Atomic Structure
ATOMIC MODEL & THEORY TIMELINE
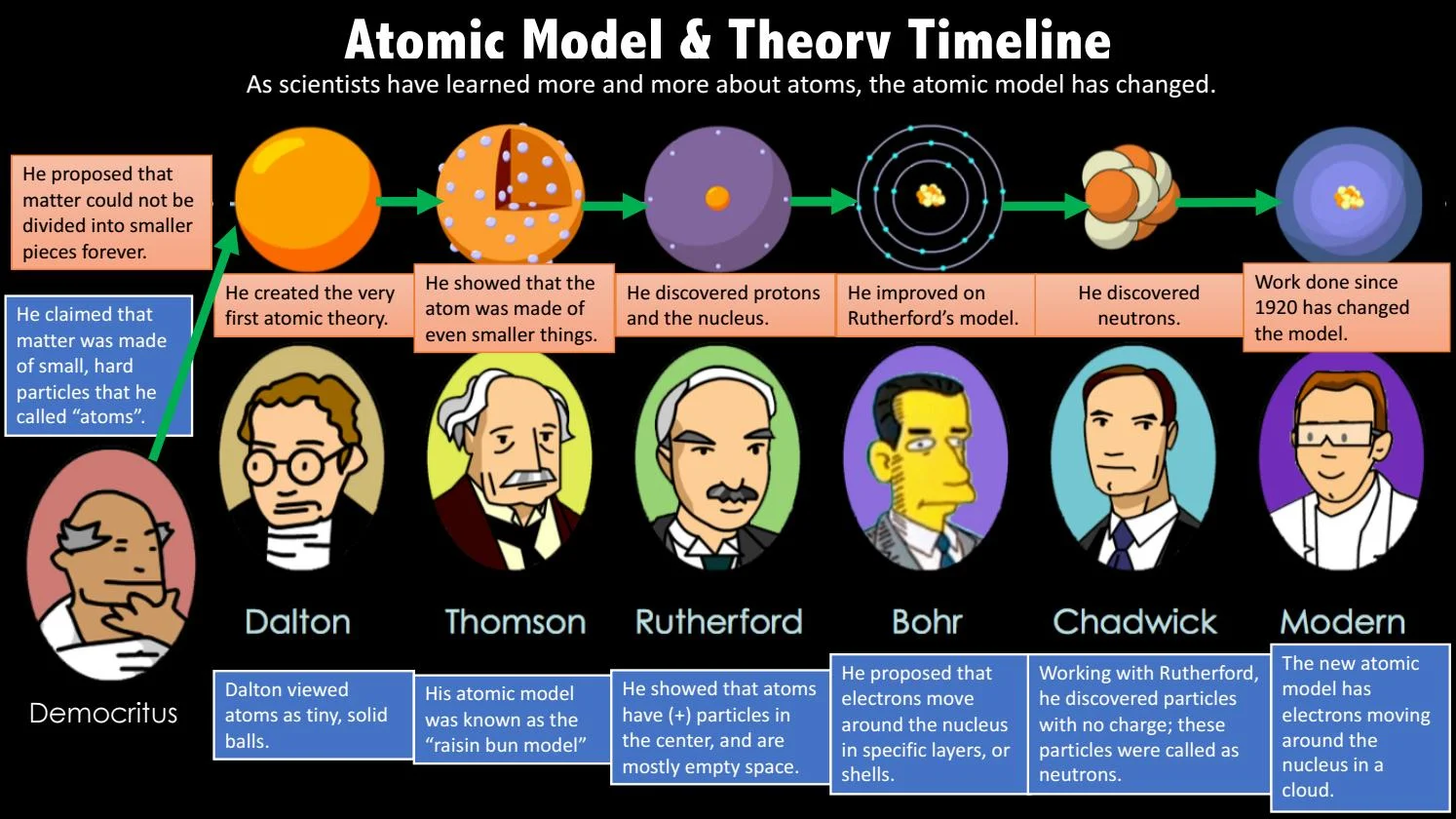
Democritus
-Atomos
-Led to the Idea “atoms in a void”
Aristotle
-Matter was made of 4 “elements”
John Dalton
-Thought of the atom as a solid billiard balls
-Proposed a modern atoms model based on experimentation
-1st scientist to have a theory about matter being composed of atoms & how atoms might look & behave
Law of Conservation of Mass
-During chemical reactions [like burning], atoms are neither created nor destroyed.
-The number of atoms remains constant throughout the reaction, thus the mass also remains the same
Law of Definite Proportions
-In a given compound, the element’s components are in a fixed ratio
-Atoms are neither lost nor gained in a chemical reaction.
!!TAKE NOTE!!
-Each element consists of a different number of protons, electrons, and neutrons
-Compounds cannot be made without electrons.
-Only electrons can be transferred or shared
-the more neutrons added, the heavier it becomes
Atom
-An atom is defined as the smallest particle of a substance that can exist or by itself or be combined with other atoms to form a molecule
JJ Thompson
-Discovered electrons through the discovery of the cathode rays
-electrons are negatively charged particles
-1st scientist to show that the atom was made of even smaller things.
-Plum pudding
-Atom is a positively charged sphere with loosely embedded electrons in it
James Chadwick
-working with JJ he discovered the neutrons
-Neutrons are also found in the nucleus
Neils Bohr
-Electrons move around the nucleus in specific layers
-composed of (e) (p) (n)
Atomic number
-The number of protons in an atom of an element
-# of protons = Number of Electrons (APE)
Mass number
-The total number of protons and neutrons in an atom
-Mass Number - Atomic Number = Number of neutrons
Isotopes
-atoms of the same element that have different numbers of neutrons called isotopes
-also commonly used in medical imaging
-sometimes written without atomic number
-The atomic number of an element doesn’t change but it’s number of neutrons can change
Relative Abundance
-The percentage of atoms with a specific atom mass found in a naturally occurring sample of an element
ATOMIC PROPERTIES
Radioactivity decay
-The emission of ionizing radiation from nuclear decay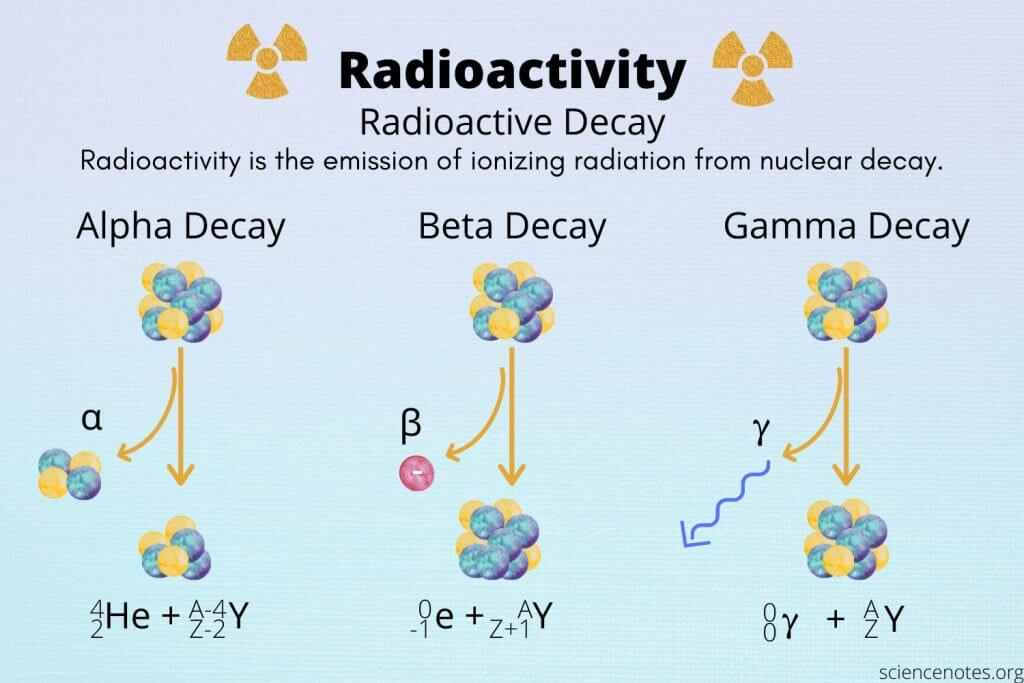
Ions
-A neutral atom becomes an ion either by losing an electron or by gaining an electron
Bohr’s Atomic Theory
-Electrons can exist only in certain circular orbits (energy levels)
-the energy of the electron in a given orbit is fixed or quantisized
-An electron can jump from one energy to another
-when it absorbs an energy it jumps from a lower to a higher energy level
-When it jumps from highher to lower energy level it emits energy
States of Electrons
-When currents is passed through a gas at a low pressure the potential energy (Energy due to position of some of the gas atoms increases)
Ground state
-the lowest energy of an atom
Exited State
-A state where the atom has a higher potential energy than it had in its ground state
Neon signs
-When an excited atom returns to its ground state as it give off the energy it gained in the form of electromagnetic radiation
Electron Cloud Model
-Electrons are most likely located in the electron cloud- a visual model that represents all orbitals in the atom
-it consisted of a dense nucleus surrounded by a cloud of electrons a various levels in the atom
-Atomic orbitals are reigons of space around the nucleus of an atom where an electron is likely to be found
Pauli Exclusion Principle
-Orbital can hold a max of 2 electrons
-to occupy the same orbital 2 electrons must spin in opposite directions
Hunds Rule
-electrons occupy equal-energy orbitals so that a maximum of unpaired electrons results
Aufban Principle
-electrons are added at a time to the lowest energy orbitals available until all the electrons of the atoms have been accounted for
Electron configuration
-the probable distribution of electrons around the nucleus of an atom