L5 - Silicate Structures
Earth Composition and Bonding
The Earth is primarily composed of Si, Mg, Fe, and O. In the crust, Al, Ca, K, and Na are also abundant. Understanding the bonding and atomic structures of these elements is crucial for comprehending the planet's function.
Silicon Tetrahedron
Si, as the most abundant non-oxygen element in the crust, exists as a ion. Its small size allows it to fit into the cavity formed by four oxygen anions. This forms a tetrahedron, which can bond with other tetrahedra, typically by joining corners, creating a bridging oxygen.
The bond is rigid, maintaining a fixed distance and angle. However, the arrangement can adopt various angles, leading to diverse structures beyond individual tetrahedra. The packing of these tetrahedra significantly influences a rock's properties.
An isolated tetrahedron carries a 4- charge. Bridging oxygens, while having a 2- charge, share it between two tetrahedra, reducing the overall charge of the repeating unit in the silicate crystal structure. Crystal neutrality is essential, so the types of ions incorporated depend on how the tetrahedra bond and affect the crystal's charge.
Silicate Structure Connectivity
The connectivity of tetrahedra dictates rock properties, and each connection type has a distinct name:
Isolated Tetrahedra (Orthosilicate)
Each tetrahedron is separate and not connected. Each isolated tetrahedron has a 4- charge: . The other ions in the structure must sum to a 4+ charge for net neutrality. Olivine is an example, incorporating or ions to achieve neutrality.

Single Chain Silicates (Inosilicate)
Each tetrahedron bonds to two others, forming a zig-zagging 1D chain. Each repeating unit contains 2 cations, 4 non-bonded oxygen anions, and 2 bonded oxygen anions. This results in a 4- charge: . Pyroxene is a single chain silicate that accommodates various cations to maintain neutrality.
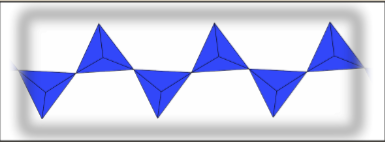
Double Chain Silicates (Inosilicate)
Half of the tetrahedra bond to three others, while the remaining half bond to two, resembling two joined single chains. The repeating unit consists of 4 cations, 5 bridging oxygens, and 6 non-bridging oxygens, resulting in a -6 charge: . Amphibole is a double chain silicate that can incorporate different cations to achieve a neutral charge.
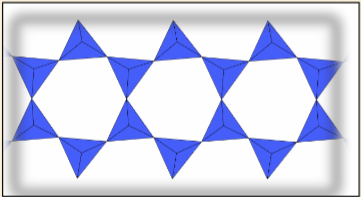
Sheet Silicates (Phyllosilicates)
Each tetrahedron bonds to three others, creating a 2D network. The repeating unit contains 2 ions, 3 bridging oxygens, and 2 non-bridging oxygens, leading to a -2 charge: . Mica and various clay minerals are examples.
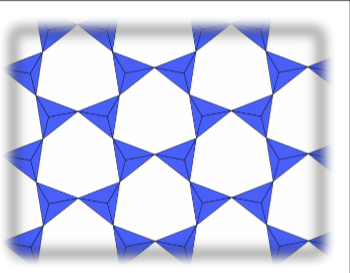
3D Frameworks (Tectosilicate)
Each tetrahedron shares all four oxygens with other tetrahedra. This results in one and two bridging oxygens, giving a net charge of 0. Therefore, no additional cations are needed for charge neutrality. Pure in a conjoined framework is quartz. Feldspars are also 3D framework silicates where some is replaced by , resulting in a net negative charge balanced by cations like and .

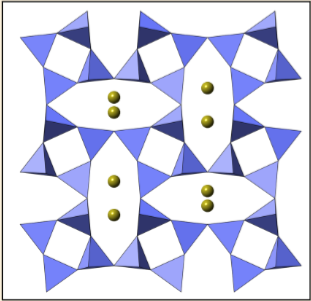
Cations in Silicate Structures
Cations such as , , , , and are larger than and fit into voids created by oxygen packing.
and fit into octahedra formed by 6 oxygen atoms.
and can fit in cubes created by 8 oxygens.
prefers fitting into cuboctahedra formed by 12 oxygens.
These polyhedra and their packing arrangement offer insights into crystal structures at a fundamental scale.
Olivine Example
Olivine's crystal structure contains two distinct distorted octahedra, M1 and M2 sites. These sites can accommodate elements with similar size and charge, maintaining charge neutrality and bond distances close to equilibrium. The similarity in size and charge between and allows both to occupy these octahedra, leading to the olivine solid solution.
Binary Phase Diagrams
A phase diagram illustrates stable phases at different combinations of parameters, typically composition and temperature, but also pressure. A binary phase diagram depicts composition variations between two end-members, such as fayalite () and forsterite ().
At high temperatures, most systems exist as a melt, accommodating a wide range of compositions. As temperature decreases, solids form, with the formation temperature depending on the melt's composition. Usually the solidification occurs over a range of temperatures.
The upper boundary between the melt and co-existing region is the liquidus. The lower boundary between the co-existing region and the solid is the solidus. When a melt cools to the liquidus, a solid forms with a composition indicated by the solidus at that temperature. As the solid forms, it removes elements from the melt, changing the melt's composition. Further cooling is needed for more solid to form.
The initial solid then reacts with the melt to adjust its composition to the new solidus value if cooled at equilibrium.
The solid's composition follows the solidus as the system cools. When the solidus composition matches the melt, complete solidification occurs, resulting in a solid with the original melt's composition. Olivine solid solution exemplifies this concept, but it extends to numerous systems exhibiting solid solutions where the melt composition favors solid solution formation.
Major, Minor, and Trace Elements
The elements in a mineral's formula (e.g., , , , and in olivine) are major elements. Ion compatibility in a crystal structure depends on size and charge, allowing minor or trace elements to substitute without significant energy penalty if they share similar charge and size. For example, , , , and can substitute into olivine's M1 and M2 sites. The suitability of minor or trace elements depends on the mineral's crystal structure (e.g., does not easily substitute into olivine but could in a mineral with a metal site).