Ap Biology
Unit 1
1.1
Oxygen is more electronegative compared to hydrogen, resulting in an uneven distribution of electrons between oxygen and hydrogen.
Covalent bonding can result in polarity.
Polarity = when there are differences in atomic electronegativity
Important things about hydrogen bonds
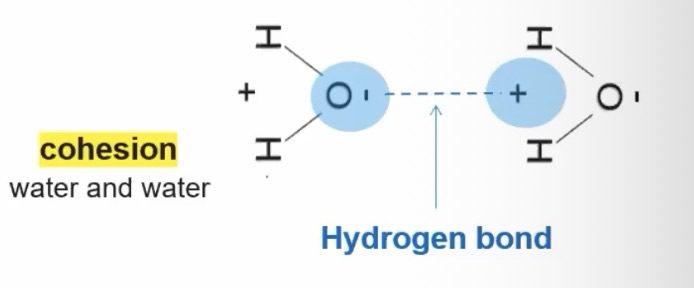
A hydrogen bond is a weak bond interaction between the negative and positive religion of two separate molecules
Water can form hydrogen bonds with other water molecules or any other charged molecule.
Cohesion = When two of the same molecules form hydrogen bonds with each other
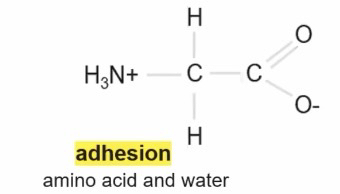
Adhesion = When two different molecules form hydrogen bonds with each other
Hydrogen bonds between water molecules can result in surface tension
Surface tension =
Result of increased hydrogen bonding forces between water molecules and the surface
Property allowing liquid to resist external forces
Properties of Water
Water’s adhesive property gives water a high solvency ability in its liquid state
Water’s cohesive property allows for unique hydrogen bond interactions to occur when water is in a solid state making ice less dense than liquid water
High Heat Capacity =
Water’s cohesive property allows it to absorb a lot of thermal energy before changing chemical states, resisting sudden changes in temperature
High specific heat = How much energy is needed to raise the temperature for a sample of a substance
Water resists changes in temperature
Water can absorb a lot of heat
Capillary action is the result of high adhesive and cohesive properties of water
Ex. Water using cohesion to stick together and then adhesion to stick to the tree and climb up
1.2
The law of conservation: States that energy cannot be created or destroyed only transformed
Living systems follow the laws of energy
Living system need a constant input of energy to grow, reproduce, and maintain organization
Living systems mainly use the energy stored in chemical bonds
Atoms and molecules from the environment are necessary to build new molecules
Most essential elements:
Carbon is used to build biological molecules such as carbohydrates, proteins, nucleic acids and lipids
Nitrogen is used to build proteins and nucleic acids
• Phosphorus is used to build nucleic acids and certain lipids
Carbon
Can bond with other carbon atoms creating carbon skeletons to which other atoms can attach themselves to
Carbon skeletons allow for the creation of large and complex molecules
Carbon containing molecules can be used to store energy and to form basic cell structures
1.3
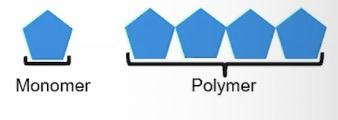
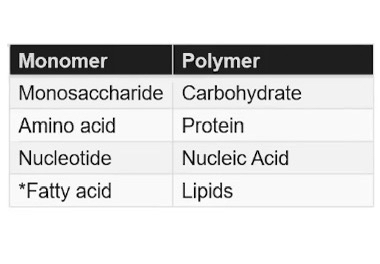
Monomers are chemical subunits used to create polymers
Polymer is a macromolecule made up of many monomers
Covalent bonds link monomers to form complex carbohydrate polymers
Monomers have specific chemical properties that allow them to interact with one another
Specific monomers are used to build specific polymers
***Lipids don’t have true monomers***
Dehydration synthesis reactions are used to create macromolecules
Hydrolysis is used to break down macromolecules back to monomers
Steps to dehydration synthesis
Two monomers get close together
One monomer loses a hydrogen atom, and the other loses a hydroxyl group OH.
Those pieces combine to make water H2OH2O, which is released.
A new covalent bond forms between the monomers, creating a larger molecule or polymer.
Steps to hydrolysis
Water is added the polymer to break polymer into monomers
-Covalent bonds are broken
A water molecule is hydrolyzed into OH and H and each subcomponent is added into a different monomer
A dehydration synthesis creates carbohydrates
Carbohydrates
Comprise of linear chains of sugar monomers connected by covalent bonds
Small direction change in the components of a molecule can result in functional differences
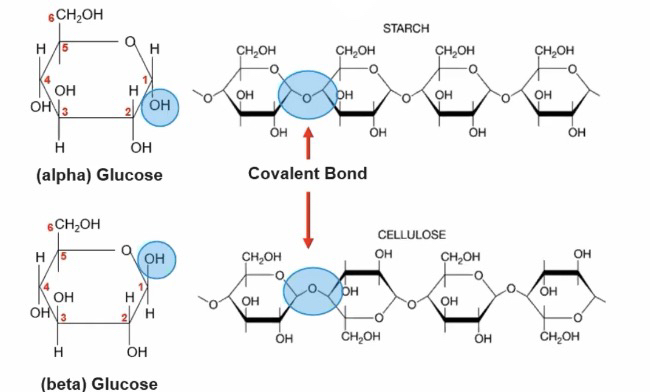
Monosaccharides: one sugar subunit
Polysaccharides: two or more sugar subunits
-Can be linear or branched
Lipids
Definition: Lipids are typically non-polar, hydrophobic molecules whose structure and function are derived from the way their subcomponents are assembled
Saturated fatty acids: Contain only single bonds between carbon atoms
Unsaturated fatty acids: Contain at least one double bond between carbon atoms.
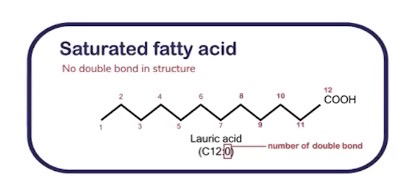
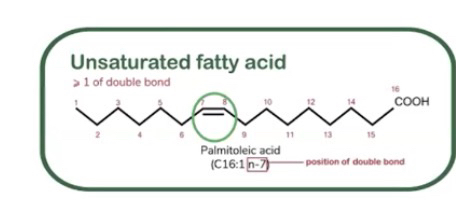
Different types of lipids
Fats: provide energy storage and support cell function. Can provide insulation to keep mammals warm
Steroids: hormones that support physiological functions including growth and development
Ex. Cholesterol which is essential to structural stability to animal cell membranes
Phospholipids: group together to form the lipid bilayers in plasma and cell membranes
Hydrophilic head: hate fats love water
Hydrophobic tail: hate water love fats
Nucleic Acids
What are the similarities and differences between the nucleic acid polymers; DNA and RNA?
In common: Both are assembled from nucleotide subunits which are comprised of
5 Carbon sugar
Phosphate group
Nitrogenous base
-Each nucleotide monomer is connected by covalent bonds forming the sugar phosphate backbone
-Each linear strand of nucleotides has a 5’ end and a 3’ end
The nitrogenous bases are perpendicular to the sugar phosphate backbone
Differences:
DNA contains deoxyribose
RNA constrains ribose
DNA contains thymine and RNA contains uracil
DNA is squally double stranded
RNA is usually single stranded
The two DNA strands in double stranded DNA is anti parallel
The linear sequence of all nucleic acids are characterized by a 3’ hydroxyl and a 5’ phosphate
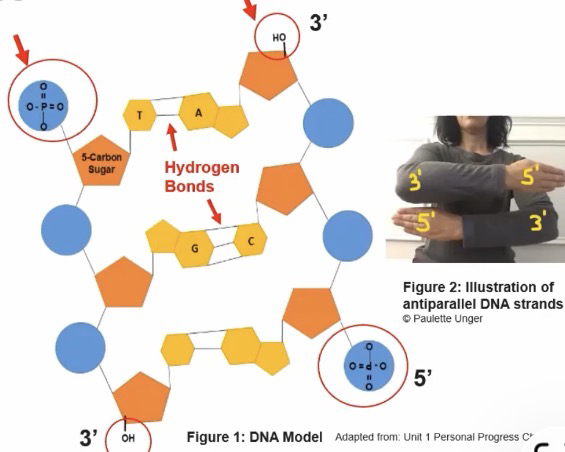
Adenine - Thymine base pairs are held together by 2 hydrogen bonds and Guanine - Cytosine base pairs are held together by 3 hydrogen bonds
Hydrogen bonds between base pairs in a DNA molecule stabilize the molecule's structure (More bonds = more stable)
During synthesis of the nucleic acid polymers, nucleotides can only be added to the 3’ end which forms covalent bonds
Proteins
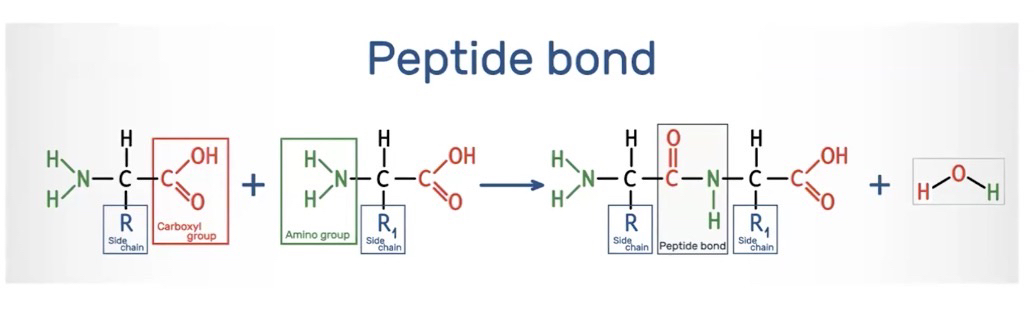
Comprise of linear chains of amino acids that have directionality with an amino terminus and a carboxyl terminus
Amino acids are connected by the formation of covalent bonds at the carboxyl terminus of the growing peptide chain
A change in an amino acid subunit at the primary level of structure may lead to a change in the structure and function of the protein at subsequent levels.
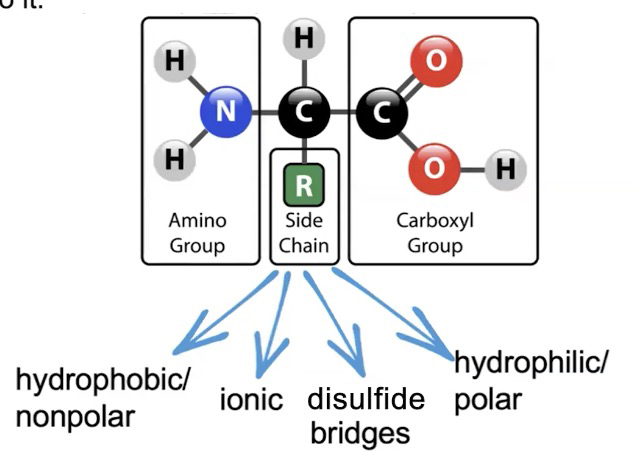
Amino acids: composed of a central carbon atom with a hydrogen atom, a carboxyl group, an amine group, and a variable R group covalently bound to it.
1.7
Proteins comprise linear chains of amino acids connected by the formation of covalent (peptide) bonds that form between a carboxyl group (-COOH) of one amino acid and an amine group (- NH,) of the next amino acid, resulting in a growing peptide chain.
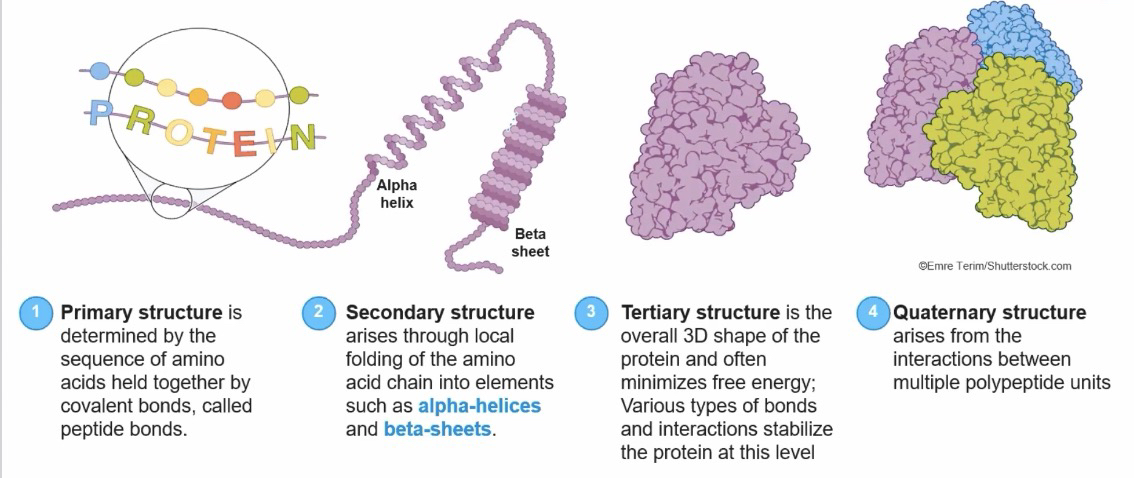
The sequence of amino acids determines a protein's primary structure and overall shape.
Secondary structures, such as alpha-helices and beta-pleated sheets, form through hydrogen bonding.
Tertiary structure results from various interactions, including hydrogen bonds, hydrophobic interactions, lonic interactions, and disultide bridges.
Quaternary structure forms when multiple polypeptides interact, and all four levels of structure determine a protein's function.
Unit 2
2.1 Eukaryotic cells
• All living cells contain a genome and ribosomes, reflecting the common ancestry of all known life.
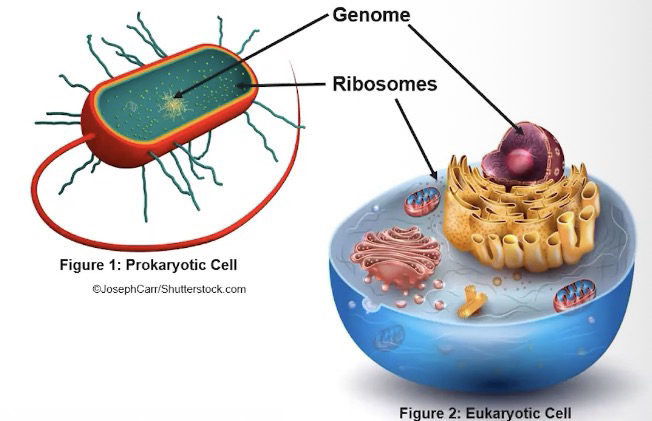
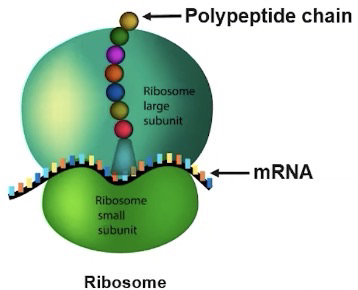
Ribosomes
They synthesize protein according to mRNA sequence and the instructions that are encoded in that mRNA sequence originate from the genome of the cell.
Ribosomes consist of two subunits that are NOT membrane-enclosed
Ribosomes are made of ribosomal RNA (rRNA) and proteins
Ribosomes synthesize protein according to mRNA sequences
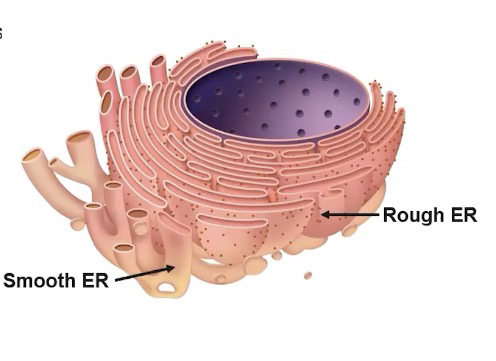
Endoplasmic Reticulum (ER)
The endoplasmic reticulum is a network of membrane tubes within the cytoplasm of eukaryotic cells
• The ER performs the following functions for the cell:
Provides mechanical support
Plays a role in intracellular transport
Rough ER carries out protein synthesis on ribosomes that are bound to its membrane
-Two forms of the ER
1. Rough ER
Has ribosomes attached to its membrane
Compartmentalizes the cell
rough ER is associated with packaging the newly synthesized proteins made by attached ribosomes for possible export from the cell
2. Smooth ER
Does NOT have ribosomes attached
Functions include detoxification and lipid synthesis
Structural differences between rough ER and smooth ER
leads to functional differences
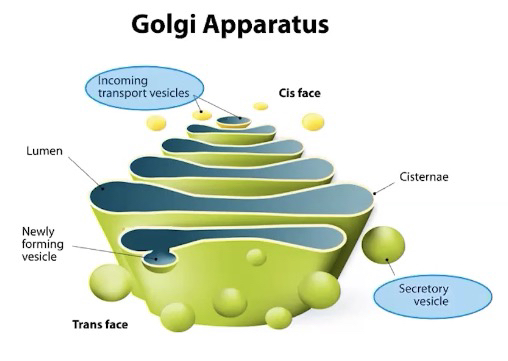
Golgi Apparatus
• Series of flattened membrane-bound sacs found in eukaryotic cells
• Involved in the correct folding and chemical modification of newly synthesized proteins and packaging for protein trafficking
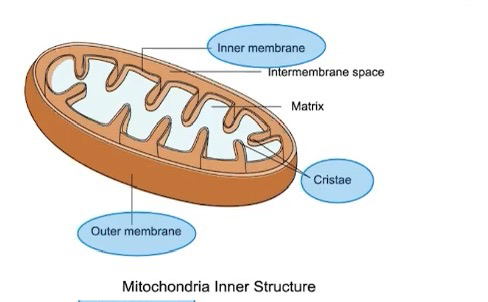
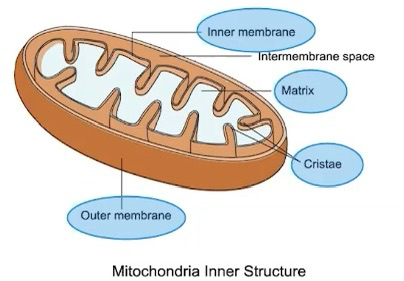
Mitochondria
Has a double membrane
Outer membrane is smooth and inner membrane is highly convoluted, forming folds called cristae
Functions in production of ATP energy that eukaryotic cells can use for cell work.
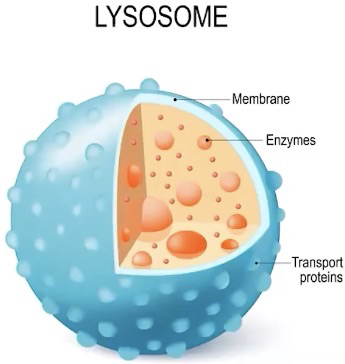
Lysosomes
Membrane enclosed sacs found in some eukaryotic cells that contain hydrolytic enzymes
Hydrolytic enzymes can be used to digest a variety of materials such as damaged cell parts or macromolecules
Ways it can contribute to cell function:
Intracellular digestion
Recycling of organic materials
Programmed cell death (apoptosis)
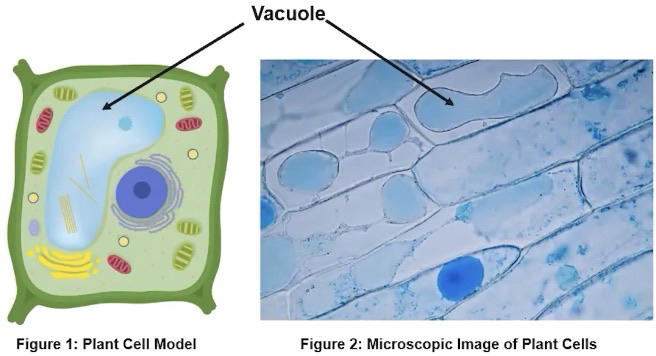
Vacuoles
Membrane-bound sacs found in eukaryotic cells
Play variety of roles ranging from storage of water and other macromolecules to the release of waste from a cell
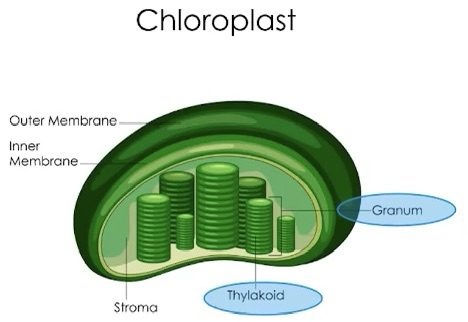
Chloroplasts
Found in eukaryotic cells such, as photosynthetic algae and plants
Double outer membrane
Specialized for capturing energy from the sun and producing sugar for the organism
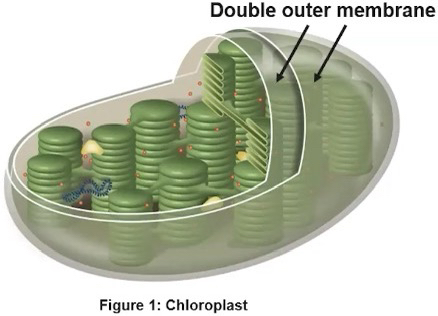
Chloroplasts
Specialized for photosynthesis and capturing energy from the sun to produce sugar
Within chloroplasts are distinct compartments:
1. Thylakoid
Highly folded membrane compartments that are organized in stacks called grana
Membranes contain chlorophyll pigments that comprise the photosystems and electron transport proteins can be found between the photosystems, embedded in the thylakoid membrane
Light-dependent reactions occur here
• Folding of these internal membranes increases the efficiency of these reactions
2. Stroma
Fluid between the inner chloroplast membrane and outside thylakoids
The carbon fixation (Calvin-Benson cycle) reactions occur here
Mitochondria
Double membrane provides compartments for different metabolic reactions
Mitochondria capture energy from macromolecules
The Krebs cycle (citric acid cycle) reactions occur in the matrix of the mitochondria
Electron transport and ATP synthesis occur in the inner mitochondrial membrane
Folding of the inner membrane increases the surface area, which allows for more ATP to be made
Vacuoles
Vacuoles play a variety of roles, including storage and release of water, macromolecules, and cellular waste products.
In plants, vacuoles aid in retention of water for turgor pressure
Turgor pressure is an internal cellular force, usually caused by water pushing up against the plasma membrane and cell wall
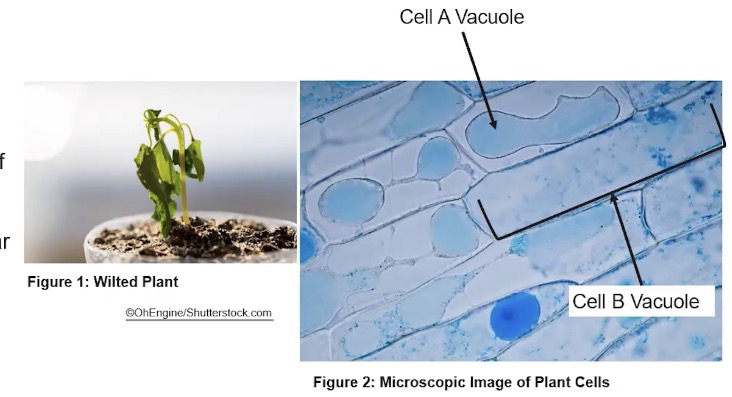
Cell A= plant’s wilted
Cell B= plant’s not wilted
2.2
Effect of Surface Area-to-Volume
Ratios on the Exchange of Materials
• Smaller cells typically have a higher surface area-to-volume ratio and more efficient exchange of materials with the environment
As cells increase in volume, the relative surface area decreases making it difficult for larger cells to meet the demand for internal resources and remove waste sufficiently
These limitations can restrict cell size and shape.
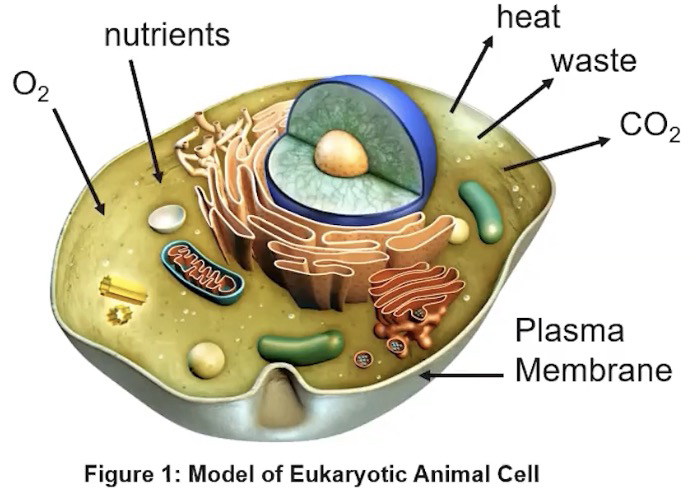
As cell volume increases or a cell become specialized for transport across its surface, structural modifications such as membrane folds are necessary to adequately exchange molecules from or into the environment.
As organisms increase in size, their surface area-to-volume ratio decreases making it harder to release heat energy and adaptations may improve an organism's efficiency in doing so.
Cells and organisms use specialized exchange surfaces like stomatal openings on the surface of a leaf
2.3
Cell membranes provide a boundary between the interior of the cell and the outside environment
-Control the transport of materials in and out of the cell
What are cell membranes made up of? Phospholipids.
-They’re amphipathic
Hydrophilic phosphate head is polar
Hydrophobic fatty acid tail is non-polar
Phospholipids spontaneously form a bi-layer in an aqueous environment
Tails are located inside the bilayer
Heads are exposed to the aqueous outside and aqueous inside environments
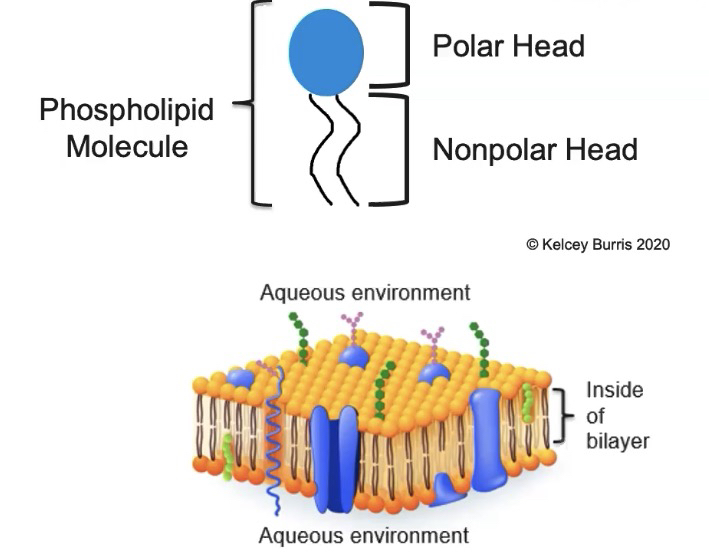
There are embedded proteins in the cell membrane
-Peripheral proteins
Loosely bound to the surface of the membrane
Hydrophilic with charged and polar side groups
-Integral proteins
Span the membrane
Hydrophilic with charged and polar side groups
Hydrophobic with non-polar
side groups penetrate hydrophobic interior of bilayer
Ex. Transmembrane proteins
Embedded proteins have various roles
Transport
Cell to cell recognition
Enzymatic activity
Signal transduction
Intercellular joining
Attachment for extracellular matrix or cytoskeleton
The framework of the cell membranes are described as the Fluid Mosaic Model
-Structured as a mosaic of protein molecules in a fluid bilayer of phospholipids
-The structure is not static and is held together by hydrophobic interactions (weaker than covalent bonds)
-Most lipids and some proteins can shift and flow along the surface of the membrane or across the bilayer
F.M.M. include steroids and carbohydrates
-Cholesterol is randomly distributed and wedged between phospholipids in the cell membrane of eukaryotic cells
-Regulates bilayer fluidity under different environmental conditions
-Diversity and location of carbohydrates and lipids enable them to function as markers
-Glycoproteins: one of more carbohydrate attached to a membrane protein
Glycol-lipids: lipid with one more more carbohydrate attached to it
Membrane permeability
The cell membrane is selectively permeable
-Small nonpolar molecules pass freely
Ex. N2, O2, CO2
Hydrophobic substances such as large polar molecules and ions can NOT freely move across the membrane
Hydrophilic substances move through transport proteins
Channel Proteins - A hydrophilic tunnel spanning the membrane that allow specific target molecules to pass through
Carrier Proteins - Spans the membrane and change shape to move a target molecule from one side of the membrane to the other
Small polar molecules, like H2O, can pass directly through the membrane in minimal amounts
The cell wall is a structural boundary and permeable barrier
As a structural boundary:
Protects and maintains the shape of the cell
Prevents against cellular rupture when internal water pressure is high
Helps plants stand up against the force of gravity
As a permeable barrier
Plasmodesmata - small holes between plant cells that allows the transfer of nutrients, waste, and ions
Animal cells do not have cell walls
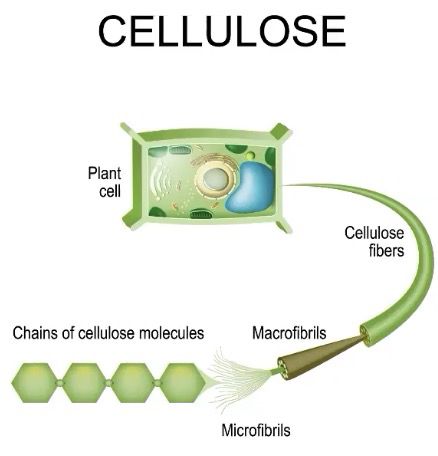
Cell Wall - comprised of complex carbohydrates
Plants - Cellulose
Polysaccharide
Fungi - Chitin
Polysaccharide
Prokaryotes - peptidoglycan
Polymer consisting of sugar and amino acids
2.5
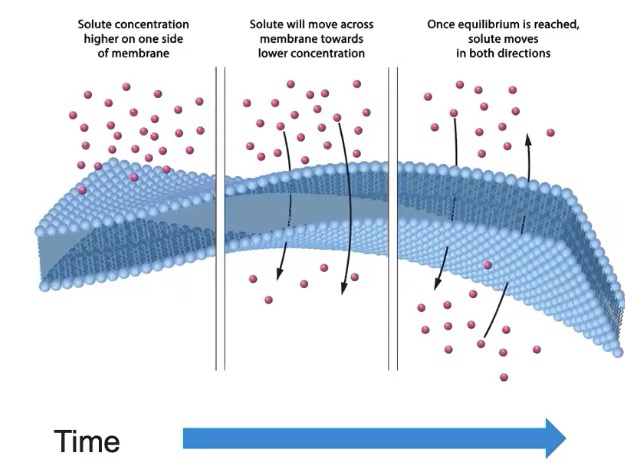
Selectively permeable membranes allow for the formation of concentration gradients.
Concentration gradient
A concentration gradient is when a solute is more concentrated in one area than another.
A membrane separates two different concentrations of molecules
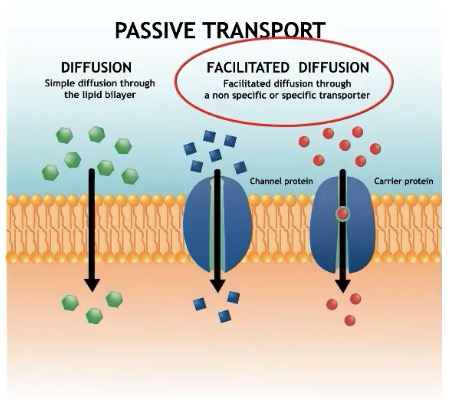
Passive transport ( High to low concentration)
-Net movement of molecules from high to low concentration without ATP
• Plays a primary role in the import of materials and the export of wastes
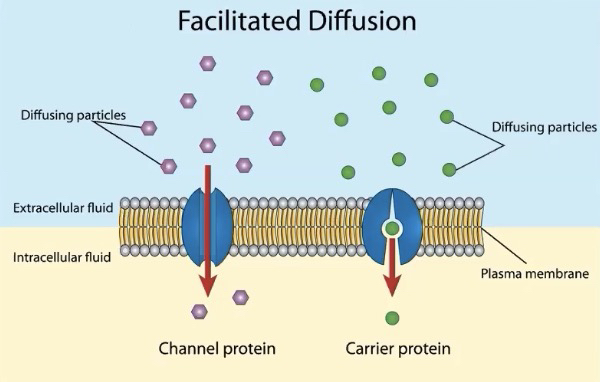
Two types of passive transport:
Diffusion - movement of molecules from high concentration to low concentration
Small nonpolar molecules pass freely (N2, O2, CO2)
Facilitated Diffusion - movement of molecules from high concentration to low concentration through transport proteins
Allows for hydrophilic molecules and ions to pass through membranes
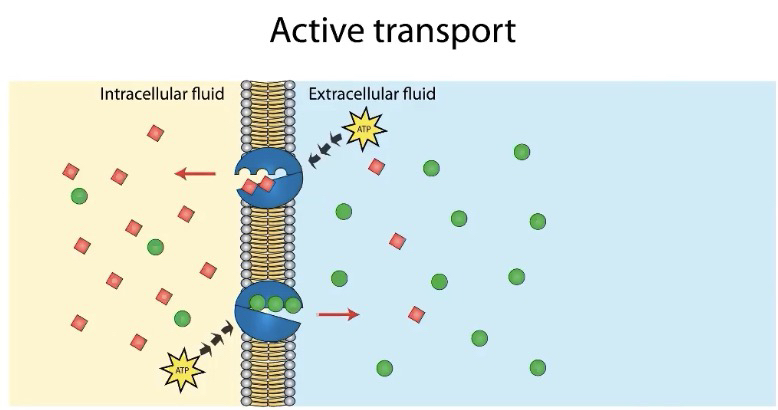
Active Transport (Low to high concentration)
Active transport requires energy.
• Active transport requires the direct input of energy (such as ATP) to move molecules from regions of low concentration to regions of high concentration.
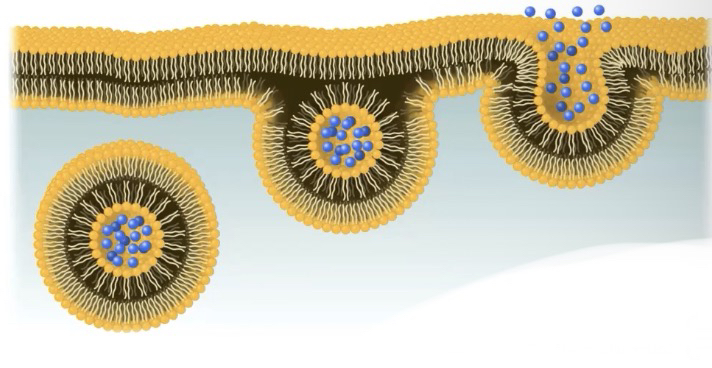
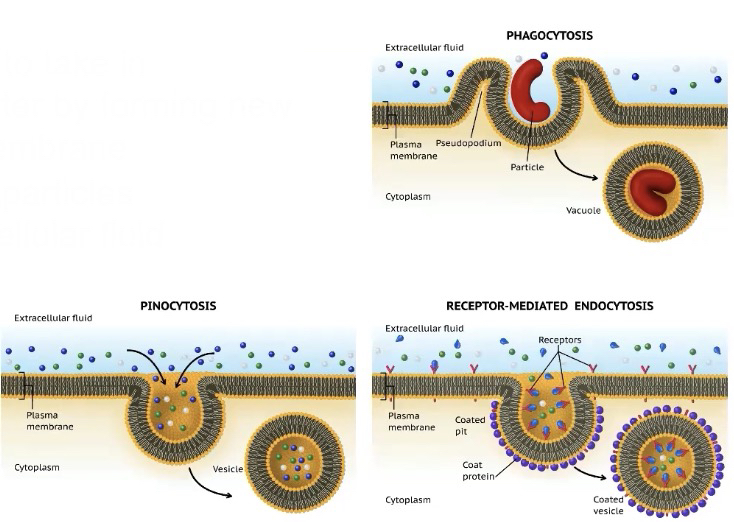
In endocytosis
the cell uses energy to take in macromolecules and particulate matter by forming new vesicles derived from the plasma membrane
Phagocytosis - cell takes in large particles
Pinocytosis - cell takes in extracellular fluid containing dissolved substances
Receptor-Mediated endocytosis - receptor proteins on the cell membrane are used to capture specific target molecules
In exocytosis
internal vesicles use energy to fuse with the plasma membrane and secrete large macromolecules out of the cell
Proteins such as signaling proteins
Hormones
Waste