Chemistry Chapter 8 and 9
Lattice Energy - enegry to conber solid ionic to gas ions
- the formation of these bonds realease energy
when looking at a lattice cube it represents a solid
Melting can relatively quantify lattice energy because it takes more energy to break
Lattice energy is one of the strongest bonding forces
Electronegativity and Polarity
- two types of strong bonds
* Covalent '
* ionic - fro the bonds inbetween there are polar bonds
- A nonpolar covalent bonds have no diffenec in electron negitivity
- Bonds with a diffeence in electo negtiviy ar Polar bonds
* For example Br-Br is nonpolar
* C-Br is Polar because one wants the elctrons mor than the other and they have 2 different poles (dipoles) - Ionic bonds (NaF) usually between Metals and nonmetals
- Electronegativity varies in position on the periodic table
- Electron affinity is the atom by itself and electron negativity is the atom in a compound
- halogens have highest electronegative because they are one away from a stable octect
- a bond between atoms are non polar covalent when the difference is les that .5
- .5 -2 is polar covalent
- greater than 2 is ionic
- A metal and a nonmetal is ionic
The s is a partial charge (negative or positive)
From left to rights elctronegeitiviiy increases
From bottom to top electronegtiviy increases
Polar covalent bonds
Dipole Moments - whats going on with bond polarity
- the more electronegative get the parial negative
- Cross arrow also show the patial negative
- For patiall negative and partal positive allways pick the one that fartheest from florine to be positive
- Dipoles always depends whay it is paird on
- \
Lewis dot symbols
Group 1A and Group 7A- tend to form one bond
all elctron assoiated witht the highest priciple qunatum number are valence
One dot goes in each box for each valence electron
Show ions with that lose wot a plus
Show ions that gain with the full shells and a minus
Drawing Lewis structures
Step1 - Cont the total numver of valence electrons Present, ad for negative charges subtract for positive charges
Step 2- Draw the skeletal structur of the compound, the lease electronegative atom is usually the central atom and eac fo the surrounding atoms
Step 3- use the remaining elecrons to complete octets of the terminal atoms by placing pairs of electrons on each atom , complete the octeet of the most electronegative atom first
Step 4 - place any reaming atoms on the central elctron
Step 5 - IF the central atom ahas fewer than eight electrons move one or more pairs from the terminal atoms to forme multipe bonds bewteen the central atom and terminal atoms.
Reasonance structures are structures with differnt ways to write them, arrows are draw to show they are reasonate
Formal charge - used to determin the most plausible lewis structure when more thatn one possiblity exist from a compund
Formal charge = valecne e- -- assoicated e-
all the adtoms non bonding electrons are assoiated with the atom
half the atoms bodnign electron s bonding atoms are assoinatied with
Free Radicals - Molecules with an odd number or electrons
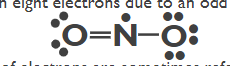
Octets are obeyed for 2P elements
Molecular Geometry
VSEPR valence chell electon pair pefulsion
linear bonds are 180 degrees
Trigonall Panar = 120 degree
Techrahedral 109.5 degrees
Trigonal bypyrimainal
Intermolecular forces
Gives insight on physical or chemicals properties
- Dispersion forces
* anything with electrons has dispersion forces (protons do not )
* an atom with more elcrons has higer dispersion forces
* The shape (contact area) of a molecule disrupts dispersion forces
* the more contact area the greater dispersion force
* The greater the dispersion the more difficult it is to boil/ melt - Dipole - dipole
* requires a permeant dipole
* they interact with other dipoles
* Dipoles connect more negative side to more positive side
* Dipole - Dipole interactions have greater intermolecular forces that molecules with only dispersion forces - Hydrogen bonds
* Stronger that dipole dipole bonds
* covalent bonds between H O N or F - dipole incuced dipoles
* Temporary dipole from a nonpolar and a polar bond - Ion dipole
- ion ion
* This is the strongest - ion/ induced
Valence bond theroy
- all single bonds are sigma bonds but all sigma bonds aren’t single bonds
- Hybridixzed bonds make bonds even
- when a bond is hybridized the obitals are combined
- a single bonds is one domain a double bond is
- Parallel P orbitals make pi bonds
Steps to hybridized bonds
- Count the electron domains, this is your amount of orbitals