Lab 5: Osmolarity
Definitions:
Plasma Membrane:
The protective barrier surrounding the contents of a cell, composed of a phospholipid bilayer that regulates the entry and exit of substances. It is selectively permeable, allowing specific molecules to pass while blocking others.
Osmosis: → know definition
The movement of water molecules through a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration, aiming to achieve equilibrium in solute concentrations on both sides of the membrane.
water go in direction of more concentrated solute
Aquaporins:
Specialized protein channels embedded in the plasma membrane that facilitate the rapid transport of water molecules
Tonicity: The term that describes the osmotic pressure gradient of two solutions separated by a semipermeable membrane, influencing water movement.
Isotonic: A solution that has the same solute concentration as another solution, resulting in no net movement of water across the membrane.
Hypotonic: A solution with a lower solute concentration than another solution, leading to water moving into cells, potentially causing them to swell and burst (hemolysis).
Hypertonic: A solution with a higher solute concentration than another solution, causing water to move out of cells, leading to cell shrinkage (crenation).
Osmolality:
The measure of solute concentration in a solution, expressed as osmoles of solute per kilogram of solvent, important in understanding the direction and extent of osmotic movement.
Osmoregulation:
The biological process whereby an organism maintains optimal water and solute concentration within its internal environment, crucial for homeostasis and proper cell function.
Osmoregulators:
Organisms that actively regulate their internal osmotic environment, maintaining a stable internal pressure regardless of external conditions (e.g., mammals, most freshwater fish).
Osmoconformers:
Organisms that allow their internal osmotic pressure to match that of their surrounding environment, common in many marine animals like jellyfish and some invertebrates.
Metanephridia:
A type of excretory structure found in certain invertebrates (such as annelids), consisting of tubules that play a role in osmoregulation and the expulsion of metabolic waste.
Hemolysis:
The process in which red blood cells burst due to the influx of water in a hypotonic environment, resulting in the release of hemoglobin into the surrounding fluid.
Crenation:
The shriveling or shrinkage of a cell that occurs when it is placed in a hypertonic solution, where water leaves the cell, causing it to lose volume and appear wrinkled.
Overview:
Osmosis is of fundamental importance in animals and affects every cell in the body.
Osmoregulatory systems in animals play a critical role in maintaining extracellular fluid composition and internal body conditions to minimize osmosis-induced changes in cell volume.
Purple Box Questions:
Describe the function of aquaporins. Where in the human body are these
membrane proteins found?
Aquaporins are specialized protein cells that facilitate the rapid transport of water molecules across cell membranes. The proteins are embedded directly in the plasma membrane of cells and play a crucial role in maintaining osmotic balance for animal cells and bodies
These membrane proteins are found in all animals all over their bodies. There is a greater amount of them in areas like the kidneys, (specifically in renal tubules), red blood cells, eyes, brains and certain epithelial tissues.

• Why is it a bad idea for humans to drink sea water?
Humans should not drink salt water because our cells cannot handle the high amounts of salt. Excess salt concentrations will create a hypertonic solution of cells to extracellular fluids causing cells to shrink and shrivel.
Animals who can consume salt water often live in marine environments (fish) and have specialized structures that allow them to filter out excess salts. An example of this is the marine birds, such as seagulls, having salt glands. These glands extract excess sodium chloride (NaCl) from the blood and excrete it through nasal fluids

• Describe how osmoregulatory challenges differ between the marine,
freshwater and terrestrial environments for vertebrate animals.
Osmoregulartors are animals whose bodies work to actively maintain a constant steady internal environment that cannot be changed by changes in the external environment.
Each type of environment presents unique challenges for osmoregulators:
Marine environments (very salty): vertebrate animals lose water through osmosis due to the high salinity of the surrounding water, necessitating mechanisms to expel excess salts while retaining water. → animals develop salt glands
Freshwater environments: these animals face the opposite challenge, as they absorb too much water osmotically, which requires them to excrete large volumes of dilute urine while conserving salts. → gills
Terrestrial environments: animals must prevent dehydration from water loss due to evaporation, often leading to adaptations such as the development of impermeable skin or concentrated urine.
Procedure/ What happened:
Experiment 1 - placed 5 earthworms is 5 different concentrations of salt water to determine Percent of Initial Mass. The solutions were 10%, 20%, 40%, 60% and 1 unknown. The earth worms were weighed on a scale before and after submersion into the water to find their individual change in mass.
Percent of Initial Mass = (Final Mass / Initial Mass) x 100%
To find the concentration on the unknown solution, we had other make an equation on excel from the class data. In the equation, y was equal to Percent Initial Mass (which was known from the experiment) , m equal slope, x was percent/ concentration of seawater and b was the intercept
y = -0.2372x + 101.81
* on the XY scatter graph that was made, we did not include trial 5 worms results (unknown) because it had to be mathematically solved used the trend.
WHY =
Osmosis is the movement of water across a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration.
^ this process is used in osmoregulation to maintain extracellular fluid composition
Percent of Initial Mass is used to indicate the change in body mass and match it to the movement of water. If it is calculated to be above 100, that means weight was gained, but if were below 100, that means weight loss.
An earthworms internal body solute concentration is between 25%-40% (controlled by their metanephridia)
in hypotonic (lower) solution water is moving into a cell / worm → worm would increase in mass and size , and could potentially burst
^ In 10% and 20% treatments the worm would swell
in an isotonic (same) solution, water is moving in and out of the cell at an equal rate → worm should have a minimal / negligible change in weight
^ In 31.49% and 40% treatments worm would have negligible weight change
in a hypertonic (higher) solution water is moving out of a cell / worm → worm would shrink in mass and size
^In 60% treatment worm would shrink
Experiment 2 - using a microscope the diameter of ten sheep erythrocytes (blood cells) were measured under three different solution tonicities. Three drops of sheeps blood was added to a vial containing 2 unknown concentrations of NaCl, the third vial was left untouched with blood cells in natural plasma environment.
for each vial / treatment a blood slide was prepared and analyzed under a microscope at 40x objective. A special ruler, ocular micrometer, measured the diameter as ‘ticks’ before it was converted to micrometers.
Dashes were converted to micrometers = Dash size * 100/40
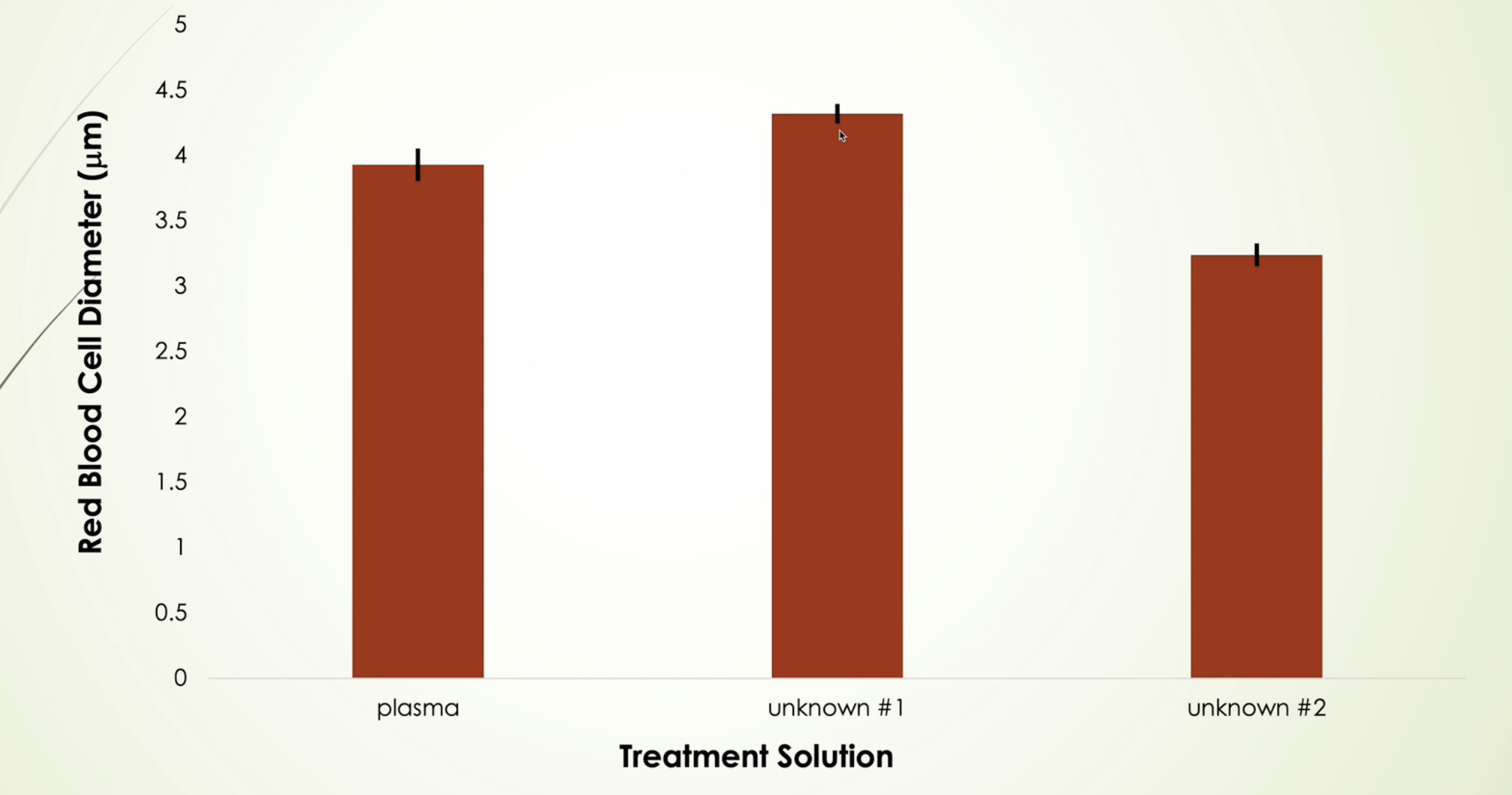
The upper confidence level is a maximum expected value . Taken for each vial
the lower confidence level is minimum expected value . This was taken for each vial
the values determined from these were used to make SEM bars on the class data of the column graph
WHY =
Blood plasma, blood’s non cellular fluid is isotonic, this means that there is no net movement of water into or out of the erythrocytes. NaCl is non permeating solute meaning it cannot diffuse across a plasma membrane, but water can still diffuse through the membrane; water could bring dissolve NaCl across plasma membrane
Under hypotonic (lower) conditions, the concentration of NaCl outside the cell is lower than inside, water will move into the erythrocytes, causing them to swell and potentially burst (lysis) . → this is seen as an increase in diameter and a more spherical shape of cell
Under hypertonic (higher) conditions, the concentration of NaCl inside the cell is higher than outside, water will move out of the erythrocytes, resulting in cell shrinkage and crenation. → this is seen as a decrease in diameter and a shrivelled spiky look
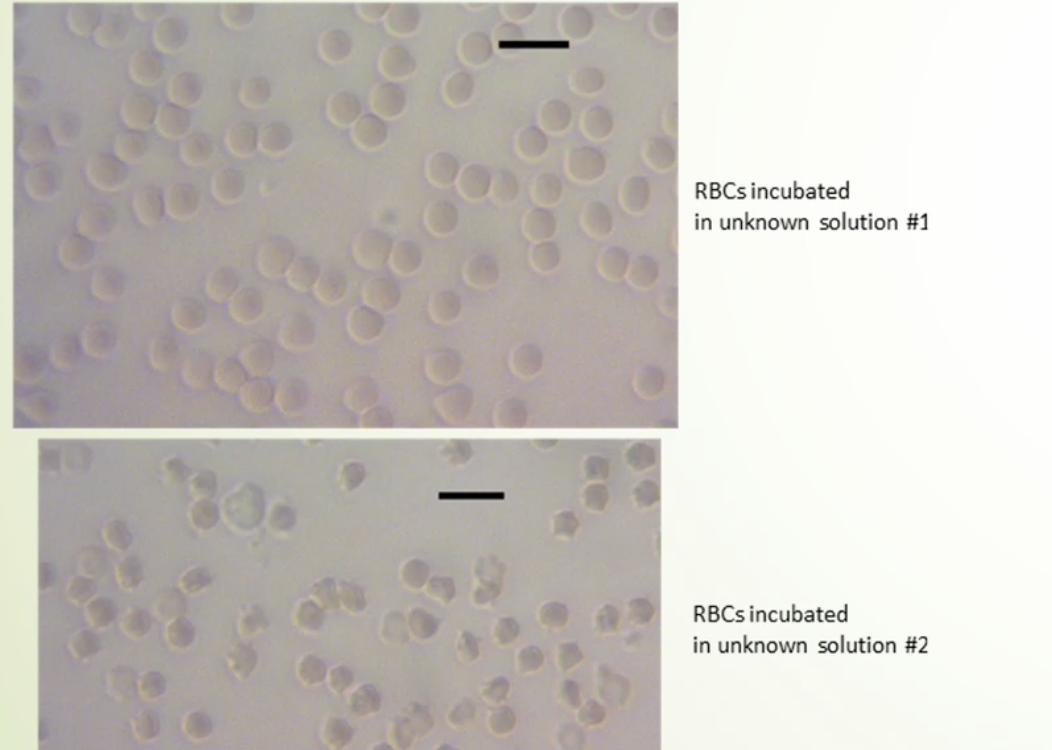
^ from this photo : #1 water moved in and cells swelled = hypotonic
#2 water moved out and cells shrunk = hypertonic
^ same results as photo above
SEM Bars:
If the error bars overlap , the treatments are likely not statistically significant
If the error bars do not overlap, the treatments are likely statistically significant
longer SEM bar means that there is more variation among results that the students provided, where as shorted SEM bars meant that there was less variation and many results were similar
Learning Outcomes:
Describe how animal cells are affected when osmolality of extracellular fluid varies.
→ When the osmolality of extracellular fluid increases, animal cells may undergo crenation due to water loss, while a decrease in osmolality can lead to cell swelling or lysis as water enters the cells.
Animal cells are affected by changes in extracellular fluids with changes in size and appearance. This can be concerning because if there is too drastic of a decrease on osmoalirty a cell burst, or commonly referred to as lysis. On the other end, if a cell has too drastic of an increase in osmolarity, it will undergo crenation and shrivel
note: that there are cellular components in RBC so it cannot completely shrivel up
Explain features of the plasma membrane in animal cells that underpin its role in osmosis.
→ The plasma membrane's selective permeability is crucial in regulating the movement of water and solutes, allowing specific ions and molecules to pass through while restricting others. This is facilitated by the presence of integral proteins, which function as channels and carriers, enabling water (via aquaporins) and essential nutrients to enter or exit the cell effectively. Additionally, the fluid mosaic model of the plasma membrane ensures that these proteins can adapt and change positions, supporting the dynamic interactions necessary for maintaining osmotic balance.
Explain the role of osmoregulatory organs in maintaining extracellular fluid
composition in vertebrates.
→ Osmoregulatory organs, such as the kidneys in vertebrates, play a key role in maintaining the composition of extracellular fluids by filtering blood, reabsorbing necessary substances, and excreting waste products and excess water. These organs adjust the concentration of solutes like sodium, potassium, and urea through processes such as filtration, secretion, and reabsorption, thereby helping to regulate osmotic pressure and maintain homeostasis within the body.
→ control concentrations of solute to maintain optimal cell function