2.2-2.9; 3.1
ALKYL GROUPS
Alkyl groups are derived from alkanes by removing one hydrogen atom, resulting in a substituent that can attach to a larger molecule.
Common examples include methyl (–CH₃), ethyl (–C₂H₅), and propyl (–C₃H₇) groups.
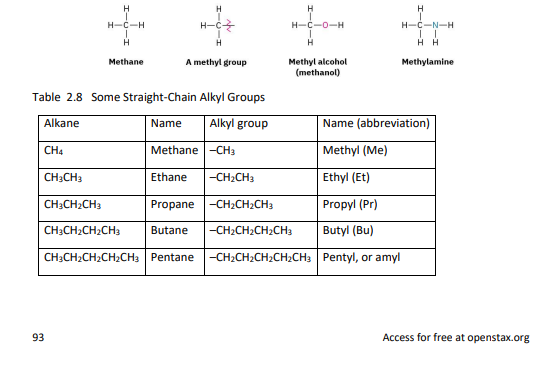
Just as straight-chain alkyl groups are generated by removing a hydrogen from an end carbon,
branched alkyl groups are generated by removing a hydrogen atom from an internal carbon.
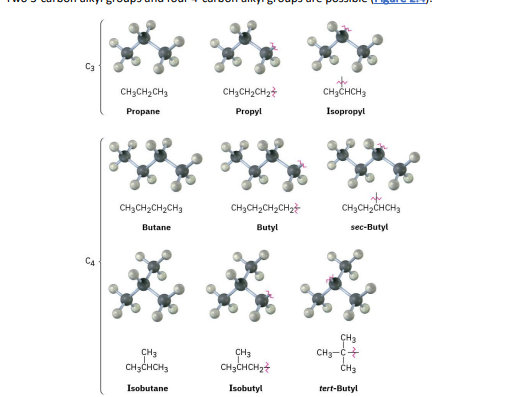
2.4 alkyl groups generated from alkanes (straight-chain)

naming hydrocarbons
parent, prefix, locant, suffix

2.5 - naming alkyl halides and alcohols
also haloalkanes
step 1: longest chain is the parent
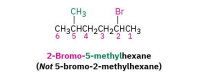
step 2: number the carbons of the parent chain beginning at the end nearer the first substituent whether alkyl or halo
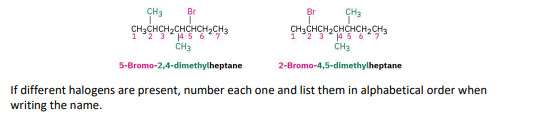
step 3: if the parent chain can be properly numbered from either end, begin at the end neaarer the substituent, alphobetical order
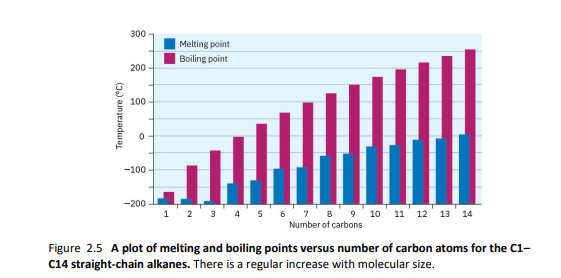
2.6- alkane properties
alkanes are sometimes paraffins/little affinity
alkanes show little chemical affinity for other substences and are chemically inert, meaningthey do not readily react with other chemical compounds under standard conditions. This lack of reactivity is due to the strong carbon-carbon and carbon-hydrogen bonds present in their structure, making them stable and unreactive in most chemical reactions. Alkanes are a type of chemical compound made up of carbon and hydrogen atoms. They are known for being very stable and not reacting easily with other substances. This means that they don't mix or react much with other chemicals, which is what scientists mean when they say they are 'chemically inert'. Because their structure is strong, with tight bonds between the carbon and hydrogen atoms, they can survive in most conditions without breaking apart or combining with other elements. This is why we often see them used as fuels, like in gasoline, where they can burn in a
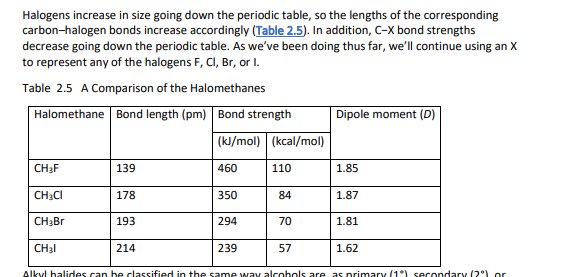
alkanes react with oxygen, halogens, and other substances given the conditions
oxygen reaction comes during combustion when alkane is used for fuel, and make CO2 and water as byproducts. large amounts of heat is released
CH4 + 2 O2 → CO2 + 2 H2O + 890 kJ/mol (213 kcal/mol)
reaction with alkane + Cl is from irradiation with ultraviolet light
-
2.7- chahnges in energy
reactions go in forwardor reverse direction
to determine the reaction, the spontaneity where products energy > reactants energy, it goes from reactant to product, where energy is released
-The energy change that occurs during a chemical reaction is called the Gibbs free-energy change (ΔG), which is equal to the free energy of the products minus the free energy of the reactants: ΔG = Gproducts − Greactants.
ΔG negative= favorable reaction. energy is lost by the chemical system and released to the surroundings, often as heat (exergonic)
ΔG postitive, energy is absored from surrounoundings, or endergonic
its faster to warm up than to cool off
Δ𝐺 ∘ = Δ𝐻 ∘ − 𝑇Δ𝑆 ∘

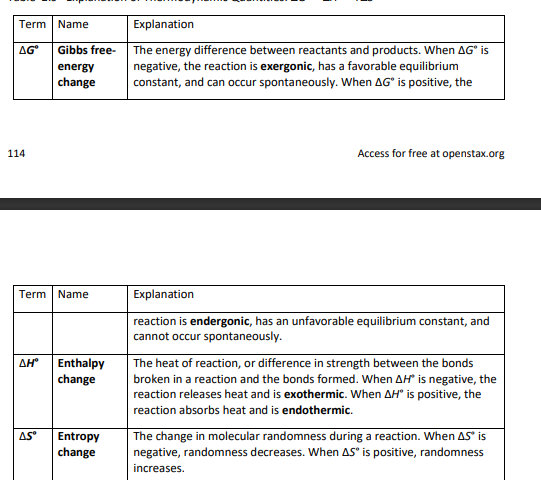
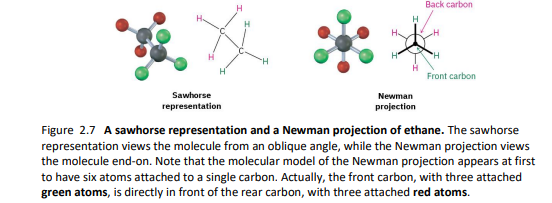
2.8- conformations of ethane
stereochemistry is the branch of chemistry concerned with three-dimensional objects
sigma bonds are cylindrically symmetrical (plane cutting through C-C looks like a circle)
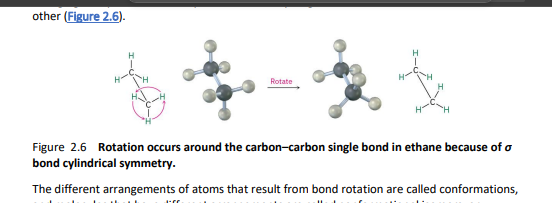
chapter 3- 3.1 naming cycloalkanes
cycloalkanes consist of rings of -CH2 units. they have general formul a(CH2)n or CnH2n and can be represented by polygons
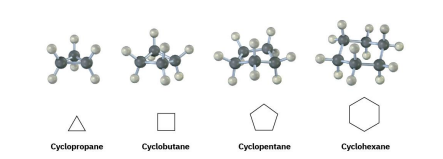
step 1: find the parent.
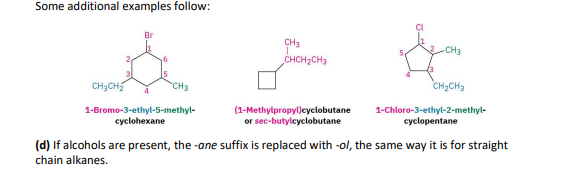
for c and h only= if its a ring, ring is parent chain and any alkyl groups are substituents. put cycloasprefix, and then use regular alkane naming ( cyclohexane)
for ring with alkyne/alkene= include such in the parent namestep 2: number substituents, write the name
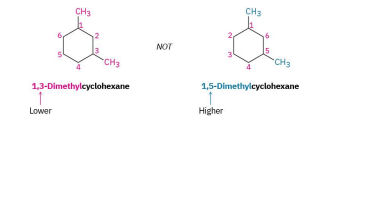
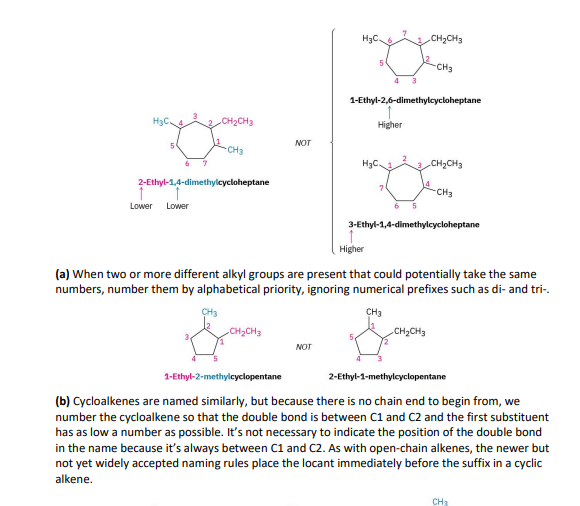
for an alkyl/halo substitued cycloalkane= choose a point of ring as 1, nnumber substituents on the ring so the SECOND substituent has as low a number as possible. if its too ambiuos, do third or fourth until a difference is found