Principles of Chemistry IGCSE Edexcel (States of Matter Pt. 1.1-1.4)
Chemistry IGCSE- Principles of Chemistry
}}1.1 States of Matter}}
The three States of Matter
The three states are solids, liquids and gases
The state changes occur at the meting point and boiling point
| Solid | Liquid | Gas |
|---|---|---|
| Regular arrangement | Randomly arranged | Randomly arranged |
| Vibrate around a fixed position | Move around eachother | Move quickly in all directions |
\
Changes of state
- The amount of energy needed for interconversion of state depends on the strength of forces between particles —>
This is a physical change involving changes in forces between the particles of the substances
\
Evaporation vs Boiling:
Both happen when a liquid changes into a gas
%%Evaporation%% only occurs at the surface of liquids; where high energy particles can escape from liquids at low temperatures
Evaporation occurs over a range of temperatures
%%Boiling%% requires heat energy which transforms into kinetic energy
It only occurs at a specific temperature which is unique to each pure liquid
\
Melting:
Requires heat energy which transforms into kinetic energy, allowing particles to move
Occurs at a specific temperature that is unique to each pure solid
\
Freezing:
Is the reverse of melting and happens at the exact same temperature as the melting point
\
Condensation:
When a gas is cooled, particles lose energy so when they bump together, they lack the energy to bounce away and instead group together to form a liquid
\
Diffusion and Dilution:
Diffusion and Dilution experiments support a theory that all matter (solids, liquids and gases) is made up of tiny, moving particles; the kinetic theory of matter
Diffusion-
Movement of particles from an area of high concentration to an area of low concentration
Therefore, particles must be able to move freely (in fact, diffusion cannot occur in solids)
\
Solutions:
| Solvent | Liquid in which solute dissolves |
|---|---|
| Solute | Substance that dissolves in a liquid to form a solution |
| Saturated solution | A solution with the maximum concentration of solute dissolved in the solvent |
}}1.2 Elements, compounds and mixtures}}
Pure substances and mixtures:
Element:
A substance made up from only one type of atom (eg. Sodium)
Compound:
A substance made from two or more elements that are chemically bonded together (eg. Carbon Dioxide)
Mixture:
Two or more elements or compounds physically combined together (eg. Salty water)
Chemical properties of each substance are not altered
\
Separation and Purification Techniques
Simple distillation:
Separating a %%solvent from a solution%% (eg. Water from salty water)
\
Fractional distillation:
Separating a liquid from a %%mixture of liquids%% (eg. Ethanol from ethanol and water or fractions from crude oil)
Works as liquids have different boiling points so, when heated, liquid with lower boiling point will evaporate first, condense and be collected.
\
Paper chromatography:
Separating %%mixtures of soluble substances%% (eg. Food colourings, inks, dye)
Used to help identify substances
Separation depends of the solubility of substance
\
Practical- Investigating the compositions of inks using chromatography
Method:
Draw a start line in pencil across the chromatography paper (approx 1-2 cm from bottom)
Use a pipette to add small dots of ink across the line and label them
Place the paper into a container with water (solvent) reaching just underneath the start line
Allow the solvent to move through the paper, remove the chromatogram before it reaches the top
Draw a line in pencil marking the solvent front
Analysis and results:
- Measure distance travelled by each spot and record in a table
- Calculate Rf value
\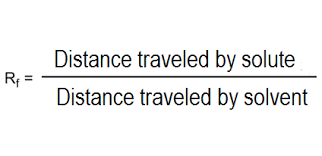
}}1.3 Atomic Structure}}
\
- All substances are made of atoms
- An atom is made up of protons, neutrons and electrons
| Subatomic Particle | Relative mass | Relative charge | Position |
|---|---|---|---|
| Proton | 1 | +1 | Nucleus |
| Neutron | 1 | 0 | Nucleus |
| Electron | Almost 0 | -1 | Shells |
\
Atomic and Mass number
Atomic number:
- Number of protons in atom’s nucleus
- Unique for every element
\
Mass number:
- Total number of protons and neutrons in atom’s nucleus
- Usually different but can be the safe across different elements
\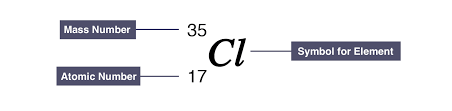
Isotopes
- Isotopes are atoms of an element that have the %%same number of protons%%, but %%different number of neutrons%%
\
Relative atomic mass
It is the weighted average of the relative atomic masses of the isotopes in that element
Their symbol is Ar
\
Calculating Ar
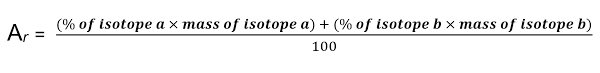
\
\
\
\