Periodic Properties of the Elements Notes
Explaining Periodic Trends
Shielding
In multi-electron systems:
Electrons are attracted to the nucleus and simultaneously repel each other.
Nucleus consists of protons (positive charge) and neutrons (neutral charge).
Outer electrons are shielded from the nucleus by core electrons, leading to a phenomenon known as the screening or shielding effect.
Outer electrons do not effectively shield each other from the nuclear charge, resulting in the outer electrons not experiencing the full strength of the nuclear charge.
Effective Nuclear Charge Zeff
Definition: Effective nuclear charge, denoted as Zeff, refers to the net positive charge that attracts a particular electron located in the valence shell.
Quantitative Definition: The simplest method to define the effective nuclear charge quantitatively is:
Where:
Z = nuclear charge (atomic number)
S = number of electrons in lower energy levels.
This method simplifies a complex problem significantly.
Shielding Effect Between Subshells
Within a specific shell, partial shielding occurs, but it is notably less than between shells.
Example of penetration based on quantum model:
2s orbital is more penetrating than 2p orbital.
2p orbital is partially shielded by 2s orbital.
Slater and Electron Shielding
John Clarke Slater (1900 – 1976): An influential American physicist who greatly enhanced the understanding of electronic structure in atoms.
He developed a more comprehensive set of rules to estimate electron shielding that incorporates electron penetration.
How to Use Slater’s Rules
For electrons in the (ns, np) groups:
Other electrons in the same group contribute 0.35 each to the shielding constant .
All electrons in the n – 1 shell contribute 0.85 each to the shielding constant.
All electrons in n – 2 or lower contribute 1 to the shielding constant.
Formula:
Here, Z is the atomic number and is the screening (Slater) constant.
To determine :
Write the electron configuration of the element and group the subshells as follows:
(1s), (2s, 2p), (3s, 3p), (3d), (4s, 4p), (4d), (4f), (5s, 5p),…
Electrons in the groups to the right of the designated electron contribute nothing to the screening constant.
For nd or nf groups:
Other electrons in the same group contribute 0.35 each to the shielding value.
Electrons with the same principal quantum number n and angular momentum quantum number ℓ contribute 1.00 each to the shielding value.
All electrons in n-1 shell and below contribute 1.00 to the shielding value.
Slater’s Rules and Orbital Filling
Observation: The 4s orbital is filled before the 3d orbital.
According to Slater’s rules:
A more stable configuration allows the electron to experience a larger effective nuclear charge Zeff.
Example: For Potassium (K, Z=19), if the last electron is placed in the 4s orbital:
Electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹
Slater’s Rules and Orbital Filling (Continued)
Example: If the last electron is placed in the 3d orbital:
Electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹
Worked Example
Task: Determine the effective nuclear charge ($Z_{eff}$) for a 4s electron in an iron (Fe) atom.
Iron’s electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁶
Trend #1: Atomic Radius – Main Group Elements
Measurement Methods:
Various methods exist for measuring atomic radius, yielding slightly different values:
Van der Waals radius = nonbonding radius.
Covalent radius = radius during bonding.
Atomic radius: Average based on numerous measurements of elements and compounds.
Trends:
Atomic Radius Increases down a group:
The valence shell is farther from the nucleus due to the increase in principal quantum number.
The effective nuclear charge remains relatively close.
Atomic Radius Decreases across a period (left to right):
Adding electrons to the same valence shell causes an increase in effective nuclear charge.
The valence shell is held closer to the nucleus.
Your Turn!
Question: Which group has its elements arranged in order of increasing atomic radius?
A. Al, P, Cl, Ar
B. Rb, Sr, Ca, Mg
C. N, P, S, Se
D. Ne, Ar, Cl, Br
E. H, He, Ne, Ar
Trends in Atomic Radius: Transition Metals
In the same group, atomic size increases down the column.
Atomic radii of transition metals maintain consistent sizing across the d block.
Valence shell is filled as ns², distinct from (n−1)d electrons.
Operations of 4s electrons determine size; inner 3d electrons serve to screen increasing nuclear charge.
Trend #2: Ionic Radius
Definition: The ionic radius refers to the size of a cation or anion.
Observed Trends:
Ionic size increases down the column; larger ions are observed due to higher valence shells.
Ions in the same group share the same charge.
Comparison:
Cations are smaller than their neutral atom counterparts.
Anions are larger than neutral atoms, with cations smaller than anions.
For isoelectronic species:
A larger positive charge results in a smaller cation.
For example: S²⁻ (184 pm) > Cl⁻ (181 pm) > K⁺ (133 pm) > Ca²⁺ (99 pm).
A larger negative charge correlates with increased anion size.
Explaining Trends in Cationic Radius
When atoms form cations,
Valence electrons are removed, resulting in a cation smaller than the original atom.
Newly appointed valence electrons experience a larger effective nuclear charge compared to previous ones, further decreasing the ion's size.
As one traverses down a group:
There is an increase in (n − 1) energy levels, resulting in larger cations.
Across a period:
An increase in effective nuclear charge leads to smaller isoelectronic cations.
Explaining Trends in Anionic Radius
Anions form when electrons are added to the valence shell:
This additional electron increases the size of the anion compared to neutral atoms.
These new valence electrons experience lower effective nuclear charge, leading to size increase.
Down a group, there is an increase in n levels, which makes anions larger.
Moving right across a period results in greater effective nuclear charge for isoelectronic anions, thus decreasing their size.
Ionization Energy (IE)
Definition: Ionization energy is defined as the minimum energy required to remove an electron from an atom or ion, especially in the gas state.
It is an endothermic process (requiring energy input and does not occur spontaneously).
The lowest ionization energy corresponds to the removal of a valence electron (the easiest to remove):
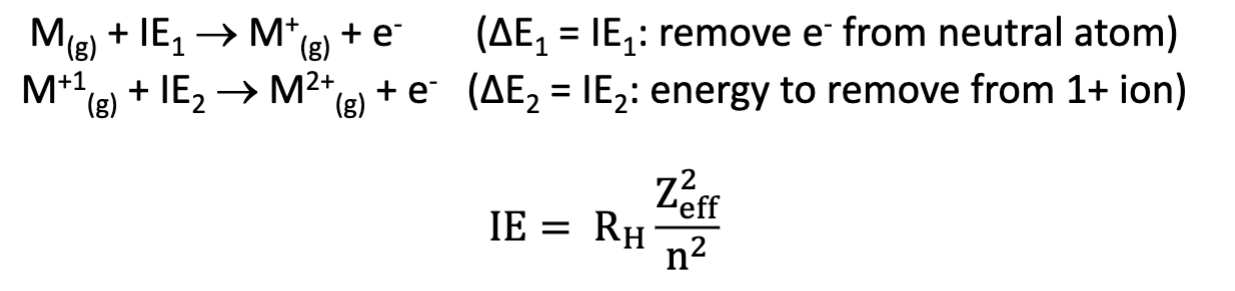
First Ionization Energies
Comparison of filled shells:
n=1, n=2, n=3, n=4, and n=5 filled shells are compared.
Distinctively, alkali metals have specific characteristics regarding their ionization energies.
Trends:
First ionization energy decreases down the group.
Larger orbitals lead to greater distances from the nucleus, thus lesser attraction and effective nuclear charge Zeff.
Increases in first ionization energy generally occur across a period due to increased Zeff on valence electrons:
Each successive electron removal costs more energy, accompanied by increased proton presence and reduced electron presence.
The outer electrons become harder to remove when they are drawn closer to the nucleus.
Each successive valence electron removal results in a regular increase in energy required, with significant jumps when core electrons are removed.
Irregularities in the General Trend of Ionization Energy
Trends are not followed when:
The added valence electron in the subsequent element enters a new sublevel (higher energy level).
The first electron pairs in the same orbital of the sublevel, with electron repulsion reducing energy levels.
.