Chem Sept. 10th
Delta charges, bond polarity, and the role of formal charges
Finishing module 1; then module 2 on Friday.
Start with a quick review: drawing organic molecules is basic; as soon as we move to molecules, start thinking about delta minus (δ−) and delta plus (δ+).
Which atoms in a molecule bear δ+ or δ−? Bonds can be polarized; not all bonds are nonpolar covalent.
Polarization tells us which atom is slightly negative and which is slightly positive, which is crucial because those charge distributions drive most chemical reactivity.
When looking at molecular drawings, consider lone-pair electrons (or lack thereof) and how many lone pairs are present. This ties directly to formal charges.
Goal: be able to determine formal charges from a structure and understand how resonance can spread charge over multiple atoms.
Formal charges: definition and calculation
Formal charge (FC) occurs on an atom in a molecule when the atom is assigned a net charge in a drawn structure.
How to determine FC (two equivalent ways shown in lecture):
Method A (explicit electron accounting):
FC = V − NB − B
V = number of valence electrons for the atom in the periodic table.
NB = number of nonbonding electrons (lone pairs counted as two electrons each).
B = number of bonds to the atom (each bond counts as 1 bond for this calculation).
Method B (standard textbook form):
FC = V − L − 1/2(Bonding electrons)
Since each bond contributes 2 electrons and each atom is assigned one electron per bond, this is equivalent to FC = V − NB − B.
In practice: FC = V − (nonbonding electrons) − (# of bonds to the atom).
Quick mental shortcut: FC can be read off by counting electrons assigned to the atom, subtracting the electrons that are unshared, and subtracting one electron for every bond (since a bond has two electrons, and we assign one to each atom in the bond).
Note on向 drawing: You may see different representations (with or without explicit lone pairs). If a formal charge exists, you should either include the lone pairs that justify it or clearly show the charge. Both representations describe the same charge distribution.
Worked examples from the lecture (formal charges)
Oxygen in a structure with 6 valence electrons, 2 lone pairs, and 3 bonds:
V = 6, NB (nonbonding electrons) = 2 (one lone pair is two electrons, but two lone pairs = 4 electrons; the slide shows a case with 2 lone electrons shown, leading to FC calculation as shown):
Using the method described in lecture: FC = 6 − 2 − 3 = +1.
Quick interpretation: this oxygen is electron-deficient relative to its neutral valence, hence δ+ on oxygen and related δ− on the adjacent atom.
Carbon in a four-bond situation with no lone pairs:
V = 4, NB = 0, B = 4 → FC = 4 − 0 − 4 = 0 (carbon is neutral).
Nitrogen in a case described during the lecture that gave it a positive character due to ring or adjacency to pi systems:
With two bonds and few or no lone pairs, nitrogen can appear with a positive formal charge in localized structures.
This electron-deficient nitrogen can be a key site for reactivity in certain intermediates (e.g., when discussing resonance and electron-rich vs electron-poor sites).
Aluminum in a typical three-coordinate environment (Al with three bonds and no lone pairs):
V = 3, NB = 0, B = 3 → FC = 3 − 0 − 3 = 0 (aluminum formally neutral in this vertex description; note other descriptions of oxidation state may differ).
Chlorine in the middle of a structure, with 7 valence electrons, 4 nonbonding electrons (two lone pairs), and 2 bonds:
V = 7, NB = 4, B = 2 → FC = 7 − 4 − 2 = +1.
In a typical C–Cl bond this would be δ− on Cl and δ+ on C, but in this particular drawing Cl bears a formal +1 due to the distribution of lone pairs and bonds in the shown structure.
Key takeaway from these examples: formal charges are sensitive to how many lone pairs you keep in the drawing and how many bonds the atom has; different valid drawings can yield different formal charges on the same atom if lone pairs are placed or omitted.
Emphasis on correctness: if a structure shows an atom with an improbable charge distribution or violates valence rules (e.g., a carbon with too many electrons without a corresponding bond), it’s a sign the structure is drawn incorrectly.
Formal charges in practice and quick checks
When you see a charge on an atom, ensure there are appropriate lone pairs and bonds to satisfy valence.
If you draw a structure with a formal charge, you should be able to reproduce the calculation to justify the charge.
If you choose not to show all lone pairs, you must still indicate the presence (or absence) of formal charges to avoid ambiguity.
It’s common to draw resonance forms to move electrons around while keeping the overall molecular charge the same.
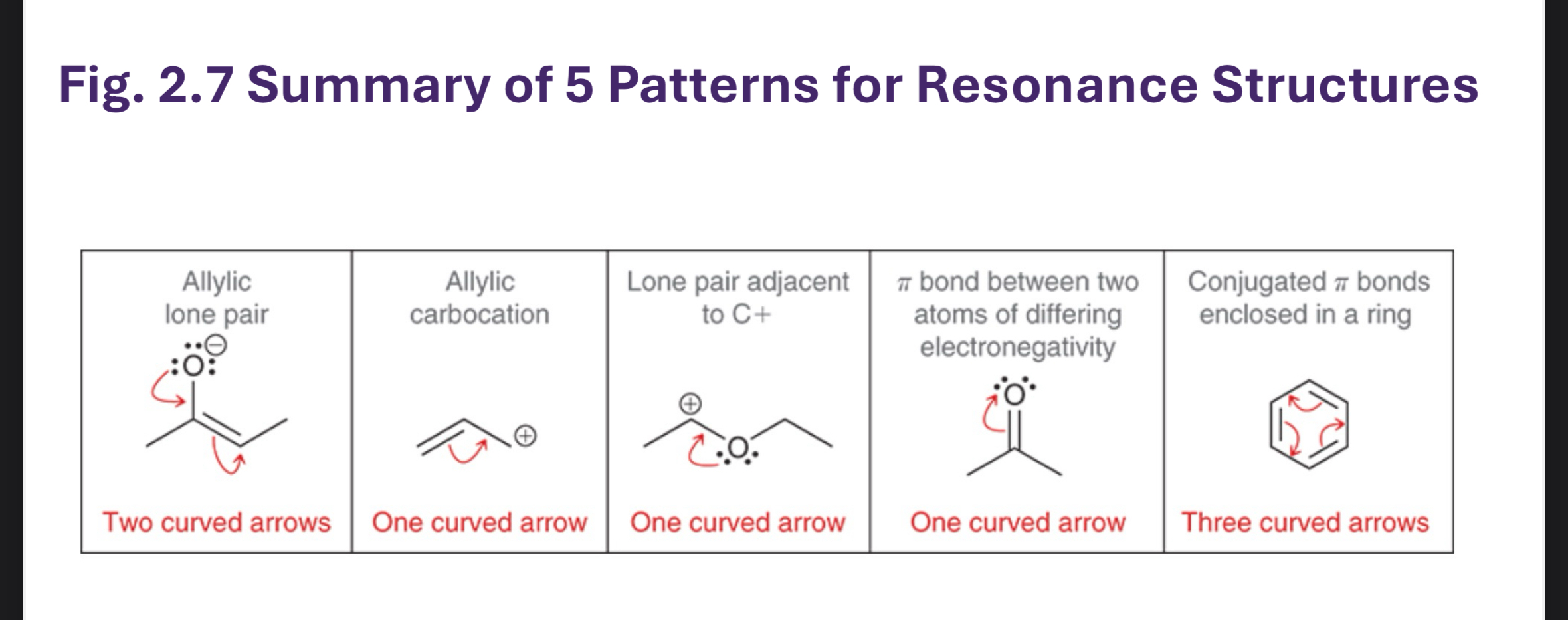
Resonance structures: concept and rules
What is resonance? The sharing of electrons in a pi system across adjacent atoms; not the movement of electrons in sigma bonds.
Localized Lewis structures vs resonance hybrids:
We often draw multiple localized structures (resonance forms) that depict electrons in different positions.
The real molecule is a resonance hybrid of these forms.
Examples of resonance forms:
A molecule with a lone pair adjacent to a pi bond (allylic lone pair) or a carbocation adjacent to a pi system can have multiple valid resonance forms.
In one form, a lone pair on an atom overlaps with a neighboring pi system; in another form, those electrons are localized in a bond elsewhere, creating a different formal charge distribution.
Major vs minor contributors:
Not all resonance forms are equally important. The major contributor typically has the most complete octets and places negative charges on the most electronegative atoms, while placing positive charges where they are less electronegative (or distributing charges to minimize separation).
The real structure is a blend (hybrid) of all valid resonance forms, with some forms contributing more than others.
Practical consequence:
For example, a resonance form may place a negative charge on oxygen (more electronegative) rather than on carbon, making that form a major contributor.
A resonance form with two equally charged atoms (e.g., an O−–C+ arrangement) might be a minor contributor if it places a positive charge on a less electronegative atom.
How to think about resonance in the course of reactions
Resonance is about electron delocalization in pi systems, not about breaking sigma bonds.
When you push electrons with curved arrows, you must conserve the overall molecular charge and keep octets intact for second-row elements unless you explicitly depict a reaction step (which would involve breaking/creating sigma bonds, not resonance).
The arrows (curly arrows) indicate the movement of two electrons: from a lone pair or a bond to another position.
If a resonance form implies breaking a sigma bond without a reaction step, that form is not valid as a resonance contributor.
You may see allylic and allyl cation/anion examples repeatedly:
Allylic position = the atom adjacent to a pi system.
Lone pairs adjacent to pi systems can participate in resonance, distributing charge over multiple atoms.
The major resonance form often places negative charge on the most electronegative atom (e.g., oxygen) and minimizes formal charges elsewhere.
Allylic systems and examples discussed in class
Allylic lone pair or allylic cation: adjacency to a pi system enables resonance via lone-pair donation or pi-electron rearrangement.
Each allylic situation can be drawn with multiple resonance forms showing delocalization of electrons across the pi system and the adjacent atom.
Consequences for reactivity:
Delocalization can stabilize certain intermediates (e.g., carbocations adjacent to pi systems) and affect regioselectivity and basicity.
The real structure is a hybrid with partial double-bond character and partial charges spread over several atoms.
Conceptual takeaway: resonance explains why we sometimes see partial bonds (e.g., a C–O bond with partial double-bond character) and why certain atoms appear more basic or more electrophilic in different resonance forms.
Ethanol and common drawing conventions
Ethanol (ROH) as an example: oxygen is valence 6; typically has two bonds (to carbon and hydrogen) and two lone pairs, giving a neutral O.
In quick sketches, you may omit drawing all lone pairs; the two-electron counts still give neutrality, but if you’re using lone pairs for arrow pushing, you should include them.
If a resonance or arrow-pushing step involves the lone pairs on oxygen, it’s important to show those lone pairs clearly to justify electron movement.
Practical implications and exam-style guidance
What to watch for when solving problems:
Always check octets for second-row elements when proposing resonance forms.
Use curved arrows to move only electrons in pi systems or lone pairs adjacent to pi systems; avoid breaking sigma bonds in resonance forms.
When assigning formal charges, be consistent with the number of bonds and lone pairs; ensure that the overall charge of the molecule remains unchanged across resonance forms.
For major contributors, favor structures with minimized formal charges, especially placing negative charges on more electronegative atoms (like oxygen) and avoiding charges on highly electropositive atoms unless required by the structure.
Real-world relevance: understanding resonance and formal charges helps predict nucleophile/electrophile sites, reaction outcomes, and stability of intermediates in organic reactions.
Instructor’s note: the current discussion emphasizes practice with resonance and arrow pushing; the next module (Module 2) will build on these ideas and show how resonance interacts with reaction mechanisms.
Quick recap of key terms
Formal charge (FC)
Valence electrons (V)
Nonbonding electrons (NB) / lone pairs
Bonds (B) and the bond count for FC calculation
Delta plus (δ+) and delta minus (δ−) charges
Resonance structures and resonance hybrid
Allylic position
Curly-arrow notation for electron movement
Octet rule for second-row elements
Connections to prior and future content
Builds on: basic Lewis structures, electron counting, and lone-pair chemistry introduced earlier in the course.
Sets up: Module 2, which will continue with resonance in more complex systems and begin applying these concepts to reaction mechanisms.
Instructor reminders and study tips mentioned in the talk
If I assign a task (e.g., practice problems), try it; it’s likely to appear on assessments.
Tests of knowledge aren’t the primary motivator here, but grades matter for you, so use the given guidance to study effectively.
Practice problem sets (e.g., Practice 2.13) illustrate common mistakes in arrow pushing and help reinforce the proper rules for resonance and electron movement.
The ongoing takeaway: be fluent in identifying δ+/δ−, calculating formal charges, and evaluating resonance structures to identify major contributors and the true electronic structure of molecules.