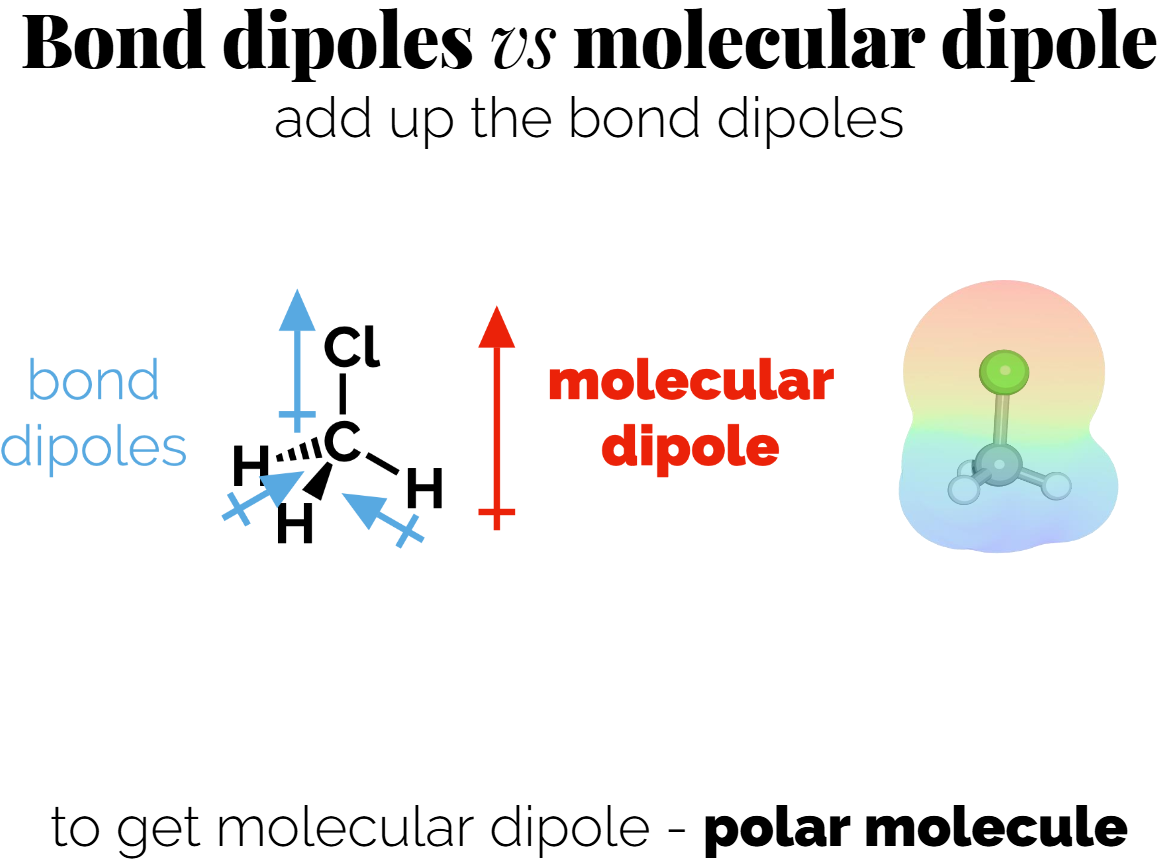
If the dipoles in a symmetrical molecule cancel each other out, the molecule may be classified as non-polar. (e.g., CO₂ has no molecular dipoles).
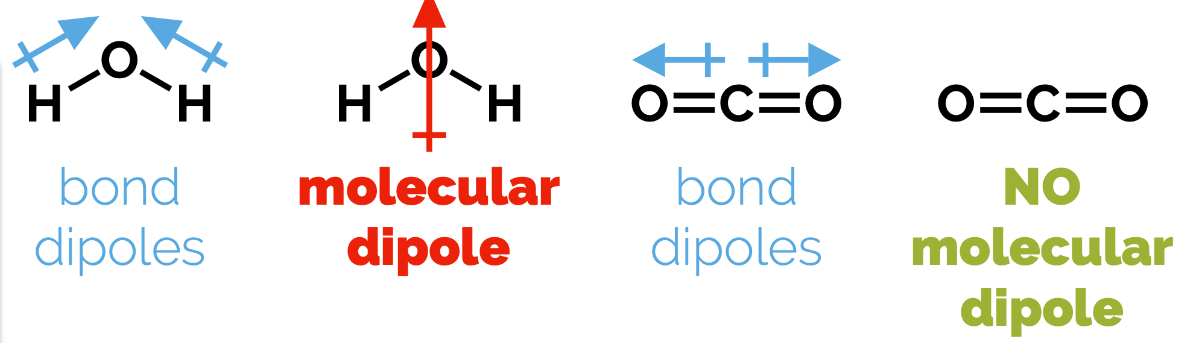
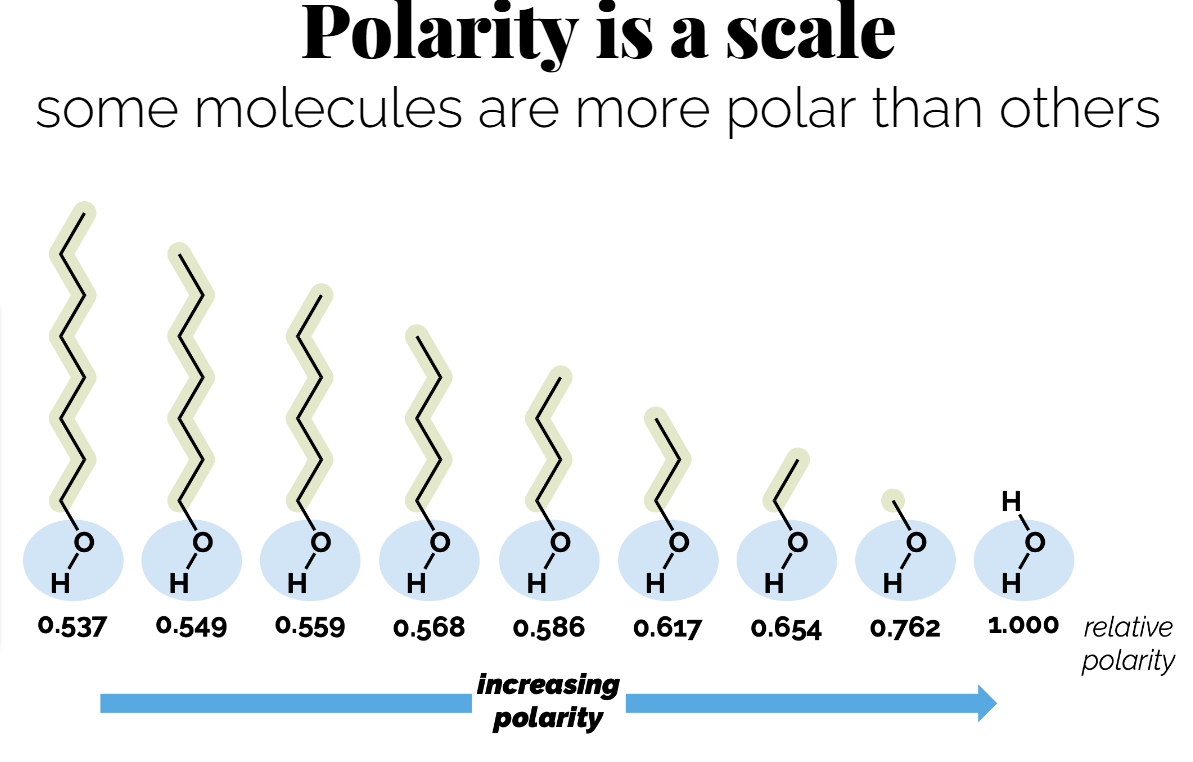
Polarity is a scale: some molecules are more polar than others.
Non-Covalent Interactions
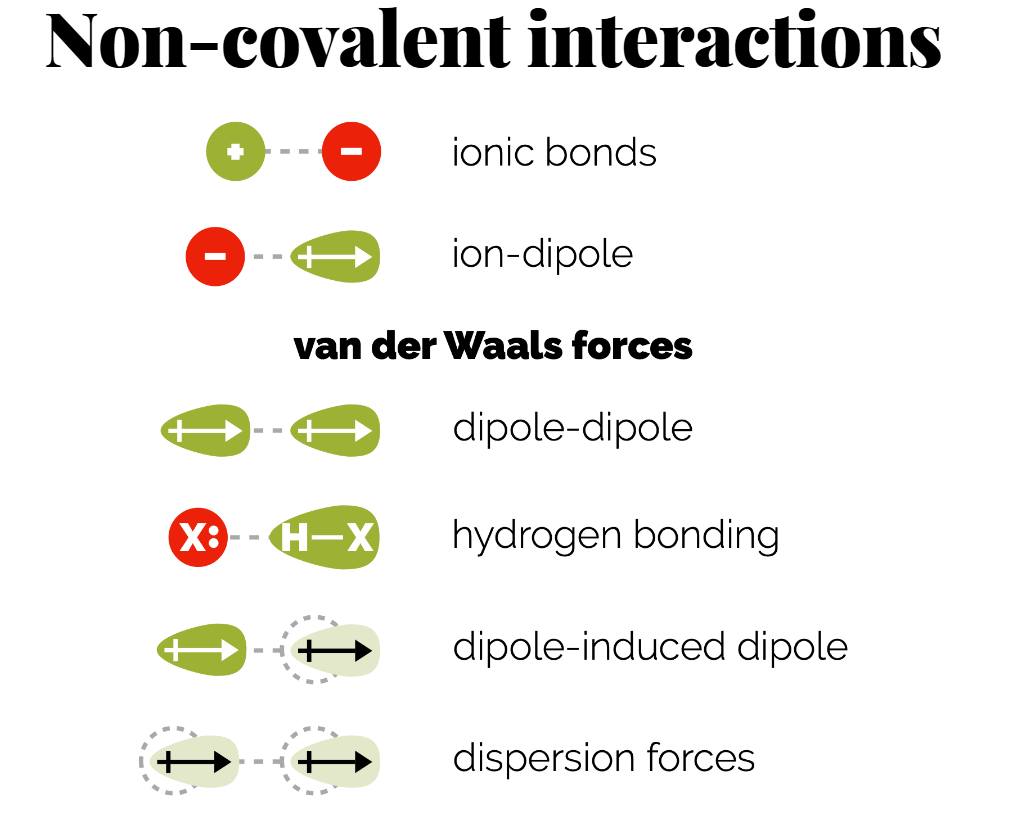
Types of Non-Covalent Interactions:
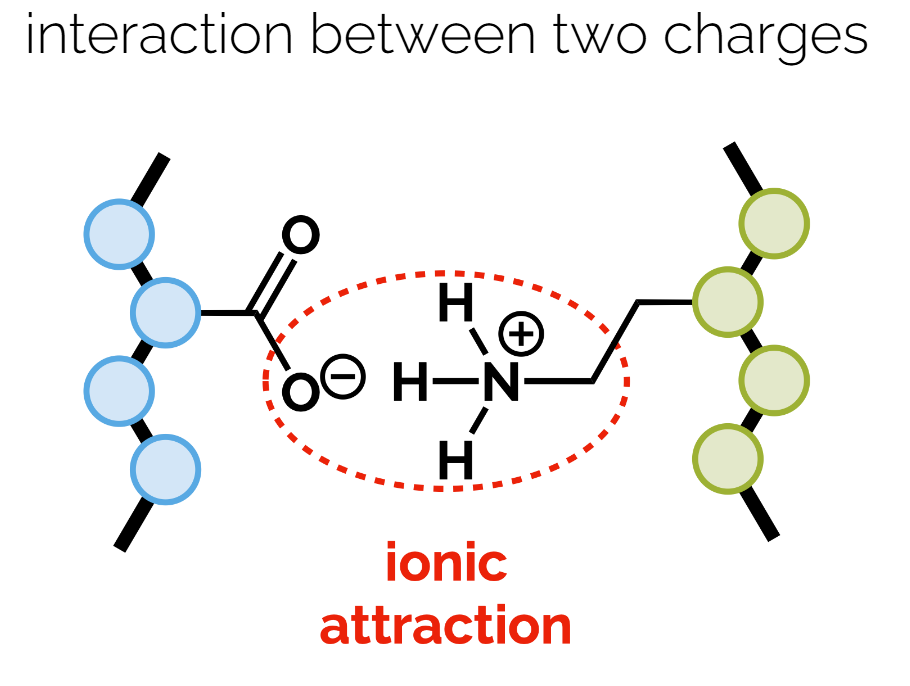
Ionic Bonds: Interactions between charged ions. Example: Salt bridges in protein structures.
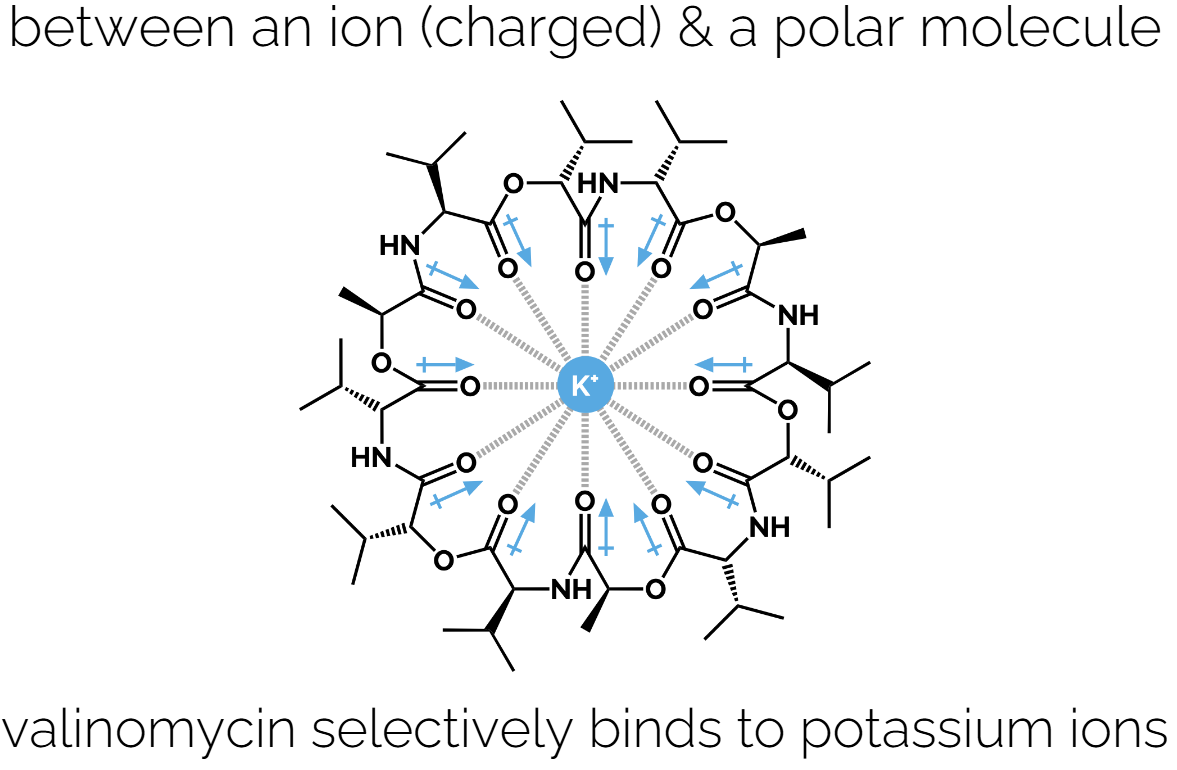
Ion-Dipole Interactions: Attraction between an ion and a polar molecule. E.g., why salts dissolve in water. Additive ( if enough water molecules then there smaller dipoles will be more attractive to the ion that the other ion)
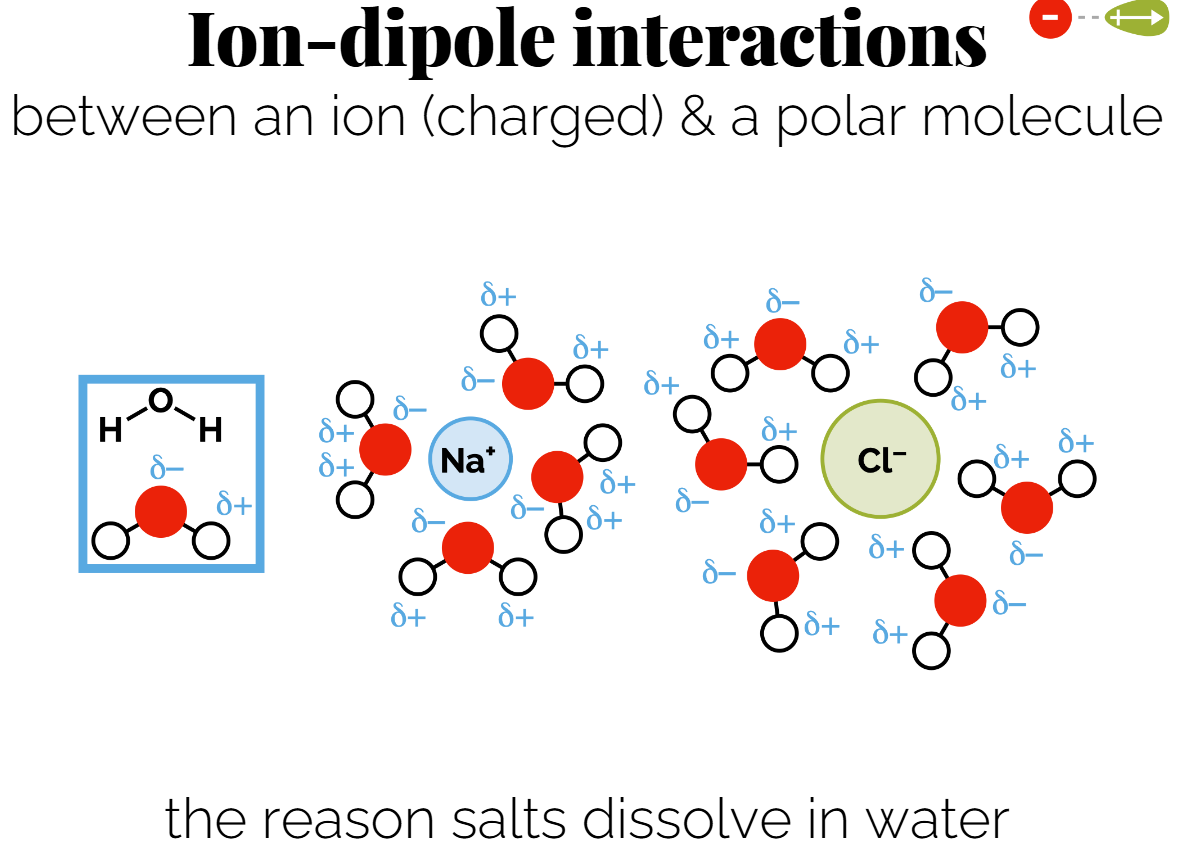
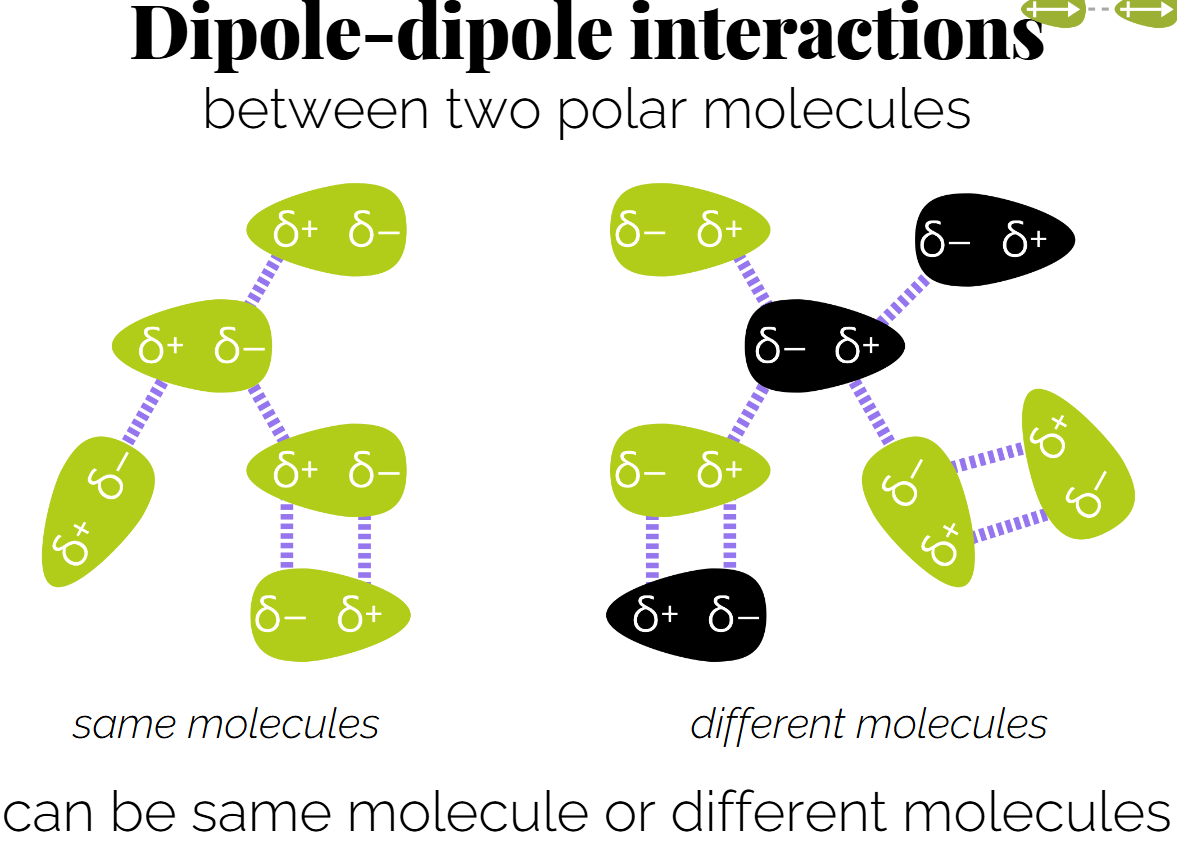
(permanent) Dipole-Dipole Interactions: Occur between two polar molecules. (can be same molecule if its a long molecule) - can explain different boiling points and solubility.
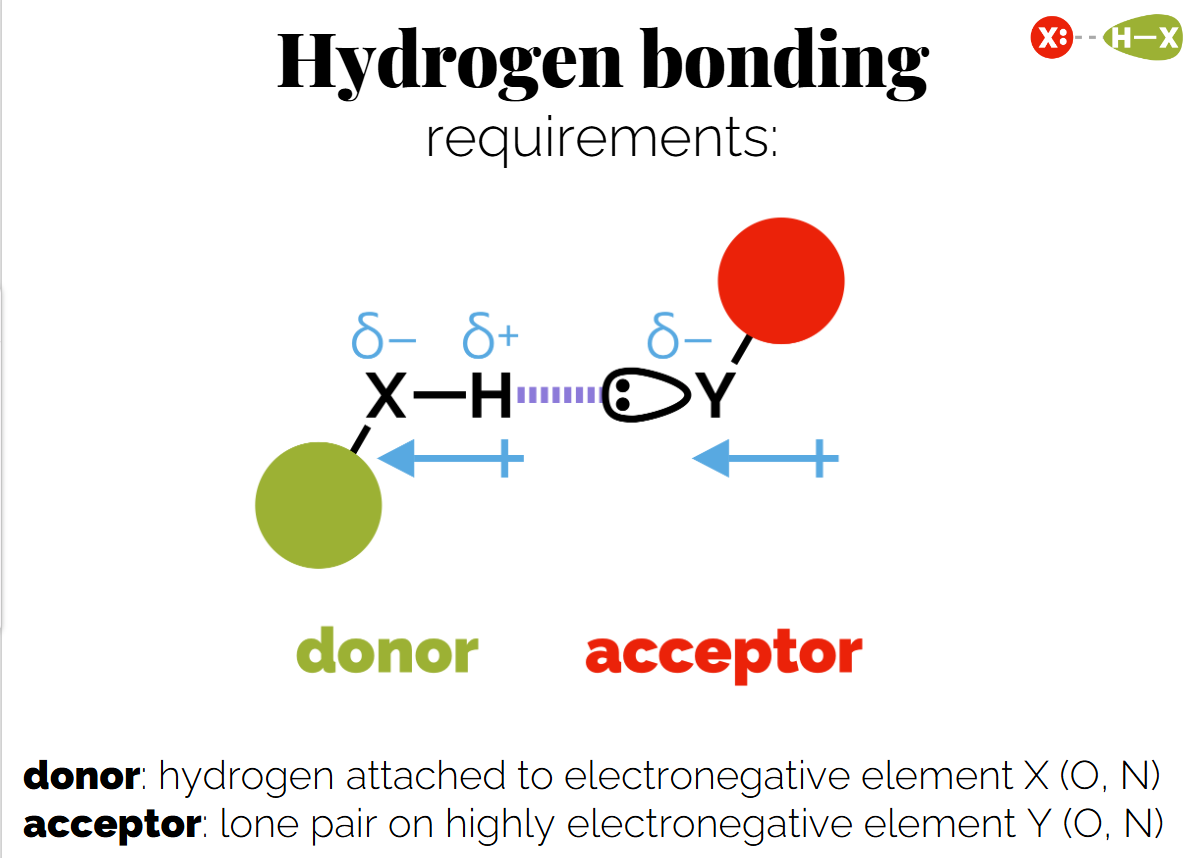
Hydrogen Bonds: A strong dipole-dipole interaction involving hydrogen and electronegative atoms (O or N).
If both molecules have acceptors and donors the overall hydrogen bonding is greater.
(temporary dipole-dipole)Dipole-Induced Dipole: Non-polar compounds can be induced to become polar when near polar molecules.
are short lived as molecules are always moving - because there are momentary the are considered weak interactions.
They are the reason everything (not just like-in like) can dissolve to an extent in water=> partitioning

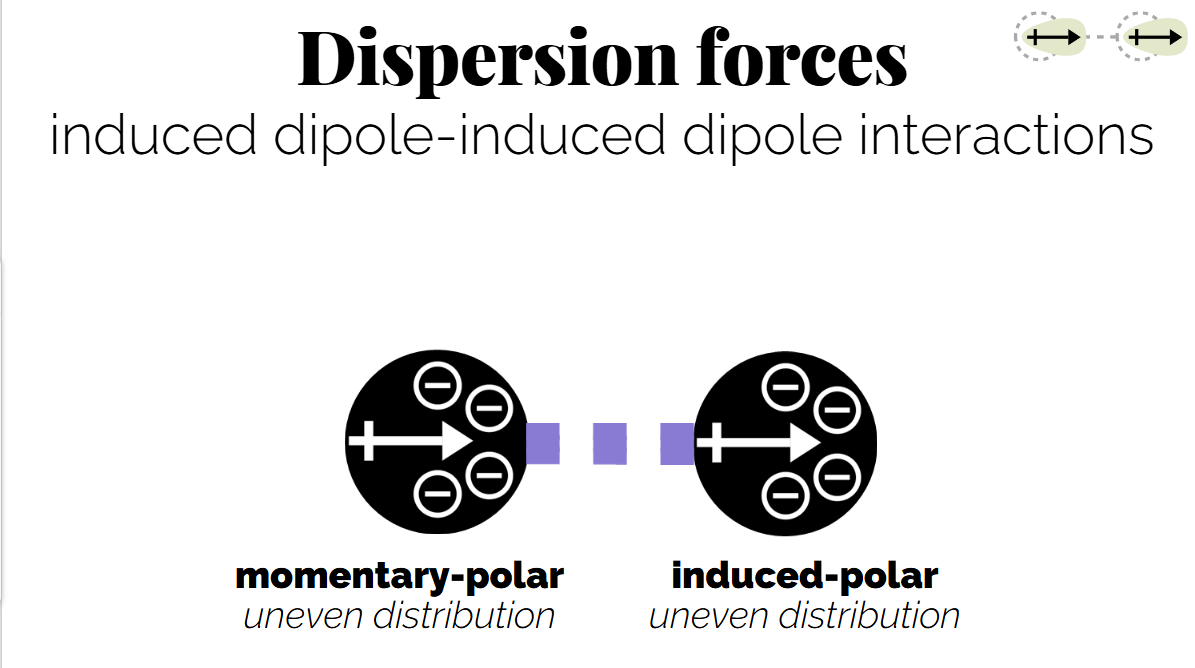
(temporary dipole-dipole) Dispersion Forces: Weak interactions that occur from transient dipoles induced in all molecules.
A dipole induced my an induced molecule, and instantaneous dipoles(from electron distribution)
The size of the molecule is important as a larger molecule = larger electron cloud = larger the transient dipole.
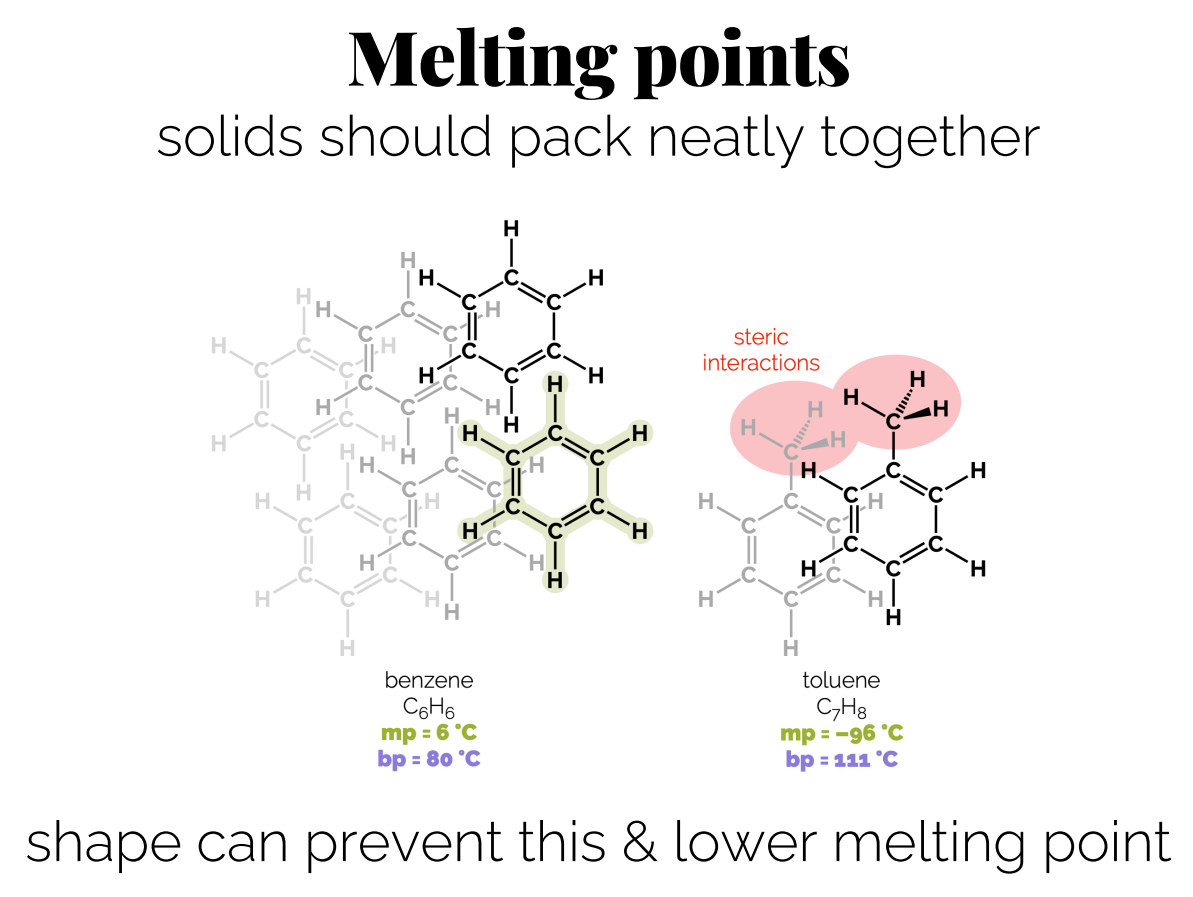
The shape of the molecule is also important as a branched molecule has a smaller surface area compared to a linear molecule, leading to weaker dispersion forces due to less contact area for interaction between molecules.
Momentary short lived-very weak(but are additive so can collectively be strong).
Dispersion forces are what hold non-polar molecules together
Physical Properties Influenced by Non-Covalent Interactions
Boiling Points: Higher boiling points correlate with more or stronger interactions,
e.g., acetone has a higher boiling point than butane due to stronger dipole-dipole interactions compared to acetone which only has weaker dispersion forces.
Melting Points: Stronger or more interactions, lead to higher melting points; substances that can pack tightly together will have higher melting points (packing only effects melting points because once they are liquid packing isn’t an issue so they can move around).
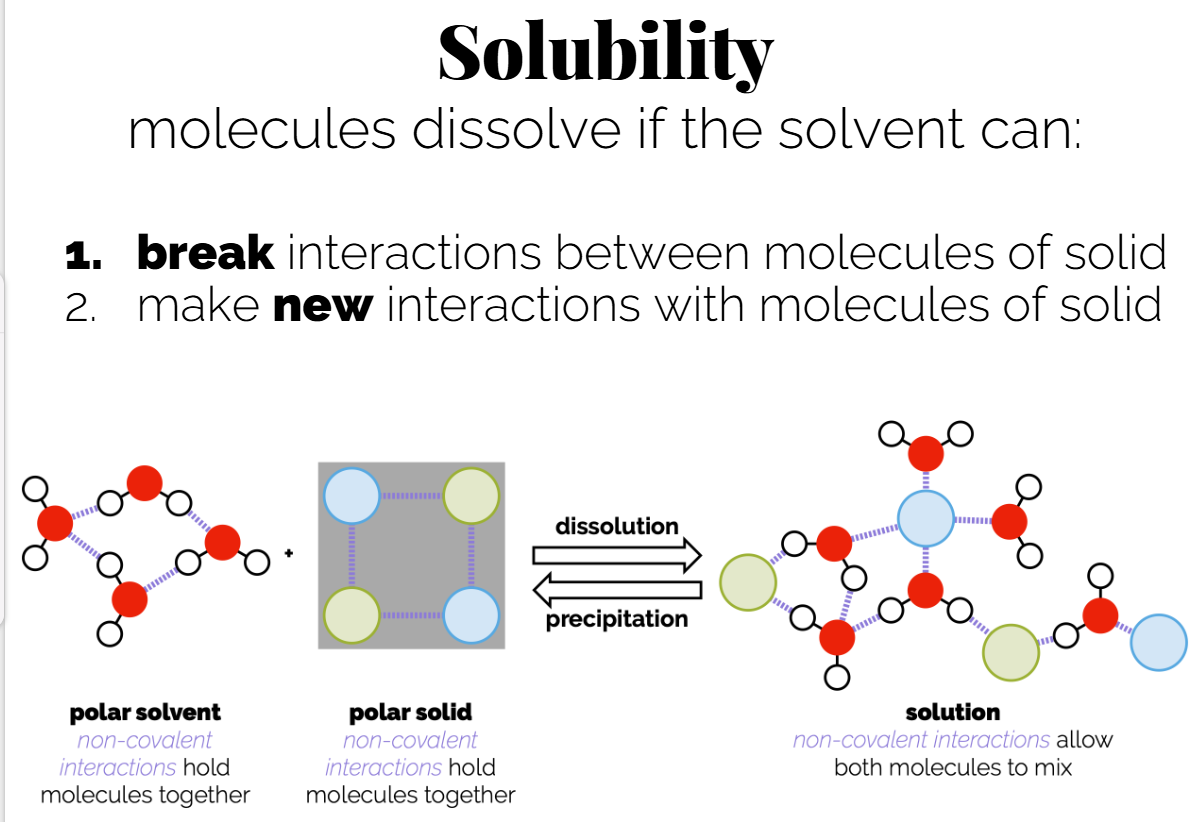
Solubility: "Like dissolves like"; polar solvents can dissolve polar solutes, while non-polar solvents dissolve non-polar solutes.
Example: NaCl dissolving in water through ion-dipole interactions.
Practical Application of Non-Covalent Interactions
Micelle Formation: In soap, polar heads interact with water while non-polar tails trap grease, illustrating the utility of non-polar and polar interactions in cleaning.