Rates of Reaction
Chemistry - Rates of Reaction
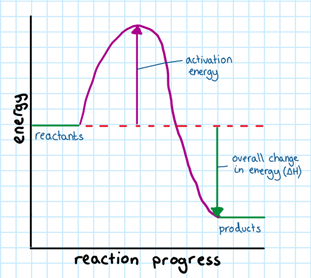
Collision Theory: The idea that particles have to collide in order to react (at the correct angle and with enough energy) for a successful chemical reaction to occur.
The minimum amount of energy needed for a successful reaction is called the activation energy. If they collide with energies below the activation energy they do not react.
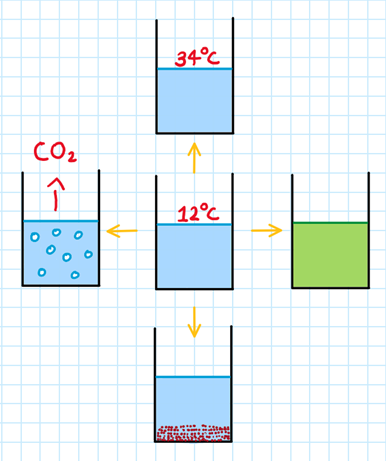
Some typical signs of a chemical reaction taking place are:
A change in temperature
A gas being produced
A colour change
Precipitate (an insoluble solid) being formed
The rate of reaction is the ‘speed at which a chemical reaction proceeds’. It is measured by change in concentration ÷ change in time.
The 4 factors that affect the rate of reaction are:
Concentration
Temperature
Surface Area
Catalysts
But how?
Concentration:
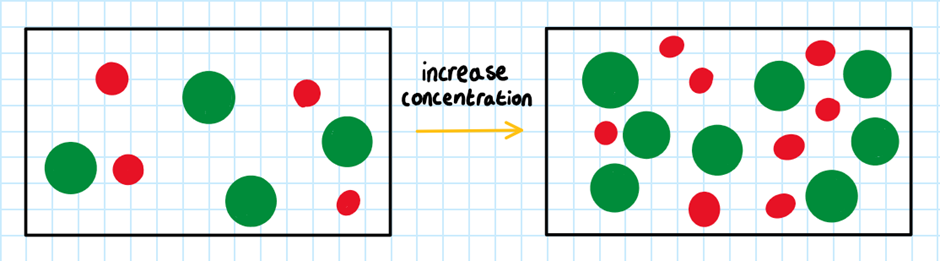
RULE 1: The higher the concentration of the reactants, the faster the rate of reaction.
When the concentration increases, there are more molecules per volume available to collide with each other. Therefore, there is a higher frequency of collision, so more chances of successful collisions. Because of this, there is an increased rate of reaction.
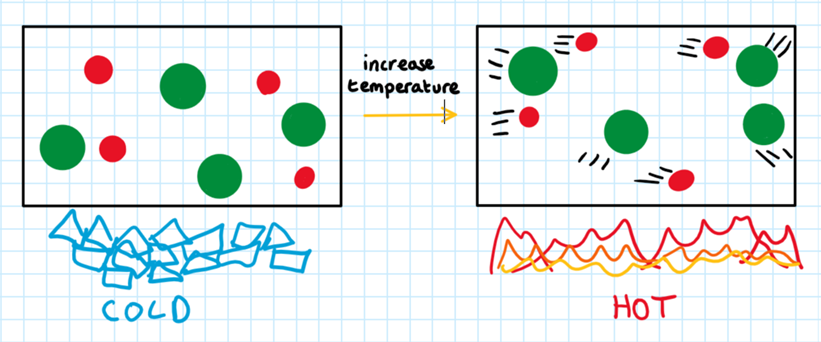
Temperature:
RULE 2: The higher the temperature of the reactants, the faster the rate of reaction
The temperature affects the rate of reaction in two ways:
It increases the energy of the molecules.
It increases the speed of the molecules
A higher temperature means that the molecules have a higher kinetic energy. This means that more molecules will have enough energy to overcome the activation energy. This increases the rate of reaction.
A higher kinetic energy means that the molecules have increased speed. This means that they move around a lot more, meaning that there is a higher frequency of collision and a higher chance of successful collisions. This increases the rate of reaction.
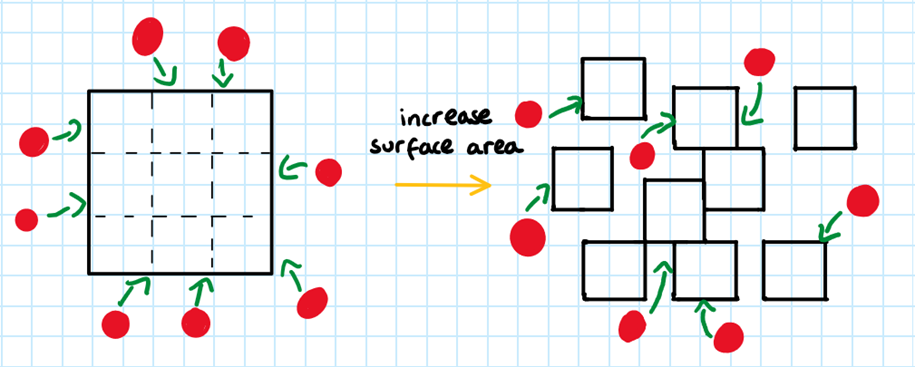
Surface Area (solids only):
RULE 3: The larger the surface area of the reactants, the faster the reaction
A larger surface area means that there is a greater area onto which particles can collide, increasing the collision frequency. Therefore, there is a higher chance of successful collisions. This increases the rate of reaction.
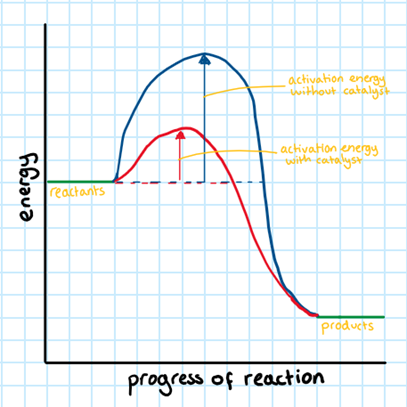
Catalysts:
RULE 4: The higher the amount of catalysts, the faster the rate of reaction.
A catalyst is a chemical substance that speeds up the rate of reaction without being consumed. You have probably already come across the concept of catalysts in biology (enzymes are biological catalysts).
A catalyst provides an alternate pathway for the reaction with a lower activation energy. This means that more molecules will now have enough energy to successfully collide. This increases the rate of reaction.
As mentioned above, the formula for the rate of reaction is change in concentration ÷ change in time. To be able to measure how fast the reaction is happening, we can use one of the five techniques below:
We can measure how fast a volume of gas is produced / gas volume produced in a certain time.
We can measure how quickly the color change occurs or when a cross is no longer visible due to the precipitate.
We can measure the mass lost over time.
We can measure the time taken for a solid to no longer be visible.
We can measure how fast the temperature change occurs.
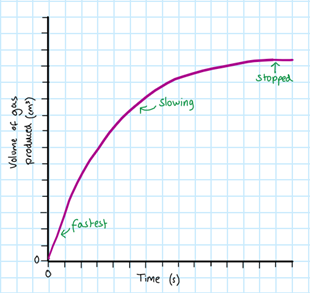
For any of these methods, you must record your results in a table, and then plot them into a graph. An example of a graph that you might draw looks like the one on the right.
As the time increases, the rate of reaction decreases. This is because as the reaction proceeds, the reactants are being used up to produce the products. This results in a decrease of concentration of reactants, which lowers the frequency of collision. Therefore the rate of reaction is lower and the graph becomes less steep. At the end, all the reactants have been used up and there is no longer a reaction. This is shown by a plateau on the graph.
The fastest rate is always at the very beginning of the reaction. This is also where we calculate the initial rate from (time = 0). We use a gradient to do this.