lecture 20th - NO MORE FLOPS
metabolic pathways. which include a series of of enzymatic reactions and these are bascially chem reactions in which substrate of a pathways is going to be converted into a product right.
product of one pathway can be the substrate for another pathway. so the first commited step is usually the rate limiting step of the pathway. and it is also the pathway that is highly regulated, because in order to control if its on or off u controll this. this first step is irresible reaction. pay attention to energy generating vs energy using steps.
the reactions we talk about gneerate or utitlize atp or generate reducing equivalents . NADH FADH and NADPH. enzymatic reactions need cofactors or cdoenzymes, meaning the 20 amino acids are not enough on its own. these cofactor/enzymes are going to be vitamin derived. these vitamins are co enzymes and cofactors that make our enzymes work.
know all of the cofactors.
cofactors can be huge. dont draw the whole structure, know the business enf. coenzyme A has. abig and complicated structure with a sulfhydryl group that is going to medicate the chem of the reaction and overall shape. show an intermediate bound to the busness end of the cofactors. like. the acetyl coA on the sulfur. know the oxidation state of a coenzyme . the coenzyme can exist in multiple oxidation states. know whihc oxidation state if for each reaction.
Know the structure of the metabolites. KNOW THE PROBLEM SETSSS
REVIEW SESSIONS ARE IMPORTANT
overview concepts - types of pathways and types of bond cleave reactions and examples of enzymatic reactions.
enzymes accelerate the rate of reactions. coupling of enzyme reactions is common in metabolic reacitons meaning the product becomes the substrate for another reaciton. by coupling. two enzymatic reactions, u can make an unfavorable reaction move forward so the first reaction is unfaborable and the second is very faborable bc a little product is made and that product is used up immediately bu the enzyme emzyme vs thats a faborable reaciton. two types of metabolic pathways, meta and anabolic pathwyas.
the catabolic pathways are where larger molcules containing energy are degraded to sort of produce smaller moclules, as these energy containg molcules are metabolized they are going to generate some inttermediates … ex. in the prescence of o2 energy containg nutritens like carbohydates fats and proteins are degraded into CO2, water and if proteins into amino acids. and those amino acids degraded in to ammonia. the energy that was in these larger molcules is going. to be stored , in these small molcules are cofactors. ADP is going to become ATP. NAD + nadp+ FAD to NADH, FADH2, NADPH. the conversion of these eneryt contianing into energy depleting molcules involve oxidation reactions. so catabolix processes are generally oxidativ e. and in the process youre going to reduce NAD FAD and NADP+ to NADPH FADH2 etc. so the energy contain in these substrates in catabolic pathways is going to be temp transfered into these small molcules. and these small molcules are going to be used in biosynthetic reactions to build larger molcules from smaller molcules. so in anabolic pathways or biosynthetic pathways. small precursor molcules are going to be assembled to make larger moclucles from amino acids, sugars, fatty acids, to make proteins polysach lipids and nucleic acids and we need energy and need electorns. those electorns come from these small moclules in which had temp stored energy or electrons when we degraded our food which contained nutrients.
types of metabolic pathways -
converging catabolic pathways, diverging anabolic pathways, cyclic pathways.
catabolic pathways convergge into a common intermediate acetyl coA that can be used up in the citrict acid cycel to generate energy which is cyclic pathways . so u can see lot of dif molcules that all in the end generate acetyl coA which u can use to shunt it ot the TCA cycle .
Anatabolic pathways they are divergent, from a limited number precurorse like actyl coA and u can make a large number. of molcules like fatty acids, triacyglycerolds, steriods etc. they start from a common intermediate and diverse.
third one is the cyclic pathways, tca cycle, urea cycle whichi s another cyclic pathway.
iclicker the metabolic pathway is irreversible
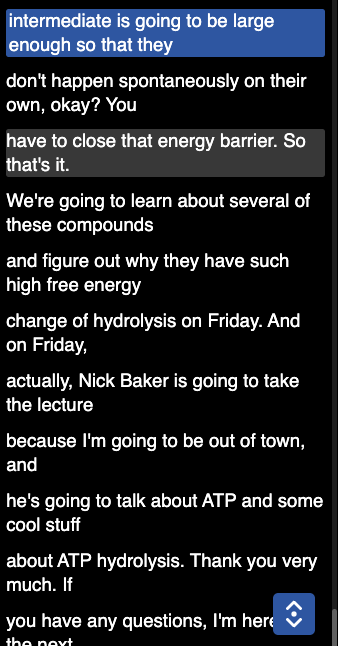
there are two types of cleavage reactions - homolytic and heterolytic cleavage of bonds. the chem bonds are electron pairs. so in homolytic bonds cleavage the electornpair is split so one electron goes to the carbon and the other goes to the hydrogen. so u form a cabon radial and a hydrogen atom.
radicals can happen,
heterolytic cleavage of a covalent bond if a protein is extracted and liberated from the carbon hydrgeon bond it is going to leave behing a carbanion or enolate anions. these are reactions in glyocys and TCA cycle(so the pair goes to the C). so the electron pair will either go to the C or the H.
know all three
phosphoryl tansfer using ATP as a phosphoryl donor
ATP is going to phorylate glucose to form glucose 6-phorphate and that is the first step of lgycolysis.
hydlase reactions - phsophate esters are going to be hydrolyezed and form phosphate esters
atp is going to
isomerase -
alpha hydroxy keton and it is going to siomerazed eto an alph hydroxy aldehyde and proceeds by a proton extraction and then u generate a enolate intermediate. and then u can reprotonate , and then u get an alhyde from this. so and enolate intermediate is made and u can reporotate and make and lahydete. so these enediole or enolate intermediate sort of helsp to isoemrize a ketone into an aldehyde. epimerases where configurationof an alpha carbon to a cabonyl
We will see examples
of epimerases, where the configuration of
an alpha carbon to a carbonyl carbon is
inverted in a reaction, okay? Turns out
this also occurs via enolate anion chemistry, okay? So in this case, a
proton is extracted here, okay? And that is going
to generate this enolate intermediate, and then
reprotonation, but on the opposite phase, okay,
it's going to invert the configuration of this
chiral center here, okay? We're also going to see
a number of reactions involving carbon
-carbon bond formation, where we have a proton
extracted, okay, from this alpha carbon to
a carbonyl, and that's going to generate,
again, an enolate anion. And that can act as
a nucleophile now on an electrophilic
carbonyl carbon, OK, to generate now a carbon
-carbon bond here, OK? OK, so we are also
going to see a number of dehydrogenation
reactions. Many of these are going to utilize NADH as a cofactor, where in a two-electron
reduction reaction, a hydride ion is added
to this carbonyl group to generate now
an alcohol here. So this is a two
-electron reduction. Again, in this slide,
I'm going to emphasize the carbonyl chemistry
and the importance of carbonyl groups in metabolic
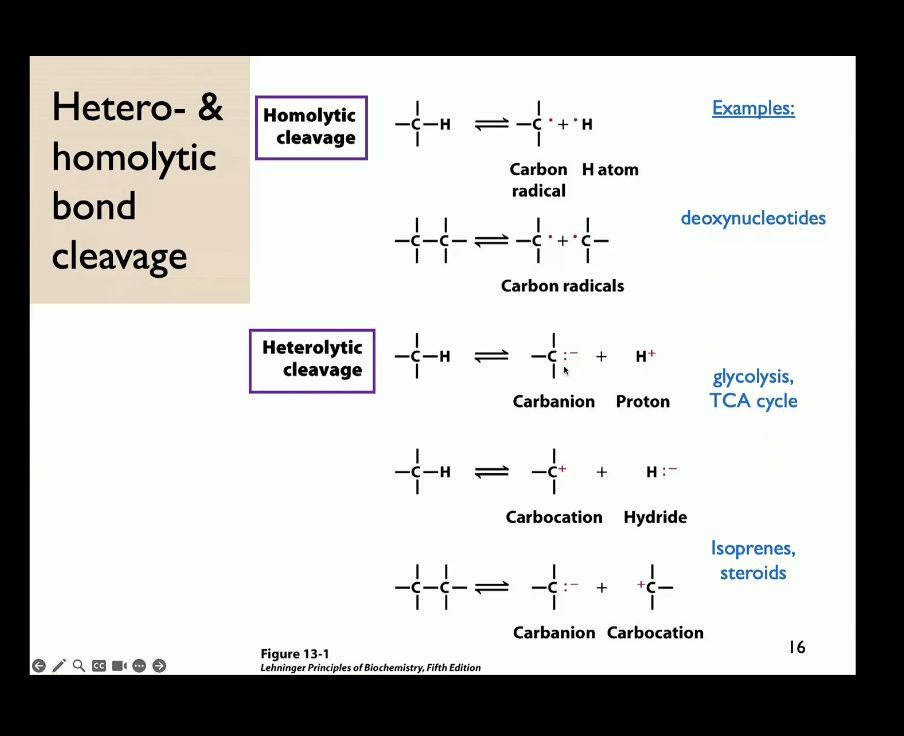
pathways. So here we have an alpha beta
unsaturated carbonyl carbon, right? Now you can draw
the resonance structures for this carbonyl
carbon here in which the electron pair is let's say
localized on this oxygen atom, right? So this is
going to make this oxygen electronegative and that
puts a positive charge on this carbon here,
right? That means that this carbonyl carbon is
now electrophilic, right? And that it can be now
attacked by nucleophiles, right? Now, you can
further draw the resonance structure that shows the
positive charge here that moves on this beta carbon,
right? So this means now you can add a
nucleophile to an alpha-beta unsaturated ketone in this
Michael addition here. So you can, you
know, by forming this alpha-beta
unsaturated ketone, now a nucleophile
can attack here and you can form a carbon
-carbon bond here. Of course, you can do the
reverse reaction as well, where you can abstract
a proton from this alpha carbon to again generate this
enolate anion intermediate, and then you can eliminate
this nucleophile to again generate this alpha
-beta unsaturated ketone. Does that make sense? Next, we're going to
talk about phosphoryl chemistry or phosphoryl
group transfer. Now, inorganic phosphate, as you know, is pentavalent. That means that
it has five bonds, right? It has five
bonds to phosphorus. Three of these oxygen
atoms are bonded to phosphorus in
single bonds, and one of these oxygen
has a double bond, right? Now, you can write four
different resonance structures for a tri
-anionic inorganic phosphate, which means that all of
these phosphorus oxygen bonds that are shown
here, they are equivalent. And also recall that inorganic phosphate is tetrahedral, and it's sp3 hybridized. Now, let's quickly
look at various oxidation states of carbon. Now, in this methyl
carbon of this alkane, the hydrogen and
carbon have similar electronegativities. OK. So
you can think of this carbon here having eight
electrons around it, right? Now, if you replace
one of these hydrogens with an oxygen atom, OK, oxygen is going to be more
electronegative, right? So it is going to own
those electrons, the pair of electrons in that
carbon-oxygen bond now. So what that means is
that this carbon is going to become deficient
by two electrons, right? Because oxygen
is going to pull those two electrons
towards itself. So we can say that in
this carbon-oxygen bond, it is still a covalent
bond, okay? But it has made this carbon deficient
by two electrons, okay? So basically what
that means is that you have oxidized this
carbon by two electrons. So in other words,
for each carbon -oxygen bond that you
make, you make the carbon deficient
by two electrons. So in an aldehyde or a
ketone, you have now two bonds with oxygen. That
means this carbon is now four electrons oxidized
relative to an alkane. Similarly, in a
carboxylic acid, which has three carbon
-oxygen bonds here, now what we're going to see
is that there's going to be this carbon which is
deficient by six electrons. And of course, in
carbon dioxide, you have four bonds to oxygen,
and that is eight electron oxidized
relative to an alkene. Now, if you think about
alkenes, the carbon -carbon bond in
double bonds are two electrons oxidized
relative to an alkane. All right. So now we're
going to, in the next 10 slides, review some
of the thermodynamics concepts that you will
need to understand as we discuss regulation
of metabolic pathways.
skip this whole part cuz wtf. . .
delta G is the change in free energy it represents enery of a reaction abaible to do some work. u will know if its forward or not. decrease in free energy when a substrate is converted into produce. A goes to B and theres a decrease in delta G. that means that the energy is realsed in the reaction that is faborable and exothermit , op is true when delta G is postive. negative delta G is spontsanoue sbut doesnt say anything about the reaction rate. or the kinets and the kinetics is determined by the height of the transition state. detla G = deltaH -Tdelta S
delta H is change in enthaply which is teh change in kids and numbers of chem bonds and non covalent interactions that are broeknd or formed during reaction, change in entahlpy is change in the heat content also. delta S is change in entropy/randomss/disorder
entropy is always increasing. going from small to large molcules u decrease the entropy, u decrease disorder bc u make a large molcuels form small. in chem reactions it will not be clear wehre changes in entropy are coming from. one example is hydrophobic molcules tend to stick together . weve all heard that hydrophic molcules like. to stick together bc if theyre isolated in wter , then water had to be ordered around the hydophobi molcules so it needs energy. so associating with other hydrophobic molcules there is a decrease in the order of the molcules or in other words an increase in entorpy. delta G is the free energy change under nomral cellular ocnditions .
if the start concentraion of substrate and products were one molar then delta G is called standard free energy change.
mejor esta
So catabolic pathways are going to be large molecules being broken down like carbohydrates, fats, protein they are broken down into smaller molecules and energy is released, htis happens with oxidation reaction so the loss of electrons. So carbs = co2 + h2o, the energy released is temporarily stored in energy carriers which are ATP, NADH, FADH2, NADPH, these are cofactors. . Then the carriers are reduced to NAD+, FAD, NADP+. These small cofactors molecules , atp, nadph, and nadh2, are going to be used to build larger molecules. So the energy and electrons come from these. This is anabolic pathway (building - using energy)
Anabolism involves the usage of the atp nadh, nadph, fadh2 in order to make larger molecules, like sugars, maino acids, fatty acids, to make proteins, lipid, polysachs.
So in catbolism, the carbs, fats, proteins are all degraded to become acetate/acetyl-COA, this is convering catbolism because glucose, fatty acids, amino acids, etc,all conver to acetyl coA. And then in the diverging pathway the acetyl coA becomes many different molecules. It wil be the building block to many different moclules. So making of cholesterol, fatty acids, steroids, the Acetyl coA can also go into cyclic pathway, like the krebs, TCA, etc. So in the cyclic pathways, it can make NADH, FADH2, and produce intermediates.
So large molcuels - acetyl coA- TCA cycle = nadh, fadh2, atp, , but its only small percent that is made, the nadh fadh2 is already made when catbolism initially happens.
There are two types of cleave reactions homolytic and heterolytic. This is where the electrons go when a bond is broken. So in homolytic bonds the electrons can split off in half, one to one element and the other one to the other one so there are two radicals.
So if its C-H = C. + .H and only the carbon is a radical. If its C-C = C. + .C so now both carbons are radicals. This happens in deoxynucleotides…
In heterolytic cleavage -
This means that both electron pairs are either going to one element or the other. So if its C-H bond and both go to the carbon then the carbon gets a - and the hydrogen a +
So C-H = C:(-) + H(+), the c- is a carbanion, and the H is proton - this is involved in glycolysis, TCA cycle
—--
If it were to go to the H then the C would be + and the H would - so that is a carbocation, and a hydride.
If its a C-C bond, then one is a carboanion and the other is a carbocation
And these both are for isoprenes and steroids.
—----
Types of common biochemcial reactions - .
Kinase where atp is going to phorhpoylate glucose to make glucose - 6 phosphate + adp. So one P is released from ATP (cuz it has three) to add it onto the glucose. This is the first step of glycolysis (but just know when a phosphate is transferred, it is a kinase reaction)
Hydrolase - hydrolysis where we use water to break a bond. So in the example we take phosphate ester and we add water to make a free phosphate and free molecule that was the R group
Isomerase involves using a ene-diol/enolate intermediate in order to go from keton to aldehyde. ( this involves a proton extraction, then a reprotonation, that is going to make an enolate, and then that is going ot help isomerize into an aldehyde) so from alpha hydroxy keton to alpha hydroxy aldehyde
Epimerase - this is gong to involve izomerization at chiral carbon but making it inverted. Configuration of an alpha carbon to carbonyl corabon is inverted, and this involves enolate anion chemistry , so a proton is extracted to make the enolate intermediate, and the reportonation but onhte opposite phase to invert configuration at chiral center
Aldolase - condensation of a enoalte with a carbonyl. So a C-C will be make by removing a proton from the alph carbon to the carbonyl and that makes an enolate anion that will nucelphile attach a electorphilic carbonyl carbon (c=0) that is going to generate a C-C
Dehydrogenase: oxidation/reduction involving hydride shifts. So we use NADH as a cofactor, we take two electrons from it so it becomes nad+(the electrons came with the H from the NADH and the H is also added to the carbon) and we add and H(a hydride) to make an alcohol.
B unsaturated carbonyl - so there is a double bond between A and B and there is a carbonyl. Resonance will allow the protons form the double bond go up to the O and then
phosphorous is penta valent meaning that inroganic phsophate has 5 bonds, 3 are single bonds to o and one is double bonded to o and there are four resoance structures that are all equivalent and it is tetrahedral and sp3 hydbridized.
oxidation states of carbon
methyl carbon of alkane, the hydrogen and cabon have similar electronegativies. think of this carbon having 8 electrons around it. if u replace on Hydrogen with an O the O is more electrongeative so the O is going to own the pair of electron in the carbon oxygen bond. this carbon is going to become deficient by two electrons. this carbon - o bond is still covalent but its made it deficient so we oxidized the carbon by 2. so for eat carbon oxygen bond u make u make it defeicent by two electrons
so if we make a aldehyde or a keont where the carbon is C=O that meaans the carbon is 4 electorn oxidized relative to an alkane. same thing with a carboxylic acid that has three oxygen bonds the cabon is defiect by 6 electrons, finally if its 4 bonds it is 8 electron oxidized ( 0=C=0)
if u think about alkenes, the carbon carbon bond in double bonds are two electron oxidized realtive to an alkane.
review material delta G is the change in free enerrgy/gibbs free energy which represents the enregy of ar eacion that is aviabel to do some work. knowing the value of delta G we can predict if a reaciton is going to move forward/spontenously.
so if in a cellular reaciton thers a decrease in free energy when the substrate is convered into produc like A to B then there is a decrease in delta G. meaning it is favorable and exothermic. so if there is an increase in free energy so delt + it is unfaborale and endothermic. delta G doesnt mention how fast the reaction happens. the kinetics of the reaction is detmeined by the heigh of the transition state.
delta G = delta H - T delta S
as long as delta G is negative it is spontenous. S is entropy, entropy is always increasing, in cells if u are creating a larger molecule from smaller, u are decreasing diy hydrosorder so u put in work. whphibic molecules like to stick together bc if they are isoalted in water, water has to be ordered around the hydrophobic molecules and tha tneeds order so by assocating with other hydrophobic molcules ther eis decrease in the order and increase in entropy , delta G is the free ernegy change under normal cellular. conditions. if the starting conce of a substrate and product were one molar then delta G is called standard free energy change. this is a delta G naught bc its a one molar concentiron, if it were one molar concention at 25 centigrade at PH 7.0 this is called delta G naught prime. bc its PH and temjperature.
when a reaciton goes from a to be at equilibirum, we can plug in delta G prime naught is equalt to -RT ln (B eq / A eq) or -rtln keq
singe of delta G predicts the directions. if its less than 0 the keq is greater than one, the reaction goes from left to right. A converts to B
if its dleta G is greater than 0 , keq is less than 1, and B goes to A. RIGHT to left
cells are not a equilibrium, delta G depends on the concentraion of susbtrates and products bc the products of a reaction may be used up by another reaction and that is going to pull the reaction foward.
So delta G = delta G naught prime + RT ln (B/A concentration )
free energy diagram of ten steps of glycolyss. steps 4,5,8 its unfaborable bc delta G is positive. and this is caluclated under standard conditions. if we calcualte the actual gree energy of the glycoltic steps from red blood cells or ehrtyoctyes, real cells u can see that the all the reactsions are faborable, and its bc the metabolic flux in a cell is influenced by conc of substrates and products. reactions that were not faborable instandard conditions, become faborbale bc the products of these reactions in actual cells are used up in the next step
indivual free energy changes of sequital reactions are additive, yielding an overall free energy change fro the coupled reaction.
how do we get unfaborable reactions to occur? it runs out delta G are additive , so if u convert A to B and that is unfabobale but the B converted into C. is favorable, then thats going to pull. reaction from A to B.
a gier is attached to a weight which is going to make the gear spontaneoulsy turn towards lowering the weight cuz its heavy, and if we do the reverse reaction the weight is heavy, it is energetically unfabole, meaning u hav e 4to pull the weight up. in figure C. the energetically faborble movment is used to turn a second gear in a direction that it would not move, sort of sponteanously.
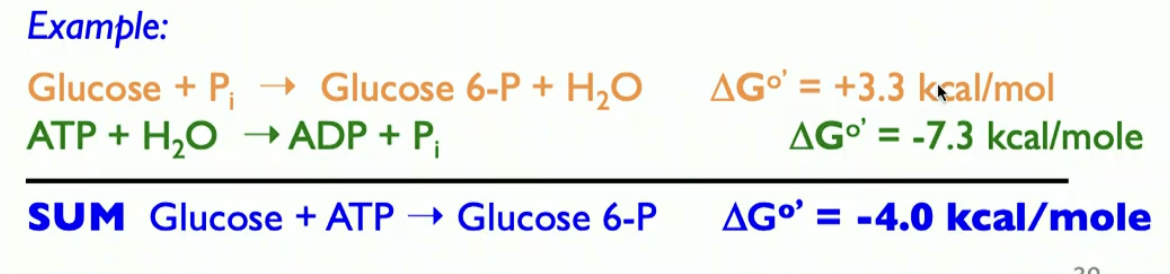
so glucose + P = a postive delta G so its UNFAVORABLE,
ATP and h20(atp hydrolysize ) delta g is negative it is favorable. and if u sum up the reactions the total delta G is negative so it is favorable. that is why ATP is used to phorphorylate a lot of substrates in biochem

we can look at equilibrium constants wehn u couple both reactions the Keq increases by 250k fold. bc the equilib constants are in a couple reaction are multiplciative, delta G are additive bu equilrium constats are mutlicpicative.
free enregy changes associated iwth reaciton, some have large free enregy changes and associated with hydrolysis reactions including anhydrides like ATP and othe rphoryl containg compounds. but many of the transomfriaotn we see in the metabolic pathways have modest/small free energy changes that are closer to one. if u look at oxidation of glucose with diatomic o going to carbon dioxide and water, if u look at palmitate , fatty acid oxidaiton, u see the delta G naught prime or the standard free energy change is very negative and huge.
anything that has a standard gree energy of hydrolysis that is less thanminus 6 kulocalories per mole is considered to be a high ernegy compound. ATP is a hgh enreyg moclules and its standard free energty change of hydrolysis is minus 7.3 kilo caolires per mole. atp is not the only energu source of cells , utp, ctp, gtp, tey all have simial. amount of standard erengy of hydrolysis minus 7.3. so all equivalent.
t is the NTPS plus acetyl coa and 1,3 bisphohsphoglycerate which are sort of highlighted here which are the high energy compounds in the cell. that is bc the high energy compounds are thermodynamically usntable and that is but theyre kinetically stable what that means is that their hydrolysis is favorable but theyre not going to hydrolyz unpreditabolity on their one.