INTRODUCTION TO PS:
PHYSICAL SCIENCE
is a branch of natural science that is concerned with the study of nonliving materials
includes Chemistry, Physics and Earth Science
CHEMISTRY
deals with the study of composition, structure, properties, and changes in matter
PHYSICS
deals with the properties and interaction of matter and energy
EARTH SCIENCE
is concerned with the study of Earth and the universe around it
it is divided into four namely: geology, oceanography, meteorology and astronomy
Matter
anything that occupies space and has mass.
States of Matter
Solid
Liquid
Gas
Plasma
Bose-Einstein Condensate
Solid
where atoms or molecules are in contact
force of attraction are strongest
crystalline sugar, salt, and metals
Solid object
occupies definite shape and volume and is independent of its container. It can no longer compressed because its particles are tightly packed
Types of Solid
Crystalline
solids that have well-defined shape
Their particles exist in highly ordered arrangement.
calcite, quartz, sugar, diamonds, etc
Amorphous
solids whose particles are organized in random patterns.
charcoal, rubber, glass
Liquid
state of matter where particles cohere but are not so rigid
molecules are usually separated with greater distance compared to that of the solids
has a fixed volume but no definite shape. Particles are held together strongly by attractive forces and are in close contact with another.
are free to move past one another
motion of particles passing one another causes it to take the shape of its container.
Movement of atoms
gives a liquid its distinctive property which is the ability to flow
Viscosity
measure of a fluid’s resistance to flow
shampoo, soda, oil and gasoline
Gases
has no definite volume and shape.
distance of atoms or molecules is much greater than in liquid.
expands to fill its container.
its particles move independently of one another and they have enough energy to overcome the attractive forces that hold them together
it bounces and presses continuously in all directions against the walls of its container
Plasma
state of matter is only under extreme condition
matter is changed into an aggregate of positively charged particles
These particles move very rapidly in a cloud of negatively charged electrons
stars, lightning, welding arc, auroras, neon signs
Bose Einstein Condensate
only developed in laboratories
occurs in temperature close to absolute zero
particles here move closer, but stopping when temperature is absolute 0.
made up of bosons
BEC
Eric Cornell & Carl Weiman
were the ones who proved the theory of two physicist, Albert Einstein & Satyendra Nath Bose.
Atom
came from the Greek word “atomos” which means “indivisible” or “uncuttable”.
smallest particle that makes up matter
fundamental building blocks of matter.
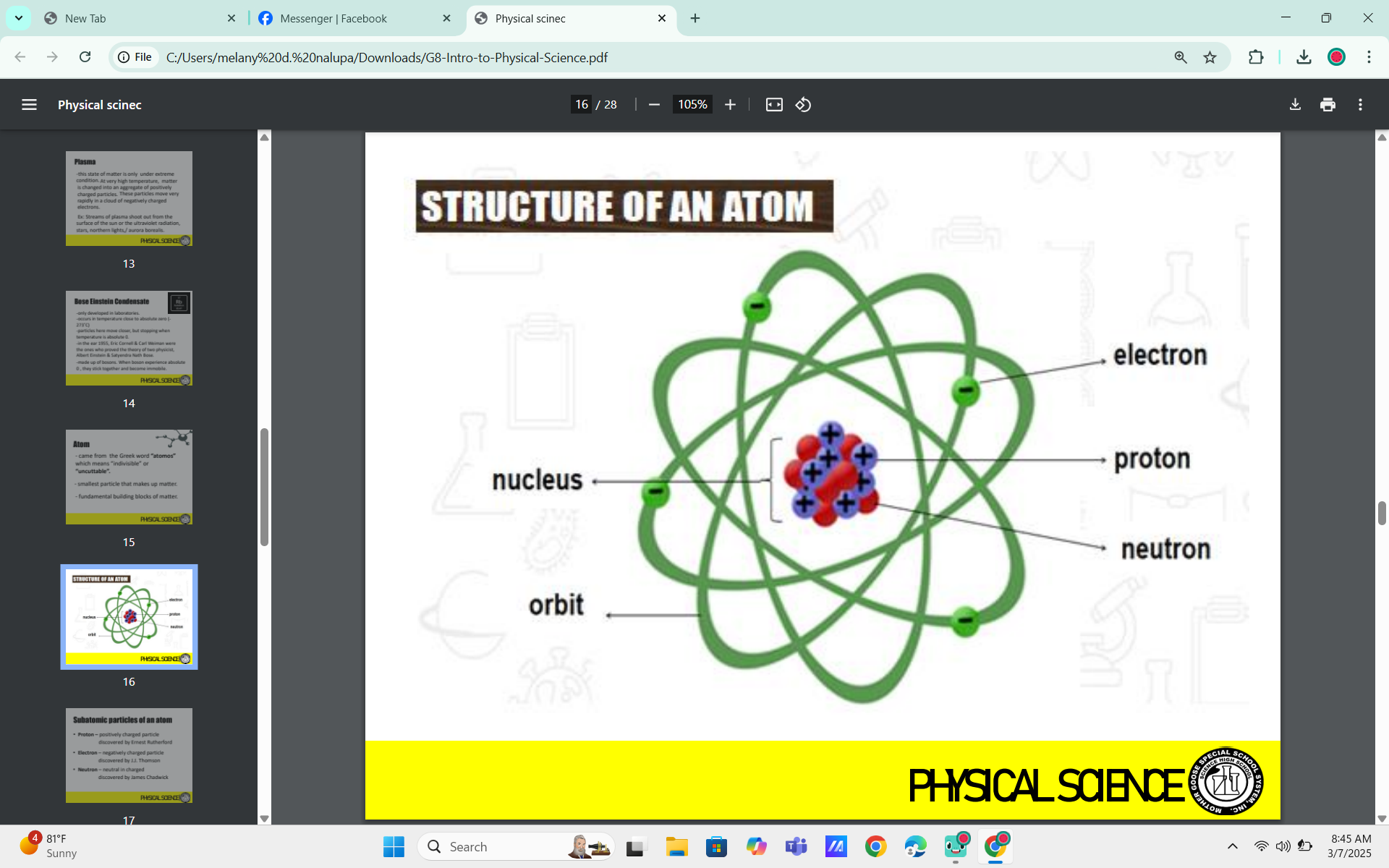
Subatomic particles of an atom
Proton
positively charged particle
Ernest Rutherford
Electron
negatively charged particle
J.J. Thomson
Neutron
neutral in charged
James Chadwick
Matter can be classified into two categories:
Pure Substances
are either elements or compounds.
Elements and Compounds
Mixtures
can either be homogeneous or heterogeneous.
Homogeneous and Heterogeneous
Element
type of matter which cannot be broken down into two or more pure substances
In chemistry
elements are identified by their symbols
consists of one or two letters usually derived from the name of the elements
118 recorded
elements in the periodic table
Compound
is a pure substance that contains more than one element
Example:
Water
contains hydrogen and oxygen
Table salt
contains sodium and chlorine
Mixtures
contains two or more pure substances combined in such a way that each substance retains its identity
Two types of mixtures:
Homogenous
or uniform mixtures.
The composition of the mixtures are the same throughout.
Heterogeneous
or non uniform mixtures
those composition varies throughout.
Solution
common type of homogeneous mixture
Solute
is the substances that gets dissolved
Solvent
dissolving medium
Several methods in the chemical laboratory can be used to separate components of a mixture:
Filtration
passing the solution using a filter medium with fine pores.
Distillation
applying heat vaporizing the liquid leaving a residue behind
liquid can be recovered in pure form
Chromatography
this is more complex, but more versatile separation of liquids based on their solubility and affinity or adsorption on a solid surface in case of paper chromatography or through the gas chromatography for mixtures of volatile solutions.
Properties of Matter
Physical property
those that you can find is by direct use of your senses and by weighing and measurement.
inherent characteristics of a substance that can be determined, observed, and measured without changing its composition.
Example: color, shape, hardness, luster, ductility, malleability, conductivity, density, solubility roughness, smoothness, sweetness, saltiness, elasticity, tensile and more
Physical properties of matter fall into two categories:
Intensive properties
do not change whether the amount of substance is small or large.
Color
if we add a 5 kg of water to a 10 kg of water, it is still blue.
BP/MP
when a 5 kg of water and 10 kg of water are boiled at 100˚C.
Extensive properties
depends on how much matter
mass, length, volume, internal energy, heat, enthalphy, entropy, time, circumference, diameter, distance, area, width, and thickness
Richard C. Tolman in 1917
Intensive and extensive properties were introduced by
Chemical property
those that when you do observation, chemical change must be carried out.