Molecules for life
Chemistry of life
All living organisms are made up of atoms, which combine chemically to form molecules, and these make up the basic units of life - cells.
Cells, tissues, and organs:
Plant and animal cells, the basic structural and functional units of living organisms, have a complex organisation that enables them to carry out the seven basic characteristics of life.
Movement
Respiration
Sensitivity
Growth
Reproduction
Excretion
Nutrition
Cells are differentiated and form tissues that perform certain functions. Tissues are organised into organs that are specialised to perform particular functions.
Molecules for Life
Chemical compounds are divided into 2 groups: inorganic and organic.
Inorganic Compounds
Inorganic compounds are chemical compounds that do NOT contain the element carbon (C) and are not produced by living organisms.
Water
The most important inorganic compound for living organisms.
NO WATER = NO LIFE.

H2O = 2 hydrogen molecules and 1 oxygen atom.
Functions of water:
Solvent
Mineral salts in the soil dissolve in water so that the roots of the plant can absorb them.
O2 & CO2 must be dissolved in water before they can enter or leave the body cells.
Medium in which chemical reactions occur
All chemical reactions in living organisms occur in water.
Reagent during hydrolysis
Water breaks polymers (larger macro-/giant molecules) down into their monomers (smaller building blocks).
Transporting agent
Digested nutrients, metabolic waste products, gases (CO2 & O2 in blood), as well as mineral salts are transported in water.
Lubricant
Makes chewing and swallowing easier.
Aids in the movement of food through the rest of the alimentary canal.
Water fluids surround the eyeballs and joints to prevent friction.
Regulates body temperature
water loss through sweat, which evaporates and cools the body.
Gives shape and rigidity
Animals such as earthworms and jellyfish have hydrostatic skeletons that give their bodies shape and rigidity.
Water in the vacuoles of plant cells causes an internal pressure that pushes against the cell wall - this pressure is known as turgor pressure and keeps plant cells firm.
Mineral salts
Minerals are inorganic substances/metals/elements that are present in rocks and soil, but are essential for all living organisms.
Minerals combine with other elements and compounds to form mineral salts, which can be absorbed by plant roots from the soil.
Humans and animals obtain mineral salts from their food and water.
Micronutrients - mineral salts that are needed in large quantities
Macronutrients - mineral salts that are needed in small quantities
Importance of mineral salts in plants and animals:
Macronutrients:
Element | Function | Deficiency disease |
Calcium (Ca) |
| Humans: rickets (children), osteoporosis, and osteomalacia Animals: weak root growth |
Phosphorus (P) |
| Humans: rare Plants: stunted plant growth |
Sodium (Na) |
| Humans: muscle cramps Plants: unknown |
Potassium (K) |
| Humans: rare Plants: yellow and brown leaf margins |
Micronutrients:
Element | Function | Deficiency disease |
Iron (Fe) |
| Humans: anaemia Plants: chlorosis (yellowing of leaves) |
Iodine (I) |
| Humans: goitre (swelling of the neck) |
Iron-deficiency anaemia is a type of anaemia caused by a lack of iron, which the body needs to make haemoglobin. Without enough iron, the blood can't carry enough oxygen, leading to tiredness, weakness, and pale skin.
Phosphates (PO4) and nitrates (NO3) are also mineral compounds.
Phosphate ions are the main source of the element phosphorus (P) in plants. Phosphates are found in most foods, and deficiency seldom occurs in humans.
Nitrate ions are the main source of the element nitrogen (N) that is needed for protein synthesis. Nitrogen forms part of the nucleotides of DNA and RNA and is also needed for the synthesis of chlorophyll in plants.
In humans, a lack of nitrogen-containing protein results in the deficiency disease kwashiorkor.
Kwashiorkor mainly results in stunted growth.
In plants, a lack of nitrate ions causes the deficiency disease chlorosis, where the leaves become yellow due to a shortage of chlorophyll.
Fertilisers and eutrophication:
In over-utilised soils, where crops are grown and harvested regularly, the soil becomes depleted of mineral salts. Fertilisers are continuously worked into the soil to counteract this mineral deficiency. Fertilisers containing mostly nitrogen, potassium, and phosphates are added to the soil in large quantities.
Many of these minerals are leached (washed away) during heavy rains, ending up in rivers and dams. The phosphates cause a drastic increase in the growth of freshwater algae, known as an algal bloom. The increase in algae blocks sunlight from other photosynthesising organisms, which results in the death of many water plants. Dead organic material leads to an increase in decomposer bacteria. Decomposition requires oxygen, and this large-scale decomposition process depletes the oxygen supply to the water. Large numbers of fish and other water organisms die as a result of an oxygen shortage. This phenomenon is known as eutrophication.
Organic Compounds
Organic compounds are chemical compounds that contain the element carbon (C) and are produced by living organisms.
Organic compounds are composed of carbon, hydrogen (H), and oxygen (O)
Some also contain nitrogen (N), phosphorus (P), and sulfur (S).
Carbohydrates:
Made up of the elements: carbon, hydrogen, and oxygen.
H atoms : O atoms = 2 : 1
Carbohydrates are made up of ring-shaped units know as saccharides.
Monosaccharides: Disaccharides: Polysaccharides: | One saccharide Two monosaccharides Many monosaccharides |
Mono - and disaccharides are sugars because they taste sweet and are water soluble.
Polysaccharides are not sugars as they do not taste sweet and are insoluble in water.
Monosaccharides are single-ring structures.
glucose
fructose
galactose
(Glucose is the base building block of all carbs)
A disaccharide is formed when 2 monosaccharides are joined.
One water molecule is removed to form a disaccharide.
This reaction is known as a condensation reaction.
maltose
sucrose
lactose
glucose + glucose
= maltose + H2O
glucose + fructose
= sucrose + H2O
glucose + galactose
= lactose + H2O
Polysaccharides are long chains of monosaccharides that bind to one another by a condensation reaction.
starch
glycogen
cellulose
chitin
Starch is the form in which glucose is stored in the plant. When the plant needs energy, the starch is broken down and the glucose is released.
Glycogen is the form in which excess glucose is stored in the liver and skeletal muscles of humans and animals. When energy is needed, glycogen is converted back to glucose.
Cellulose forms the structural component of plant cells.
Chitin forms the structural component of cell walls in fungi and exoskeletons in arthropods and molliucs. Under extreme pressure, chitin becomes limestone and marble.
Importance of carbs in animals and plants:
Main source of energy
Most important fuel molecule.
During respiration, energy is released from glucose while CO2 and H2O are formed as waste products.
Source of reserve energy
Structural component
Plant cell walls consist of cellulose, which strengthens the cell wall.
The test for the presence of glucose we use the Benedict’s solution (a deep blue liquid)
Iodine on the food.
Prepare a water bath. Take a heat-resistant beaker, fill it with water, and heat it over a Bunsen Burner.
Pour ± cm3 of Benedict’s solution into a test tube.
Add about 10 drops of the test solution (Energade, Oros, or fruit juice) to the Benedict’s solution.
Place the test tube into the water bath and examine the solution after 4 mins
The results can be presented as follows:

Blue - no glucose present
Green - low concentration of glucose
Yellow
Orange
Brink
To test for starch:
Take a piece of test material (bread or potato) and place it in a saucer.
Drop 2-3 drops of yellow-brown iodine solution onto it.
If the test material turns a blue-black colour, starch is present.
Lipids (fats and oils)
Lipids are made up of the same 3 elements as carbs: carbon, hydrogen, and oxygen.
The ratio of H : O, lipids contain more than double the number of H atoms that O atoms.
Lipid molecules consist of 2 types of building blocks - glycerol and fatty acids.
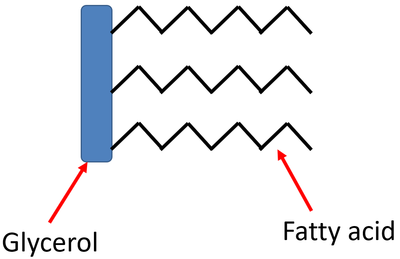
Lipids are:
insoluble in water
soluble in ether and alchol
hydrophobic - water-repelling
Saturated Fats | Unsaturated Fats |
Saturated fats help the body make cholesterol, a substance important for cell membranes and hormones | Come mostly from plants (e.g., olive, canola, sunflower oil) and are liquid at room temperature |
However, too much saturated fat can raise cholesterol levels in the blood. Cholesterol travels in the blood as either LDL (bad) or HDL (good). HDL removes excess cholesterol, while high LDL levels increase the risk of cardiovascular disease by causing fatty deposits (plaque) in arteries. This leads to a narrowing called atherosclerosis, where blood clots may form.
The coronary artery supplies the heart's muscle tissue with oxygen-rich blood. If it becomes completely blocked by a clot, the heart muscle is deprived of oxygen, and a heart attack occurs. If the artery is only partially blocked, the heart gets less oxygen, leading to chest pain (called angina) and tiredness during physical or emotional stress.
Importance of lipids in animals and plants:
Source of energy
A large amount of energy is released when fat molecules are broken down
Insulating material
Fat is a poor conductor of heat. Marine mammals (whales, dolphins and seals) have a layer of blubber (fat) under the skin that insulates their body heat from the cold sea water. This layer of bubbler also helps them to float.
Shock absorption
Kidneys and eyes are surrounded by a layer of fat to protect them from mechanical injury.
Structural component of cell membranes
Cell membranes consist of a double layer of phospholipid molecules with a protein layer on either side
Waterproofing
A fatty substance, cutin, forms a cuticle that covers the epidermal cells of plants. The cuticle prevents excessive water loss. Birds have oil glands that secrete oil to make their feathers waterproof.
Absorption of vitamins
Fat-soluble vitamins A, D, E, and K can only be absorbed when dissolved in fat
Source of water
Animals that hibernate in cold weather, or others that survive in hot, dry conditions, store fat that is broken down to CO2 and H2O during cellular respiration.
The camel, which thrives in desert areas, is an animal that survives using metabolic water.
We test for fats using ether:
Grind the test material (eg, peanuts) to a fine consistency.
Place a small amount in a test tube.
Add 5 cm3 of ether and shake the test tube well.
Allow the test tube to stand for approximately half an hour.
Filter the ether (finely ground peanuts will stay behind).
Allow a drop of the filtrate (liquid that filtered through) to fall onto a clean filter paper.
The ether will evaporate quickly.
If any fats are present, a translucent, greasy spot will form on the filter paper.
Proteins
A protein is a polymer (a large molecule) with amino acids as monomers.
Amino acids are made up of the elements carbon (C), hydrogen (H), oxygen (O), and nitrogen (N). In some amino acids, phosphorus (P), sulphur (S), and iron (Fe) are also present.
There are only 20 different amino acids.
The sequence and type of amino acids determines the type of protein.
The bond/link formed between 2 amino acids is known as a peptide bond.
Dipeptide: 2 amino acids linked by a peptide bond
Tripeptide: 3 amino acids linked by peptide bonds
Polypeptide: long chain of amino acids linked by peptide bonds
Protein: 51+ amino acids linked by peptide bonds
Every protein has its own particular shape. The long chains of amino acids form folds and loops that can fold back onto themselves. The number of amino acids, sequence of amino acids, and specific shape of a protein determines its function.
Denature - protein loses its shape and can no longer perform its function
Importance of proteins in animals and plants:
Structural component of protoplasm
About 15% of protoplasm consists of protein
Building materials
Structural component of cell membranes
Together with phospholipids, it forms the cell membrane that surrounds a cell.
Plays a role in the permeability of cell membranes
Proteins act as carrier molecules that transport other molecules through the cell membrane.
Source of reserve energy
Excess amino acids are broken down into urea and glucose.
Hormones regulate processes in the body
Some hormones, like insulin and adrenaline, are proteins and act as chemical messengers
Haemoglobin transports O2 and CO2 in the blood
Protect the body against disease
Antibodies are blood proteins that respond to antigens and fight bacteria and viruses.
Chromosomes carry hereditary material
Chromosomes are made up of proteins and DNA
Act as buffers
Proteins keep the pH of body fluids constant as they can release hydrogen atoms (to an alkaline solution) and take up hydrogen atoms (from an acidic solution).
Enzymes control metabolic processes in the body
Enzymes are proteins that accelerate chemical reactions.
Test for proteins: (The Biuret test)
Grate or grind the test material (beans) or use soup powder or milk powder. Add a little water and boil until it forms a thin soup.
Pour about 5 cm3 of soup into a test tube and add 2 - 3 drops of dilute copper sulphate solution (light blue)
Add 5 - 10 cm3 of sodium-hydroxide solution (clear) to the mixture.
Shake the test tube gently.
Heat the mixture over a Bunsen burner to obtain a deeper colour.
Blue = no protein present
Purple/violet = protein present
Enzymes
Enzymes are proteins that accelerate chemical reactions inside living cells. Without them, vital reactions essential for life would take place too slowly to sustain life.
Substances that accelerate chemical reactions are known as catalysts.
Enzymes are, therefore, biological catalysts.
In order for a chemical reaction to occur, the reaction needs activation energy to initiate the reaction.
An enzyme is a biological catalyst that accelerates a chemical reaction by lowering the activation energ, without itself being changed by the reaction.
Chemical reactions that take place in living cells are known as metabolic reactions.
Anabolic reaction
Catabolic reaction
During this type of reaction, a complex molecule is built up from simple molecules. Energy is usually required.
During this type of reaction, a complex molecule is broken down into simple molecules. Energy is usually required.
During the digestion of food, complex molecules (larger macromolecules) are broken down into simple molecules (smaller, soluble molecules) by adding H2O.
This catabolic process is known as hydrolysis.
Enzyme action:
During a chemical reaction, a substance/s is/are changed from one form to another.
The substance on which the enzyme acts is known as the substrate.
The substance formed during the reaction is the product.
Enzymes are proteins, and each protein has a particular shape to enable it to perform its specific function.
Enzymes are substrate-specific; in other words, a specific enzyme can only act on a specific substrate.
There is a region on the enzyme known as the active site. The substrate molecule fits into the enzyme’s active site as a key fits into a lock. This ensures the specificity of the enzyme.
A temporary enzyme-substrate complex is formed.
The enzyme lowers the activation energy for the reaction.
The substrate changes chemically and leaves the active site.
The enzyme remains unchanged during the reaction and it ready to bind with the next substrate molecule.
The action of enzymes is known as the lock-and-key model.
Influence of temperature and pH on enzyme action:
Temperature:
Enzymes are very sensitive to temperature changes
Low temperatures make enzymes temporarily inactive
The temperature at which most enzymes work the best is known as their optimum temperature
At high temps, usually above 45’C, the shape of the enzyme begins to change and the substrate no longer fits into the active site. The enzyme loses its structure and function - it has denatured.
At lower temperatures, enzymes become inactive. If enzymes freeze, they also denature.
Denaturing is an irreversible process. When an enzyme denatures, it cannot return to its original shape when the temp is lowered again. When an enzyme becomes inactive at low temps, however, it can become actiive again once the temperature rises.
pH:
Each enzyme has a narrow pH range within which it can function. This is known as the optimum pH.
Some enzymes work optimally in acidic medium, whereas others work better in neutral or alkaline medium.
Enzymes denature at extreme pH levels.
Properties of enzymes:
Enzymes are spherical proteins.
Enzymes are sensitive to changes in temperature - high temperatures denature enzymes, whereas lower temperatures make them temporarily inactive.
Enzymes are sensitive to changes in pH - enzymes denature if the pH changes drastically (too acidic/alkaline)
Enzymes are substrate-specific
Enzymes can be used over and over again
A small amount of enzyme can change a large amount of substrate.
Enzymes in everyday life
Biological washing powders contain enzymes that break up/remove biological stains. These enzymes usually act on proteins and are known as proteases.
Stains caused by blood, egg yolk, and sweat consist of proteins and are difficult to remove from clothes. The enzymes in washing powder break the proteins into smaller, soluble molecules that are easily removed by cleaning agents in the washing powder.
Lactose-free milk and meat fertiliser are also examples of products that contain enzymes.
Nucleic acids
Nucleic acids are made up of the elements carbon (C), hydrogen (H), oxygen (O), nitrogen (N) and phosphorus (P). Monomers of nucleic acids are called nucleotides.
There are 2 types of nucleic acids:
DNA (deoxyribonucleic acid)
RNA (ribonucleic acid)
DNA is found in the nucleus of a cell and forms part of the chromatin network and chromosomes (during cell division).
RNA is found in the nucleus and cytoplasm of a cell and forms part of the ribosomes.
Function - DNA carries the hereditary characteristics and controls the structure and function of the cell.
Function - RNA plays a role in protein synthesis to ensure that the amino acids bind to one another in a certain sequence according to the instructions provided by the DNA
Vitamins
Vitamins are essential for normal metabolism, growth and development of the human body.
They are needed in small quantities and are produced by plants.
Vitamins must be in solution to perform their functions. Vitamins can be classified as water-soluble or fat-soluble.
Water-soluble: |
|
Fat-soluble: |
|
Insufficient vitamins cause deficiency diseases.
Vitamin | Source | Deficiency Disease |
A | yellow vegetables, fish oil, liver, egg yolk |
|
B1 | brown rice, wholegrain bread, yeast, legumes |
|
C | citrus fruit, tomatoes, guavas, green leafy vegetables |
|
D | oily fish, egg yolk, liver, milk |
|
E | leafy vegetables, sunflower seeds, wholegrain and wheatgerm |
|