IM= SOB
Fetal Heart Development
The heart develops near the head of the embryo in the cardiogenic area.
Cardiogenic cords form and develop a lumen, becoming endocardial tubes.These tubes eventually fuse to form a single primitive heart tube, which then undergoes folding and segmentation to give rise to the chambers of the heart.
These tubes migrate and fuse into a single primitive heart tube.
The primitive heart tube forms five distinct regions (from head to tail):
Truncus arteriosus
Bulbus cordis
Primitive ventricle
Primitive atrium
Sinus venosus
Venous blood initially flows into the sinus venosus.
Contractions propel blood from tail to head (sinus venosus to truncus arteriosus).
Heart Borders
Right border: right atrium
Inferior border: right ventricle and a portion of the left ventricle
Left border: left ventricle and a portion of the left auricle
Superior border: both left and right atria
Apex: left ventricle
Tracing the Heart Borders
Superior border: From the inferior margin of the second left costal cartilage (3 cm left of the median plane) to the superior border of the third right costal cartilage (2 cm from the median plane).
Right border: From the end of the superior border to the sixth right costal cartilage (2 cm from the median plane).
Inferior border: From the end of the right border to a point in the fifth intercostal space near the midclavicular line.
Left border: Connects the left end of the superior and inferior borders.
Fetal Circulation
Oxygenated blood from the umbilical vein/inferior vena cava (IVC) is diverted through the foramen ovale to the head.
Deoxygenated blood from the superior vena cava (SVC) goes through the pulmonary artery and ductus arteriosus to the feet and umbilical arteries.
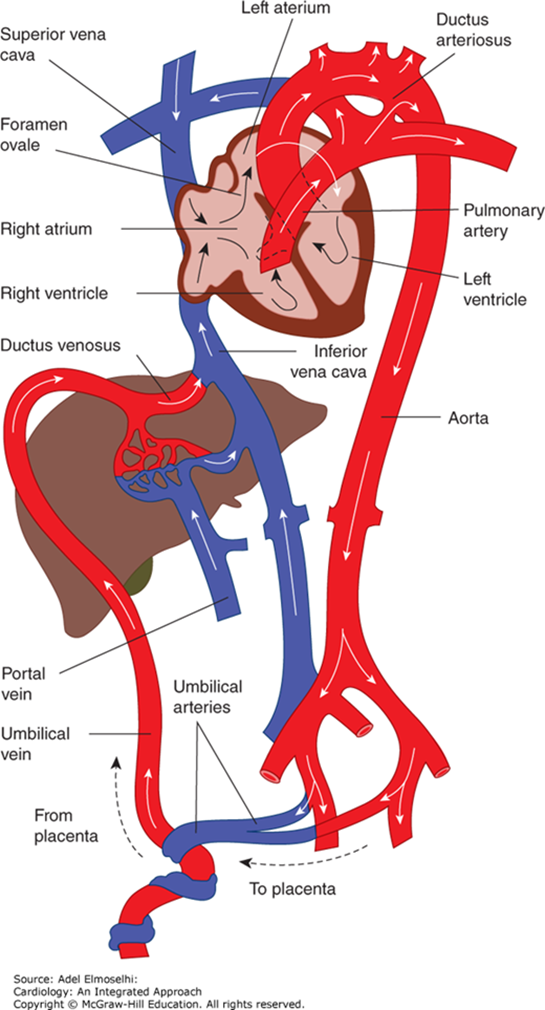
Blood Flow
Placenta → IVC → RA → Foramen Ovale → LA → LV → Ascending Aorta → Head & Upper Extremities
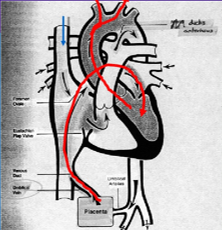
SVC → RA → RV → Pulmonary Artery → PDA → Descending Aorta → Lower Extremities and Placenta
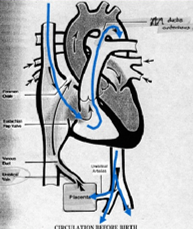
Only a small amount of blood reaches the lungs.
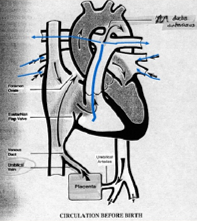
Key Differences in Fetal Circulation
Different anatomically and physiologically from postnatal circulation.
Fetal hemoglobin (Hb) has a higher affinity for oxygen.
Pulmonary vessels are constricted due to:
Lungs filled with fluids
Hypoxia causing pulmonary vasoconstriction
One umbilical vein and two umbilical arteries.
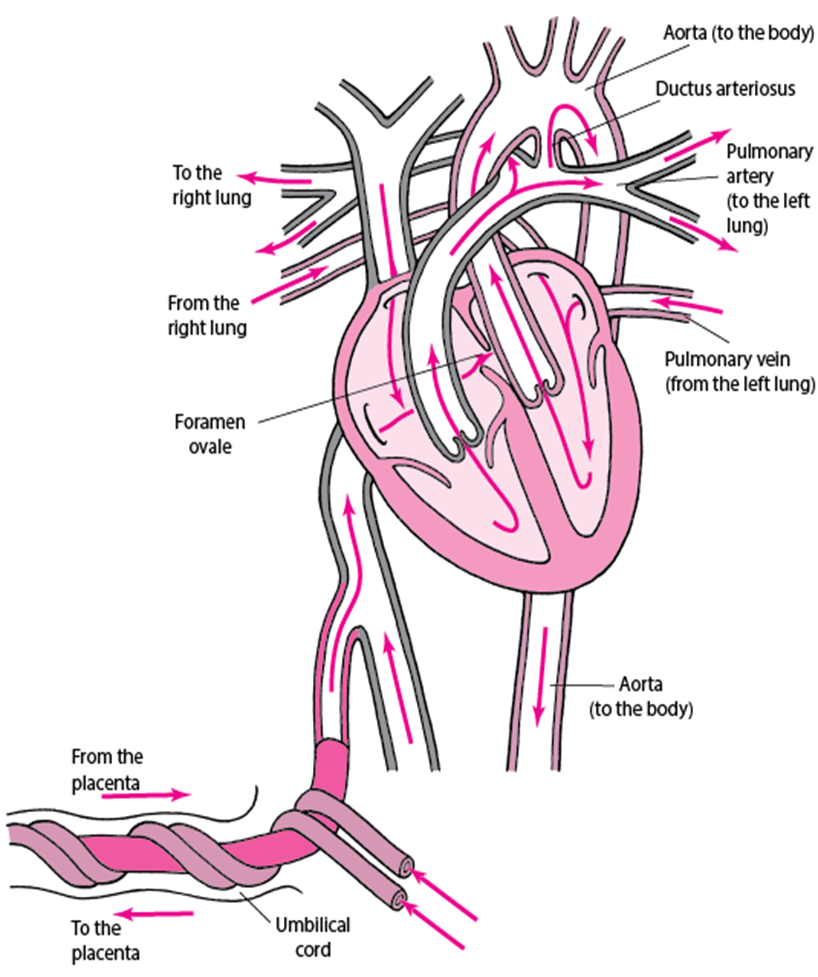
Ductus venosus bypasses hepatic circulation into the inferior vena cava.
Right atrium (RA):
40% of blood passes through the foramen ovale into the left atrium (LA) → LV → systemic circulation
60% passes to the right ventricle (RV)
Right ventricle (RV):
Due to high pulmonary resistance, 90% of blood shunted through ductus arteriosus into systemic circulation
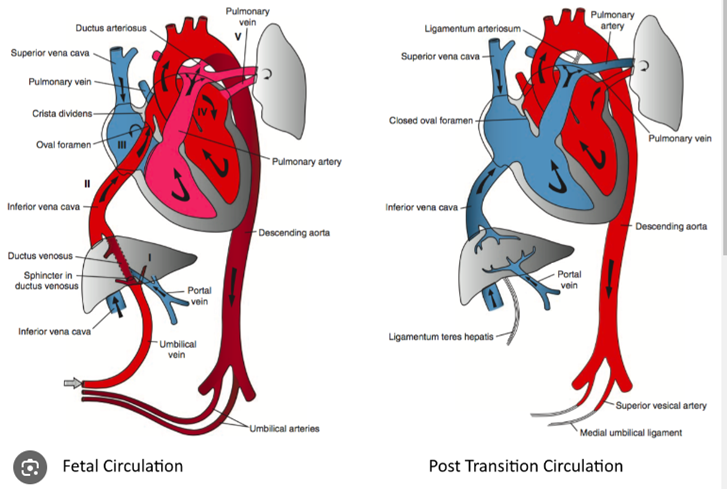
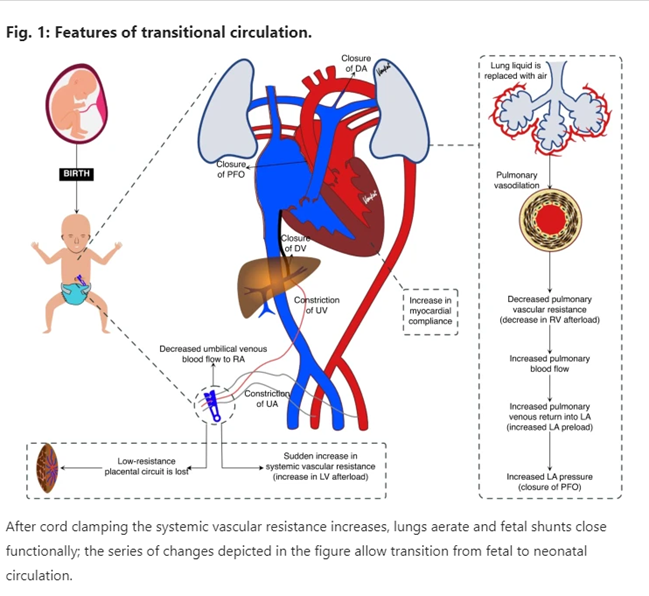
Transitional Circulation at Birth
Taking the first breath:
Replaces fluids with air, increasing oxygen and decreasing pulmonary vascular resistance.
Reduces right-sided pressure.
More blood shunted to the lungs for ventilation.
Cord clamping and removal of low-resistance placental circulation:
Increases systemic vascular resistance and left-sided pressure.
Regression of medial smooth muscle layers due to persistent low pulmonary pressure.
Factors affecting post-natal pulmonary vascular development:
Alveolar hypoxia, prematurity, congenital heart defects.
Post-Transition Features
Closure of:
Foramen Ovale (PFO)
Ductus Venosus (DV)
Umbilical Arteries (UA)
Umbilical Vein (UV)
Ductus Arteriosus (DA)
Low-resistance placental circuit lost.
Sudden increase in systemic vascular resistance (increased LV afterload).
Lung liquid replaced with air.
Pulmonary vasodilation.
Increased myocardial compliance.
Decreased pulmonary vascular resistance (decreased RV afterload).
Increased pulmonary blood flow.
Increased pulmonary venous return into LA (increased LA preload).
Increased LA pressure (closure of PFO).
After cord clamping, the systemic vascular resistance increases, lungs aerate, and fetal shunts close functionally, allowing transition from fetal to neonatal circulation.
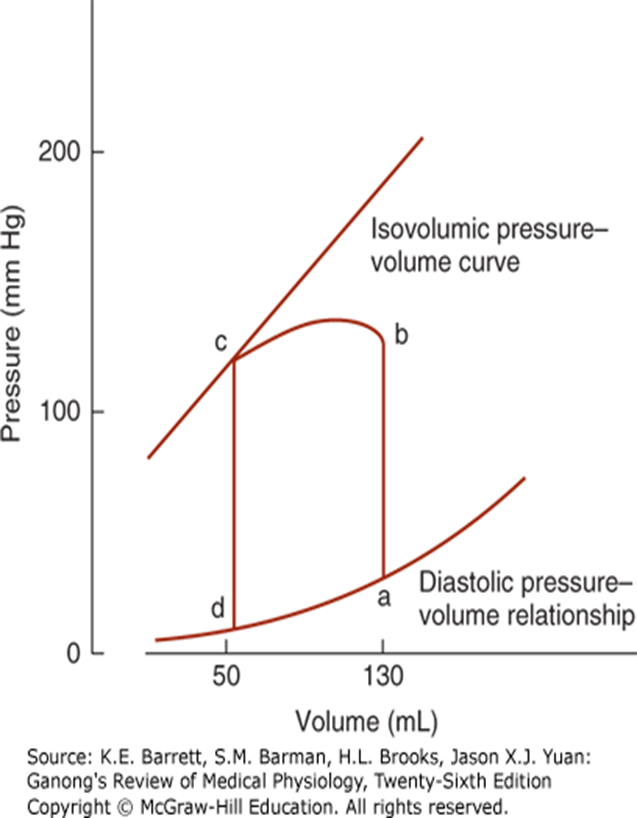
Ventricular Pressure-Volume Loop
Diastole: Ventricle fills (d to a), pressure increases.
Isovolumetric contraction: Pressure rises sharply (a to b).
Ventricular ejection: Pressure rises (b to c).
Aortic valves close: Pressure falls during isovolumetric relaxation (c back to d).
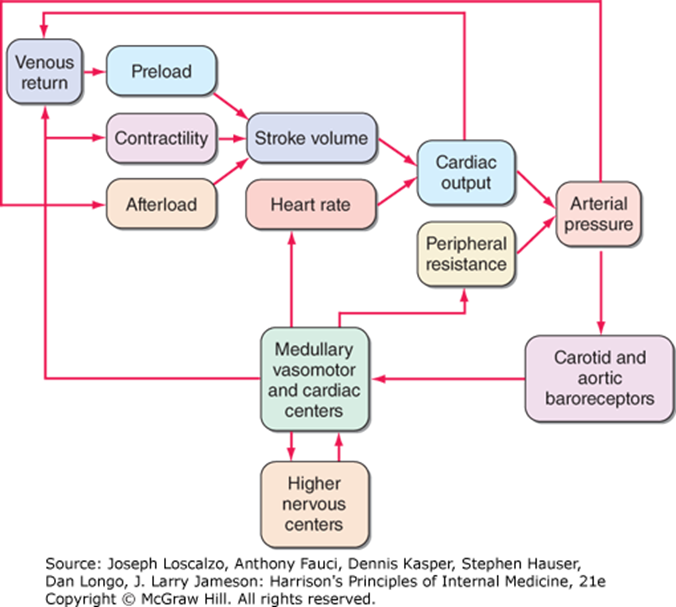
Factors Affecting Stroke Volume and Cardiac Output
Preload: Venous return affects preload
Afterload: Arterial pressure and peripheral resistance affect afterload which determines the amount of force the heart must generate to eject blood during systole.
Contractility: Myocardial strength and the availability of calcium ions influence contractility, impacting overall cardiac efficiency.
Heart Rate
Medullary vasomotor and cardiac centers, higher nervous centers, and carotid and aortic baroreceptors all are factors.
Myocardial Contractility
Intramyocardial [Ca^{2+}]
Cardiac adrenergic nerve activity
Circulating catecholamines
Cardiac rate
Exogenous inotropic agents
Myocardial ischemia
Myocardial cell death (necrosis, apoptosis, autophagy)
Alterations of sarcomeric and cytoskeletal proteins:
Genetic
Hemodynamic overload
Myocardial fibrosis
Chronic overexpression of neurohormones
Ventricular remodeling
Chronic and/or excessive myocardial hypertrophy
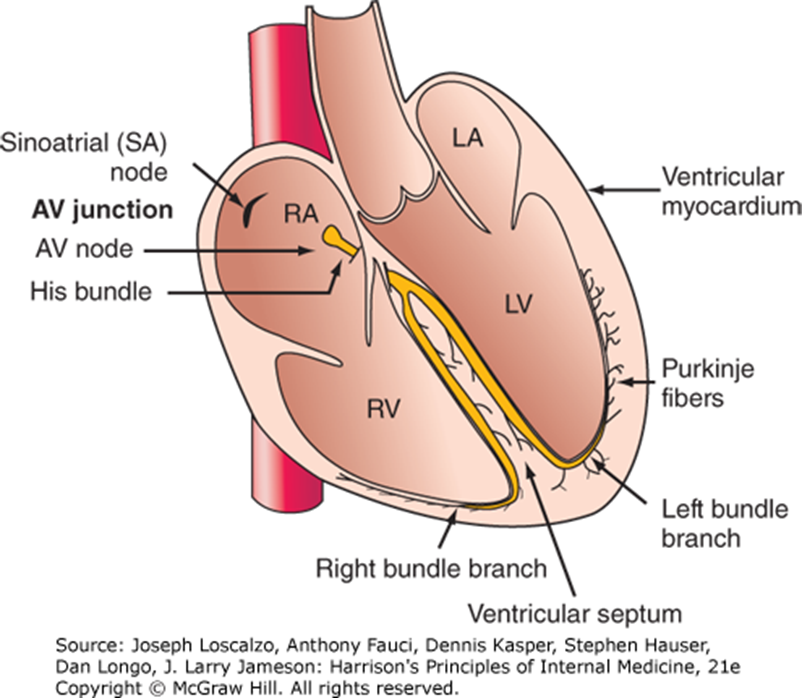
Cardiac Conduction System
Sinoatrial (SA) node → AV junction → AV node → His bundle → Left and Right bundle branches → Purkinje fibers → Ventricular myocardium
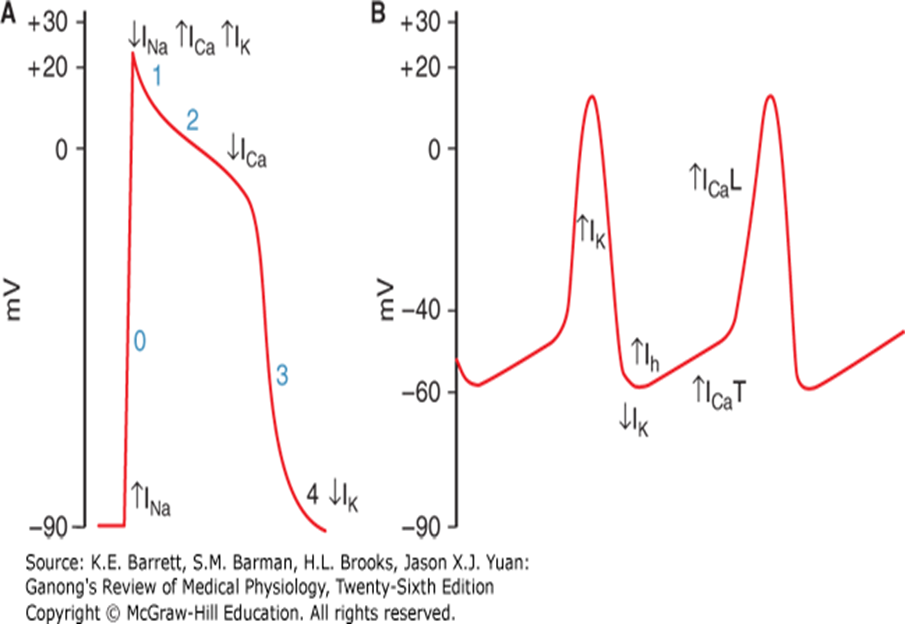
Action Potentials in SA and AV Nodes
Largely due to Ca^{2+} influx.
Phase 0: Upstroke phase
Phases 1 & 2: NOT present
Phase 3: Repolarization
Phase 4: Spontaneous depolarization or pacemaker potential
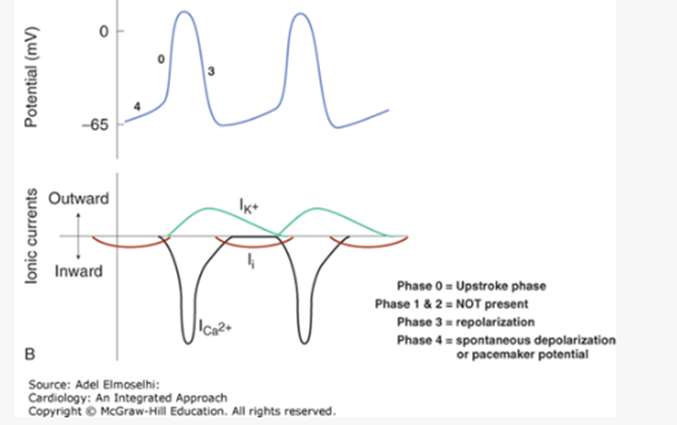
ECG Waves and Intervals
P wave: Atrial depolarization
QRS complex: Ventricular muscle depolarization
T wave: Ventricular repolarization
PR interval: Conduction time from atrium to ventricle
QRS duration: Intraventricular conduction time (time for all ventricular cells to activate)
QT interval: Duration of ventricular action potential
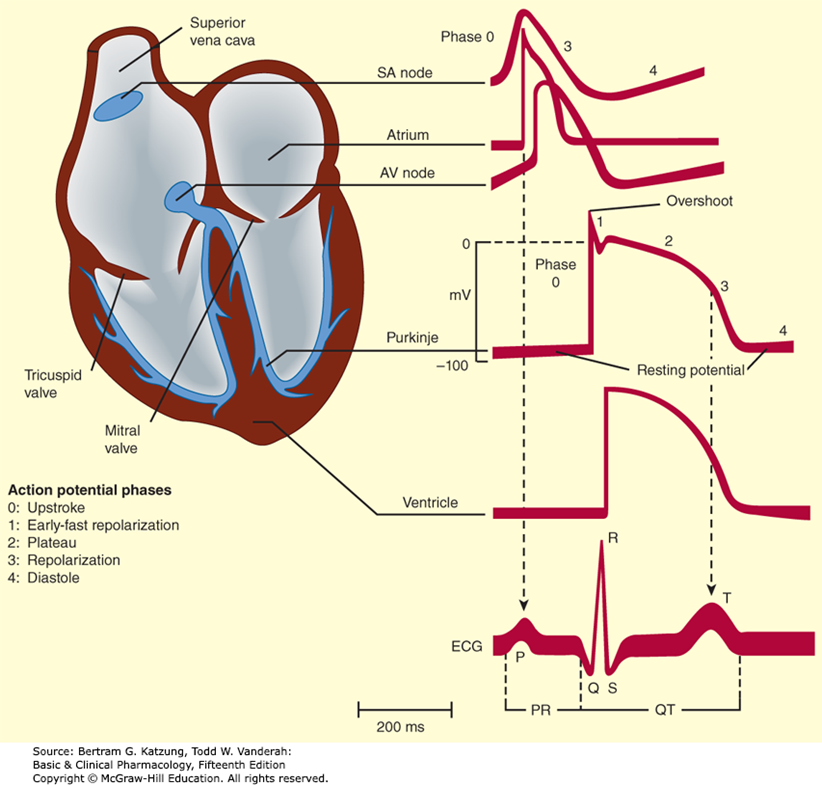
Cardiac Metabolism
Fatty acid oxidation (FAO) is a major energy contributor in the normal heart.
FAO is less energy efficient than glucose oxidation due to higher oxygen consumption.
Optimizing cardiac energy metabolism involves inhibiting FAO and inducing glucose oxidation.
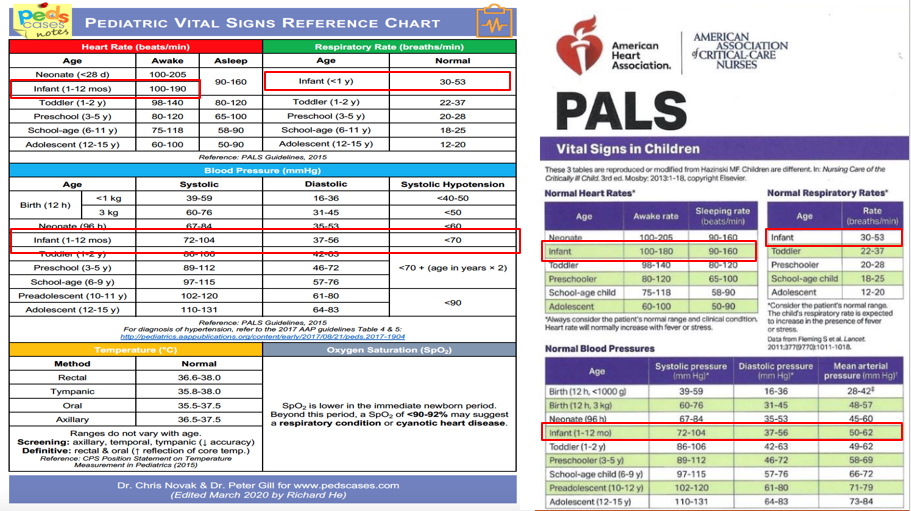
Pediatric Vital Signs Reference
Heart Rate (beats/min), Respiratory Rate (breaths/min), Blood Pressure (mmHg), Temperature (°C), Oxygen Saturation (SpO2)
Given for different age categories: Neonate, Infant, Toddler, Preschool, School-age, Adolescent
Also references systolic hypotension and normal blood pressures across age categories
Cyanosis
Bluish discoloration of skin and mucous membranes due to insufficient oxygenated blood mixed with deoxygenated blood.
Occurs when oxygen saturation falls below 80%.
Represents 5g of deoxygenated hemoglobin.
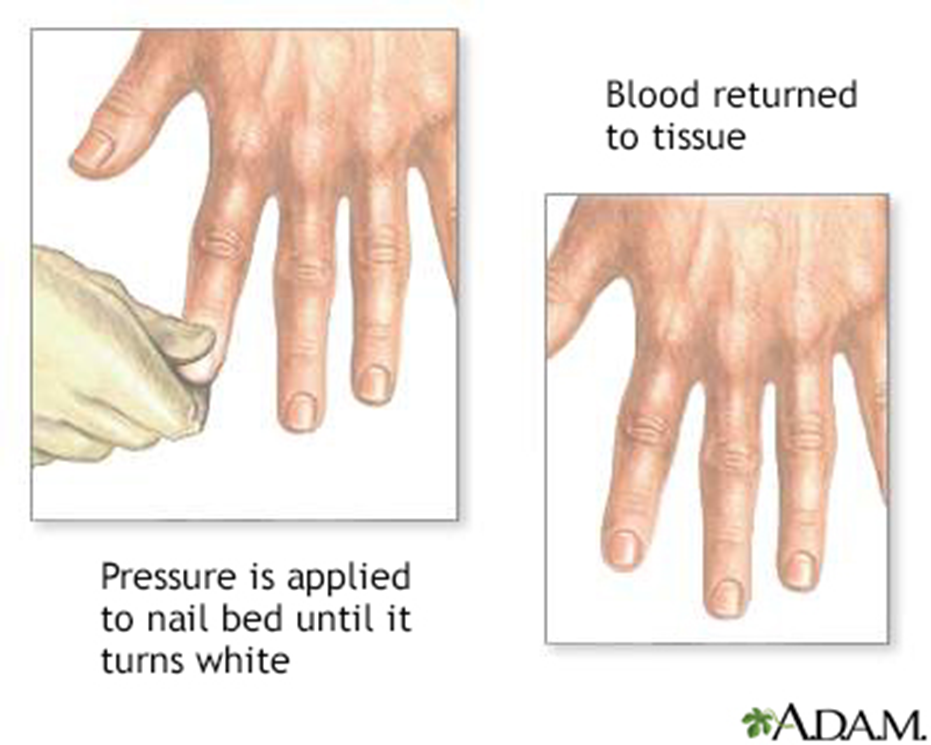
capillary refill time is a clinical test used to assess peripheral perfusion and can provide valuable information about a patient's circulatory status. An abnormal capillary refill time, defined as a refilling duration of more than 2 seconds, may indicate poor peripheral circulation.
Peripheral Cyanosis Causes
Reduced cardiac output:
Cold exposure: prolonged exposure to cold temperatures can lead to constriction of blood vessels, resulting in reduced blood flow and subsequent cyanosis in peripheral regions.
Redistribution of blood flow from extremities: in cases of shock or certain cardiac conditions, the body prioritizes blood flow to vital organs, leading to a decrease in circulation to the hands and feet, which may cause cyanosis.
Arterial obstruction: can occur due to various factors such as thrombosis, embolism, or atherosclerosis, further contributing to decreased blood supply to essential organs and exacerbating symptoms of ischemia.
Venous obstruction: can also significantly impact blood circulation, leading to increased venous pressure and potential edema in affected areas, thereby accentuating the risk of ischemic complications. DVT
Central Cyanosis Causes
Decreased arterial oxygen saturation:
Decreased atmospheric pressure (high altitude)
Impaired pulmonary function
Alveolar hypoventilation
Inhomogeneity in pulmonary ventilation and perfusion
Impaired oxygen diffusion
Anatomic shunts
Congenital heart disease
Pulmonary arteriovenous fistulas
Multiple small intrapulmonary shunts
Hemoglobin abnormalities
Hemoglobin with low affinity for oxygen
Methemoglobinemia
Sulfhemoglobinemia
Carboxyhemoglobinemia (not true cyanosis)
Classification of Congenital Heart Disease
A cyanotic:
↑ Pulmonary blood flow:
Atrial septal defect
Ventricular septal defect
Patent ductus arteriosus
Atrioventricular canal
Obstruction to blood flow from ventricles:
Coarctation of aorta
Aortic stenosis
Pulmonic stenosis
Cyanotic:
↓ Pulmonary blood flow:
Tetralogy of Fallot
Tricuspid atresia
Mixed blood flow:
Transposition of great arteries
Total anomalous pulmonary venous return
Truncus arteriosus
Hypoplastic left heart syndrome
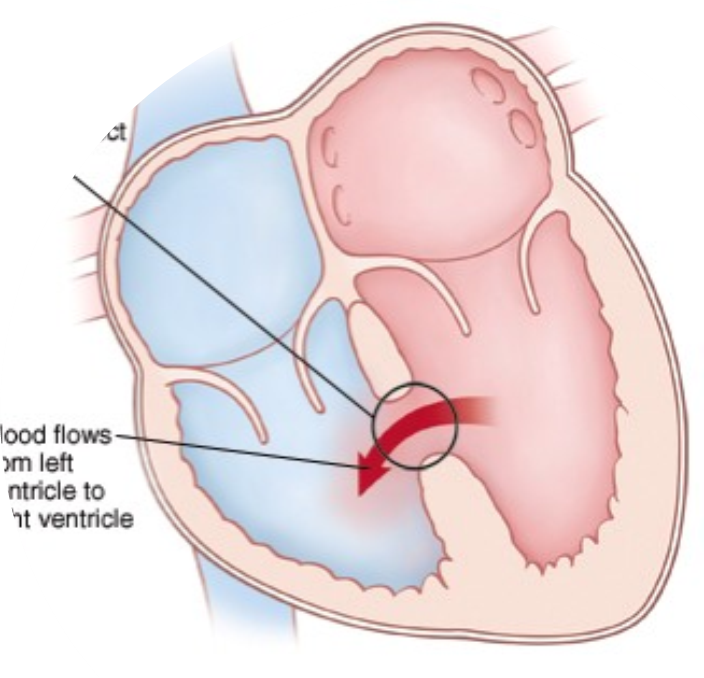
Ventricular Septal Defect (VSD)
Most common congenital heart defect (~30%).
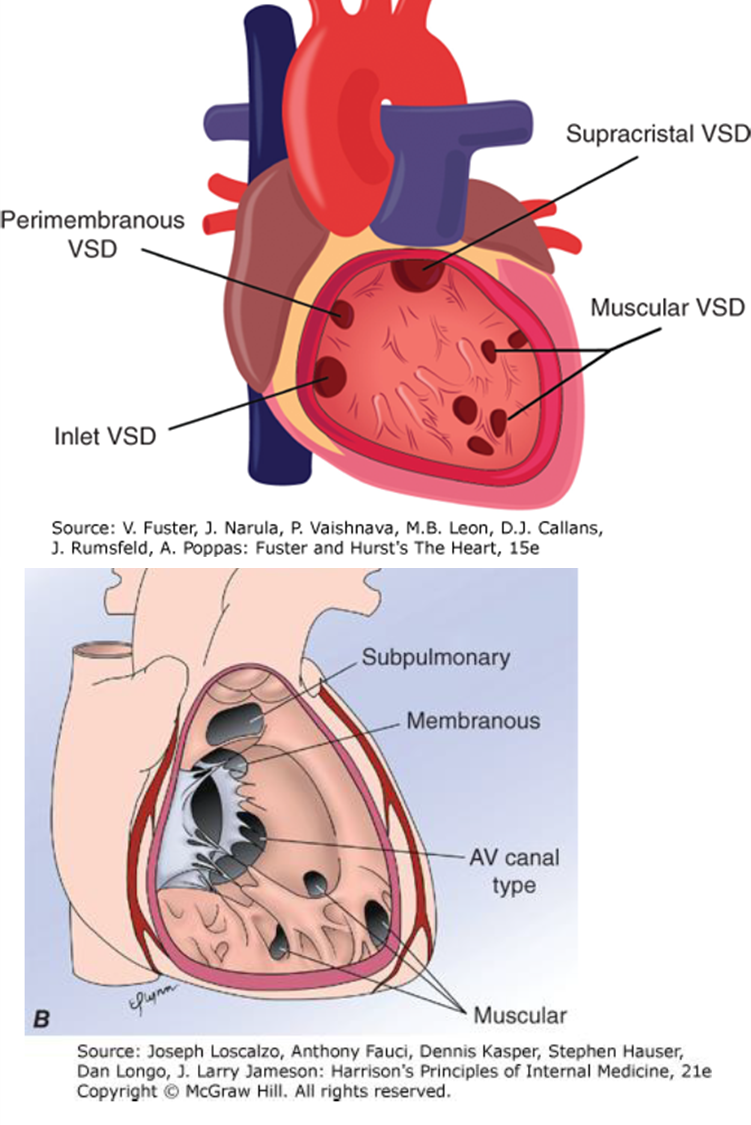
Types of VSDs
Membranous (Perimembranous): Most common (75-80%).
Muscular
Inlet (AV canal type)
Supracristal (Subpulmonary)
Pathophysiology of VSD
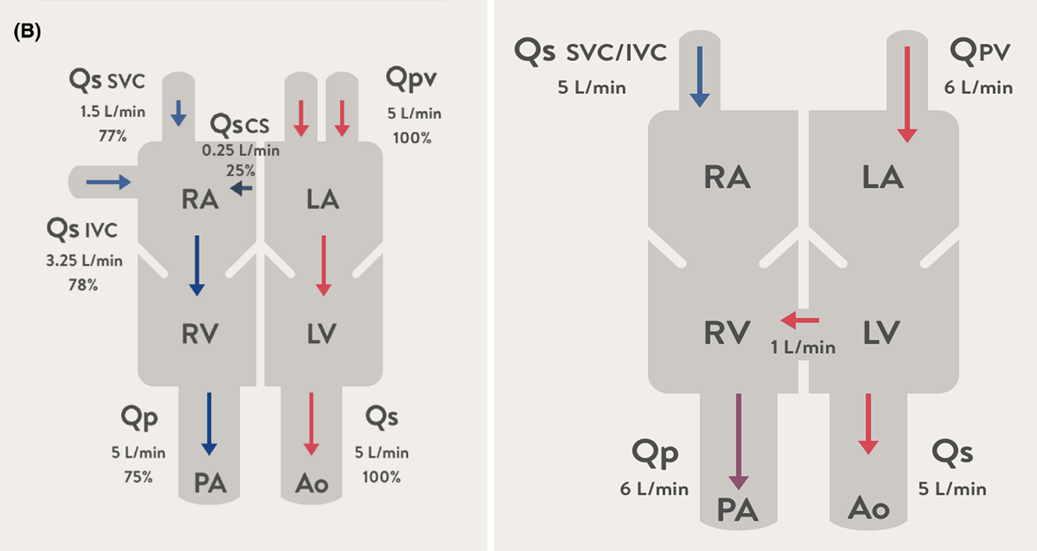
Classified according to defect diameter:
Small: < 3 mm
Medium: 3 - 5 mm
Large: 6 - 10 mm
Restrictive VSDs: Smaller defects, limit blood flow and pressure transmission to the right heart. Pulmonary pressure is normal or minimally elevated. Small VSDs result in a relatively small left-to-right shunt. Heart failure, pulmonary hypertension, and Eisenmenger syndrome do not develop.
Nonrestrictive VSDs: Blood flows easily across larger defects, pressure equalizes between ventricles, large left-to-right shunt. Causes pulmonary artery hypertension, increased pulmonary vascular resistance, right ventricular pressure overload, and right ventricular hypertrophy. Can lead to Eisenmenger syndrome (shunt reversal). Consequences: Over time, untreated nonrestrictive VSDs can result in heart failure, arrhythmias, and decreased exercise tolerance due to the heart's compromised ability to pump effectively.
Moderate VSDs result in intermediate manifestations.
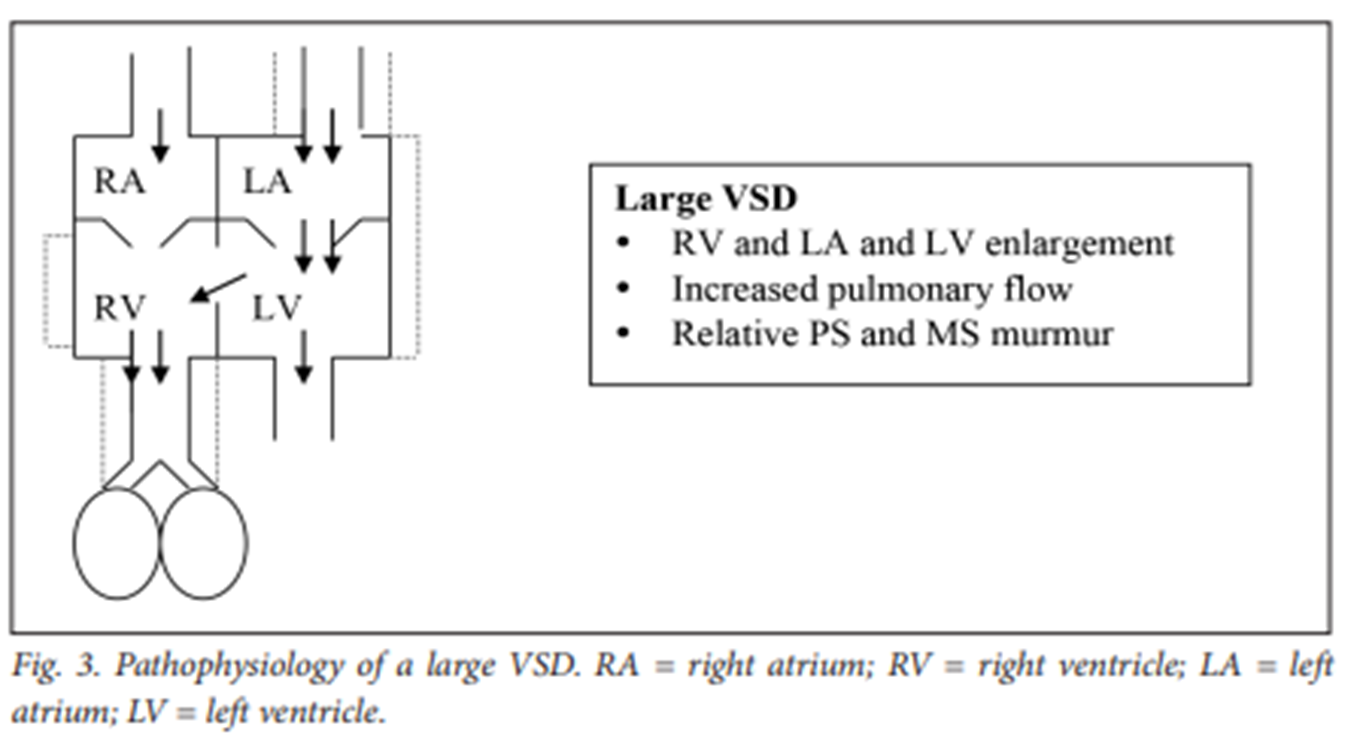
Large VSD Pathophysiology
RA, LA, RV, and LV enlargement.
Increased pulmonary flow.
Relative PS and MS murmur.
Left-to-right shunt reduces LV output. Compensatory mechanisms increase intravascular volume to maintain normal cardiac output, resulting in LV volume overload.
High LV volumes elevate LA filling pressures, causing pulmonary venous congestion.
Forward systemic flow can be reduced, leading to congestive heart failure.
Transmission of LV pressure to the pulmonary vascular bed.
LV and RV become a functionally common chamber.
Pulmonary artery pressure equals aortic pressure in systole.
The combination of volume and pressure contributes to the development of pulmonary vascular disease or Eisenmenger’s syndrome.
Quantifying Shunt Volumes
Qp/Qs ratio: Measured during cardiac catheterization or echocardiography to determine shunt size.
Ratio of total pulmonary blood flow to total systemic blood flow.
Qp/Qs = 1:1: Normal, no shunting.
Qp/Qs > 1:1: Pulmonary flow exceeds systemic flow, net left-to-right shunt.
Qp/Qs < 1:1: Net right-to-left shunt.
Oxygen Saturation
In VSD, oxygen saturation will be higher in the right ventricle due to shunting of oxygenated blood from the left ventricle.
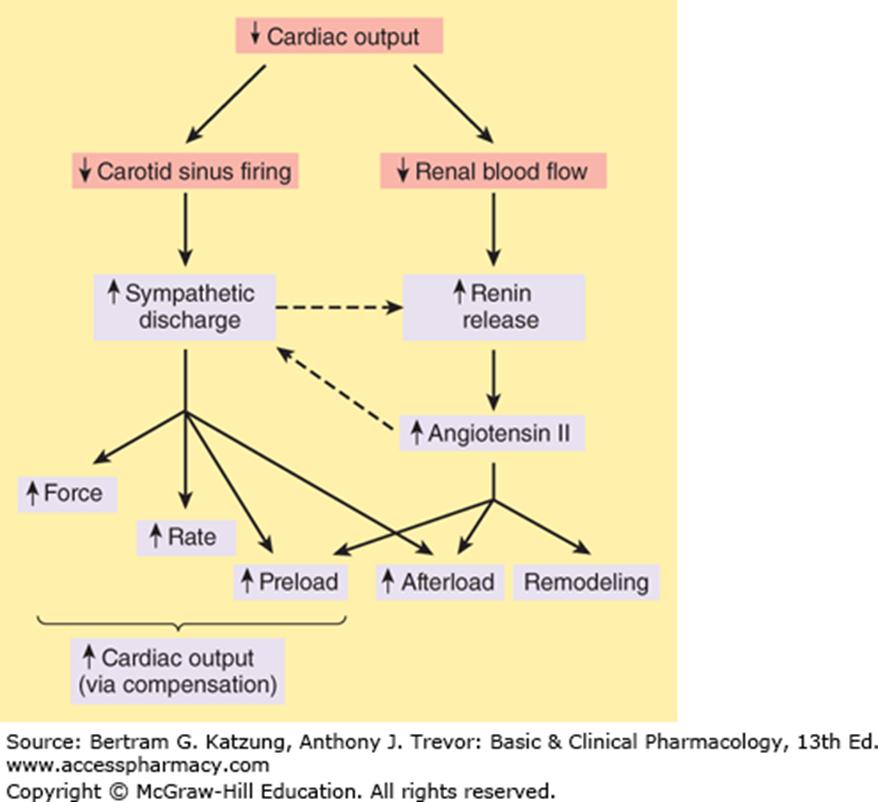
Body's Compensation for Reduced Cardiac Output

Sympathetic discharge, renin release → Angiotensin II
Increase in force and rate, preload, afterload, and remodeling.
Chest Infection and Heart Failure
A patient with acyanotic congenital heart disease (VSD) with congestive heart failure exacerbated by chest infection.
Examination
Crepitations or Crackles
Abnormal respiratory sounds heard with a stethoscope, resulting from air passage through fluid in the respiratory tract.
Associated with hypoxemia, particularly in younger children.
Causes: Emphysema, Bronchitis or pneumonia (the presence of secretions)
, Interstitial lung disease (inflammation or fibrosis).
, Pulmonary edema, Atelectasis
Hepatomegaly
Examination of the abdomen shows hepatomegaly (e.g., 4 cm below right costal margin, liver span of 13 cm).
Heart Sounds
First sound (“lub”): Closure of AV valves at the start of ventricular systole.
Second sound (“dup”): Closure of aortic and pulmonary valves after ventricular systole.
Third sound: Soft, low-pitched sound in early diastole, coinciding with rapid ventricular filling.
Fourth sound: Heard before the first sound when atrial pressure is high or the ventricle is stiff (e.g., ventricular hypertrophy).
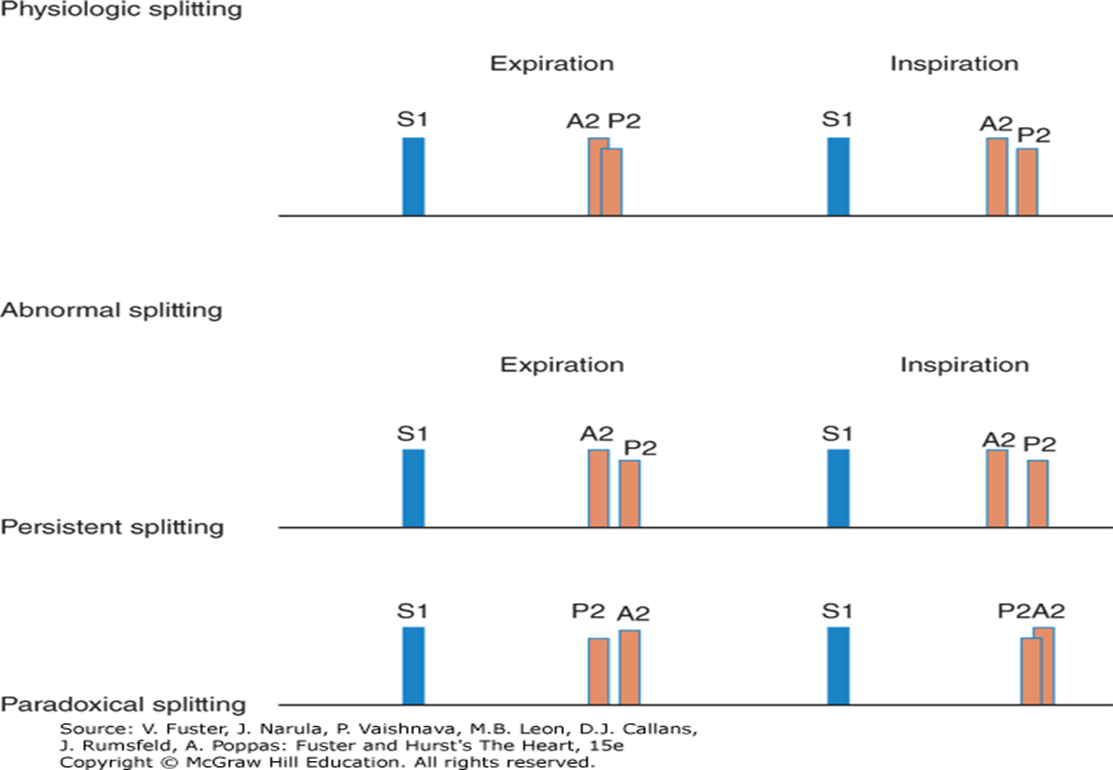
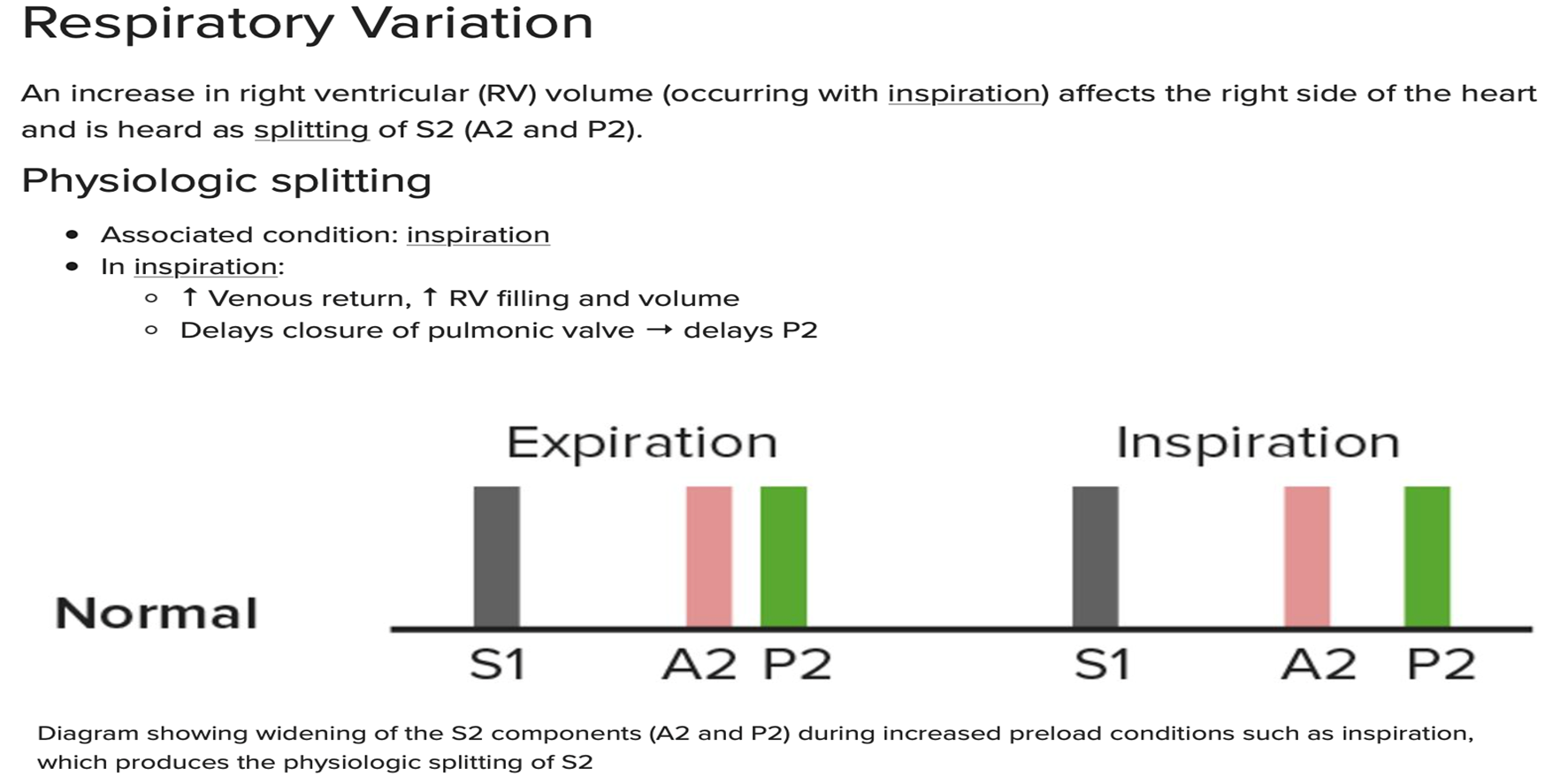
Split of Second Heart Sound During Inspiration
Physiologic splitting: Increase in right ventricular (RV) volume (with inspiration) affects the right side of the heart and is heard as splitting of S2 (A2 and P2).
Inspiration: ↑ Venous return, ↑ RV filling and volume
Delays closure of pulmonic valve, delaying P2
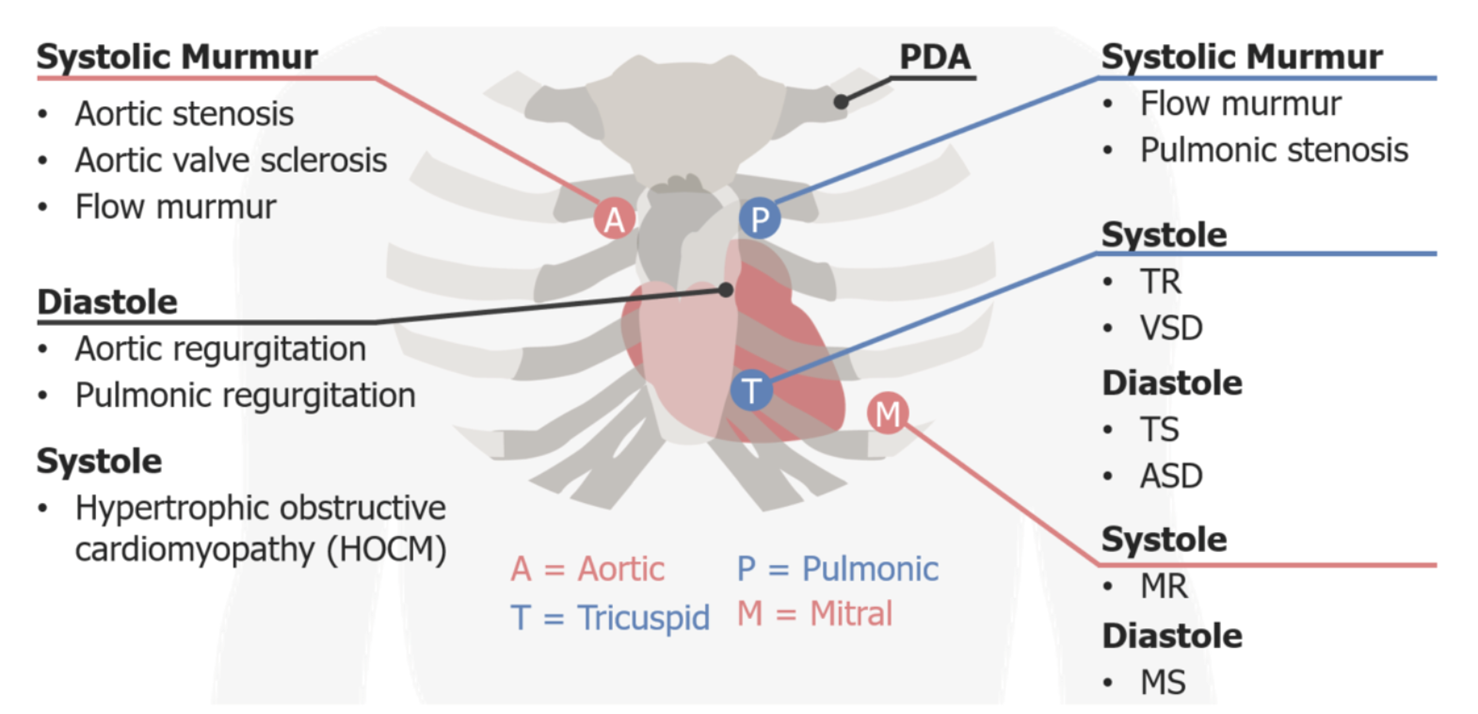
Auscultation Areas
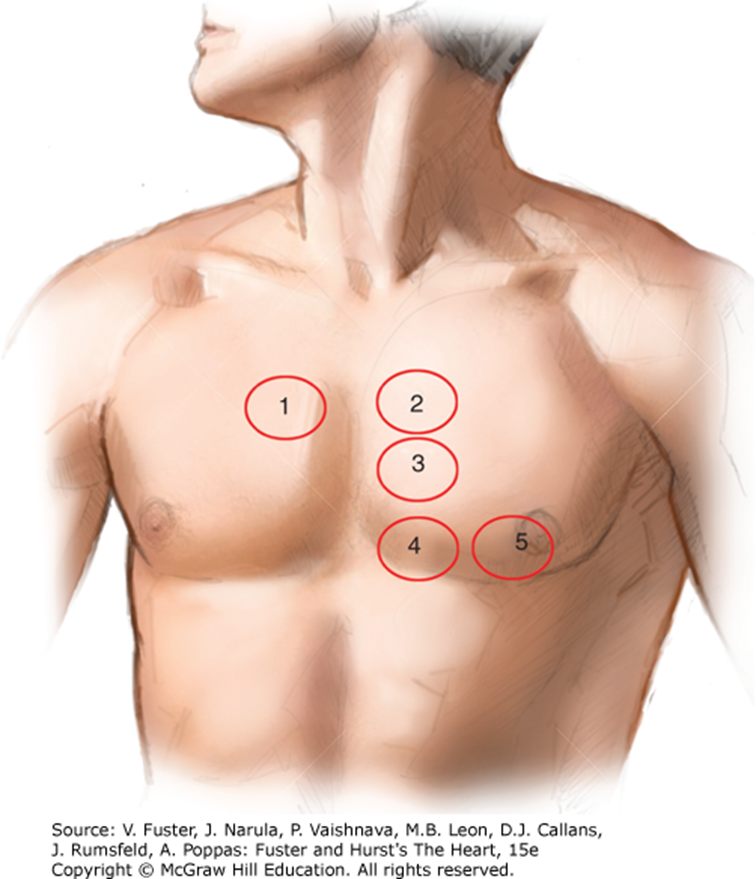
The 5 areas of auscultation can be recalled using the mnemonic, "All People Enjoy Time Magazine."
Aortic area: right 2nd intercostal space adjacent to the sternum
AS
Aortic valve sclerosis
Systolic flow murmurs
Pulmonic area: left 2nd intercostal space adjacent to the sternum
PS
Systolic flow murmurs
Erb's (auscultation) point (left sternal border): left 3rd intercostal space
Systolic murmurs: HOCM
Diastolic murmurs:
AR
Pulmonic regurgitation (PR)
Tricuspid area: left 4th-5th intercostal space adjacent to the sternum
Systolic murmurs:
TR
VSD
Diastolic murmurs:
TS
ASD
Mitral area (apex): left 4th intercostal space, midclavicular line
Systolic murmurs:
MR (holosystolic)
Mitral valve prolapse
Diastolic murmurs: mitral stenosis (MS)
Grading of Murmurs
Grade I: Faint murmur, heard only after a few seconds of auscultation.
Grade II: Moderately loud murmur, heard immediately.
Grade III: Loud murmur, not associated with a thrill.
Grade IV: Loud murmur, associated with a thrill.
Grade V: Very loud murmur, heard with the edge of the stethoscope.
Grade VI: Loudest murmur, heard with the stethoscope removed from the chest.
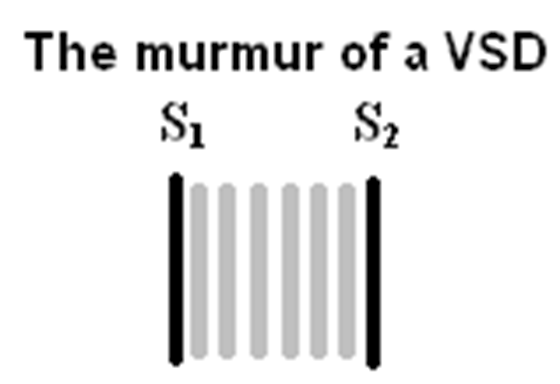
Pansystolic/Holosystolic Murmur
Present in patients with VENTRICULAR SEPTAL DEFECT.
Differential diagnosis of holosystolic murmur at left lower sternal border is tricuspid regurgitation.
TR murmur is exaggerated during inspiration.
TR murmur in neonates can be audible at first day of life while VSD appear once pulmonary vascular resistance drops and left to right shunt will establish.
Pathologic Murmurs
Systolic Ejection:
Semilunar valve stenosis (AS/PS/truncal stenosis)
ASD
Coarctation
Pansystolic:
VSD
AVVR (MR/TR)
Diastolic:
Semilunar valve regurgitation
AI/PI/truncal insufficiency
AV valve stenosis (MS/TS)
Continuous:
Runoff lesions
PDA/AVM/aortopulmonary collaterals
Investigations
CBC
Urea and Electrolytes
Liver Function Test
Blood Gas
Chest X-ray
ECG
ECHO
Arterial Blood Gas
Components: pH, PCO2, PO2, HCO3, SPO2, Lactate
Compensation
When either PCO2 or bicarbonate levels remain altered for a period, the body changes the other variable in the same direction. Compensatory response: pH = 6.1 + log \frac{[bicarbonate]}{0.03 * PCO_2}. To preserve the ratio of bicarbonate to PCO2.
Respiratory compensation: Quick changes in ventilation alter PCO2 in the blood plasma.
Metabolic acidosis: Ventilation increases, PCO2 decreases.
Metabolic alkalosis: Ventilation decreases, PCO2 increases.
Renal compensation: Slower, involves H+ secretion or HCO3− reabsorption.

Chest X-Ray Findings
Cardiomegaly and plethoric lungs (increased pulmonary blood flow from left-to-right shunts).
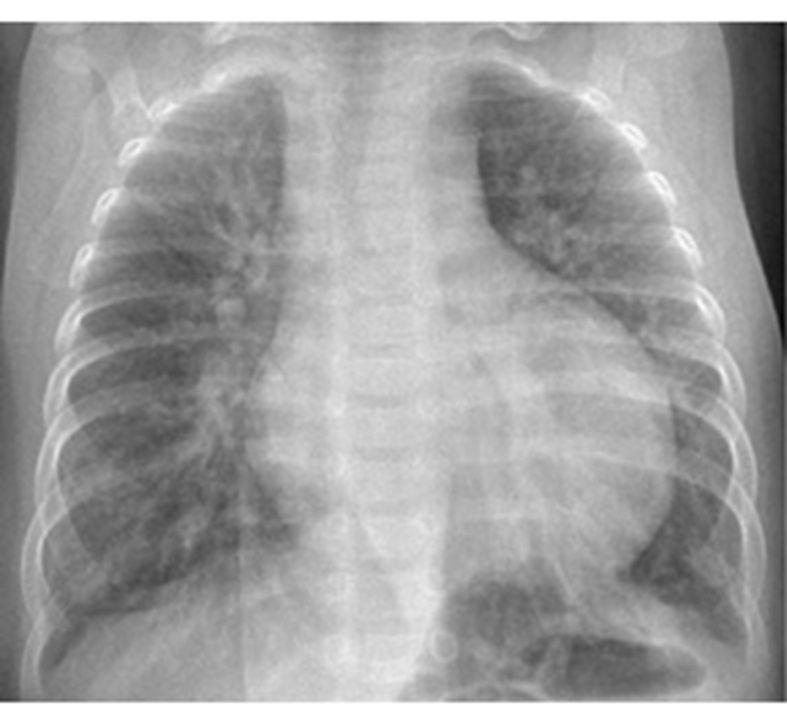
Signs of Plethoric Lungs
Plethoric lung fields are indicative of increased pulmonary blood flow (perfusion) and are seen in left-to-right shunts.
Plain radiograph: prominent pulmonary vasculature.
Dilated and tortuous pulmonary vessels extend further into the peripheral one-thirds of the lungs
Diameter of a pulmonary artery is greater than the accompanying bronchus
Causes of Cardiomegaly
Coronary artery disease
Hypertension
Valvular heart disease
Congenital heart disease
Pulmonary condition (COPD, PHTN)
Myocarditis
Infiltrative (Sarcoidosis, Amyloidosis)
Autoimmune
Arrhythmia
Systemic disease (Anemia, Vitamin B12, Hypothyroidism)
Familial Cardiomyopathy
Toxins/Medication (Chemotherapy , Cocaine , Radiation)
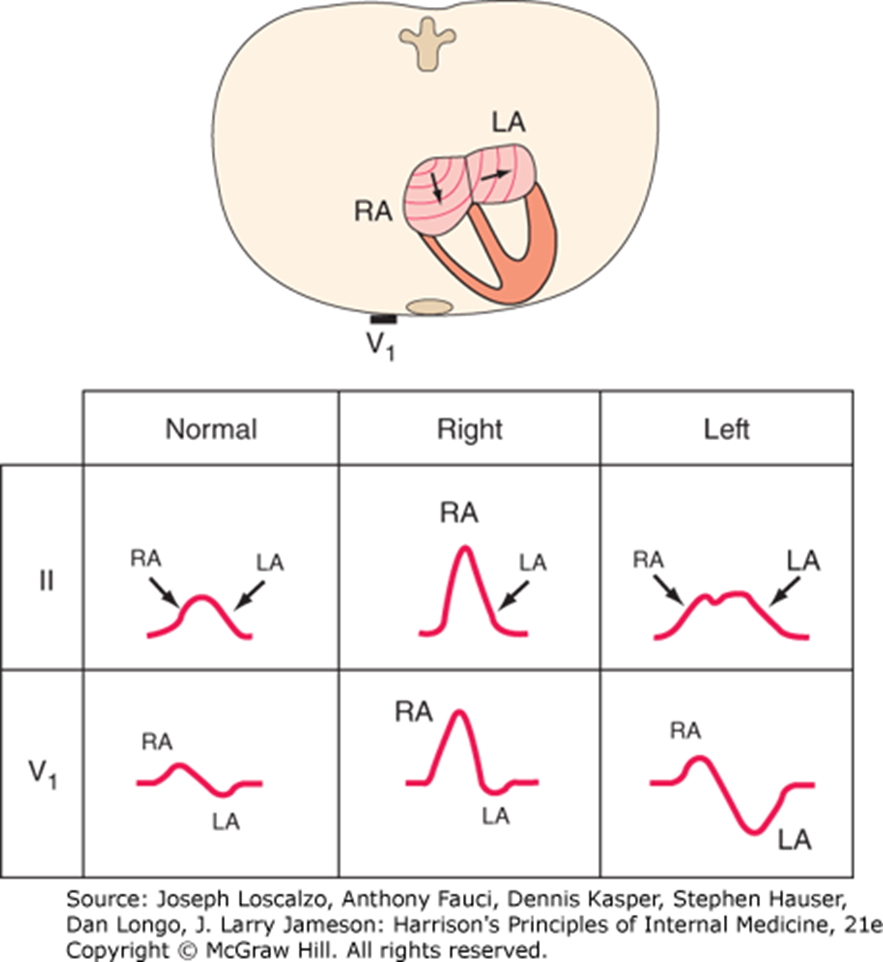
Atrial Hypertrophy
Right Atrial: P-wave amplitude ≥2.5 mm in inferior leads (II, III, aVF).
Left Atrial: Broad, notched P waves in Lead 1 or II. Biphasic P wave with a prominent negative component in Lead V1.
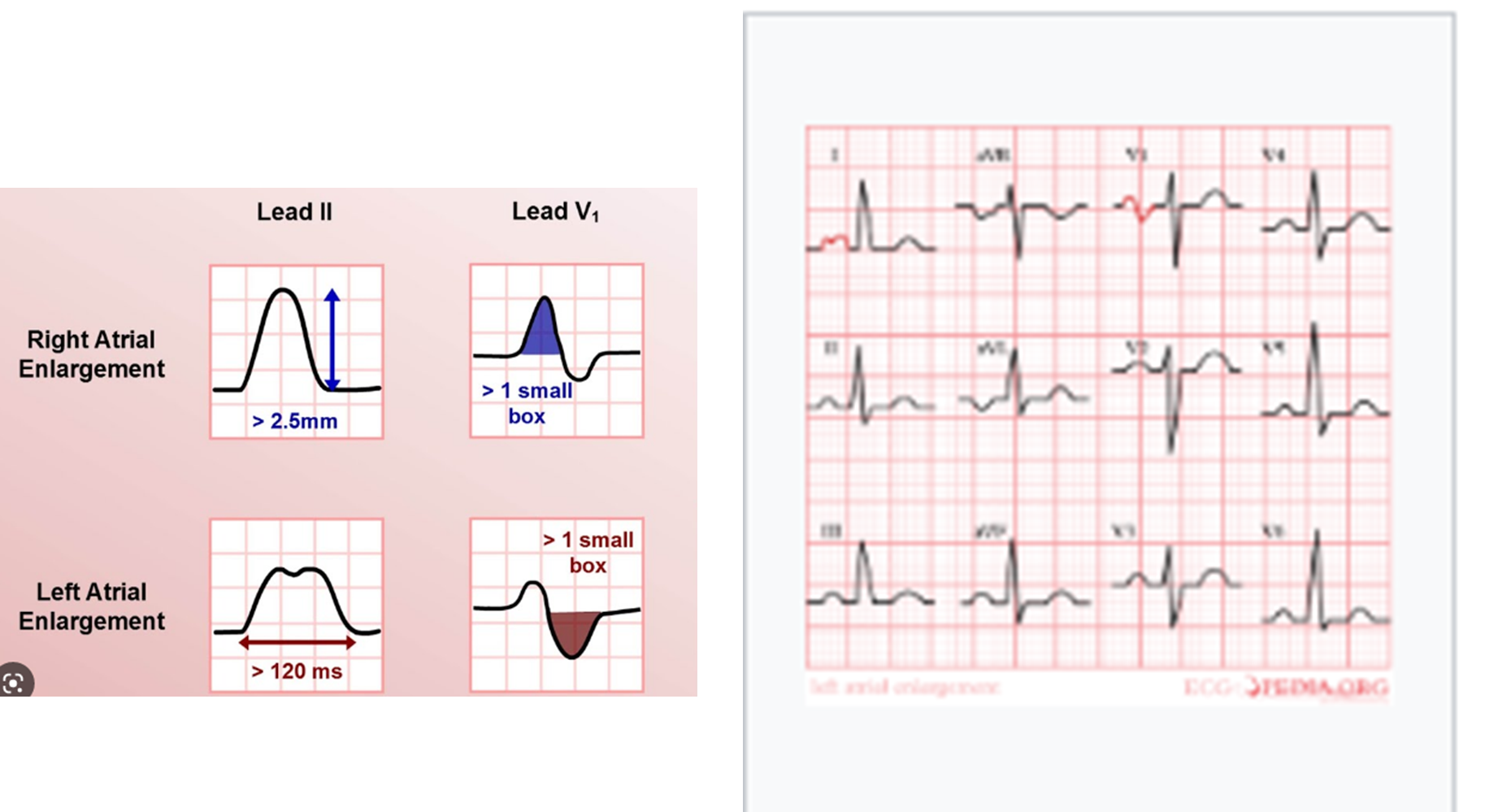
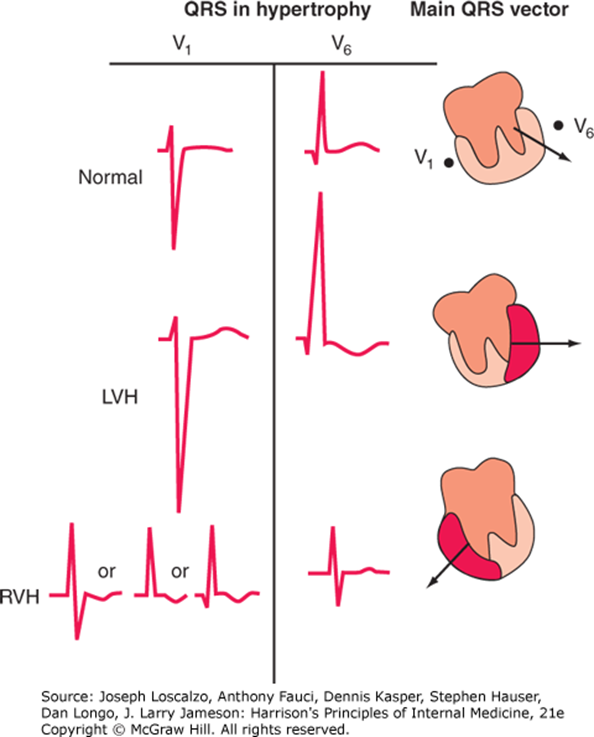
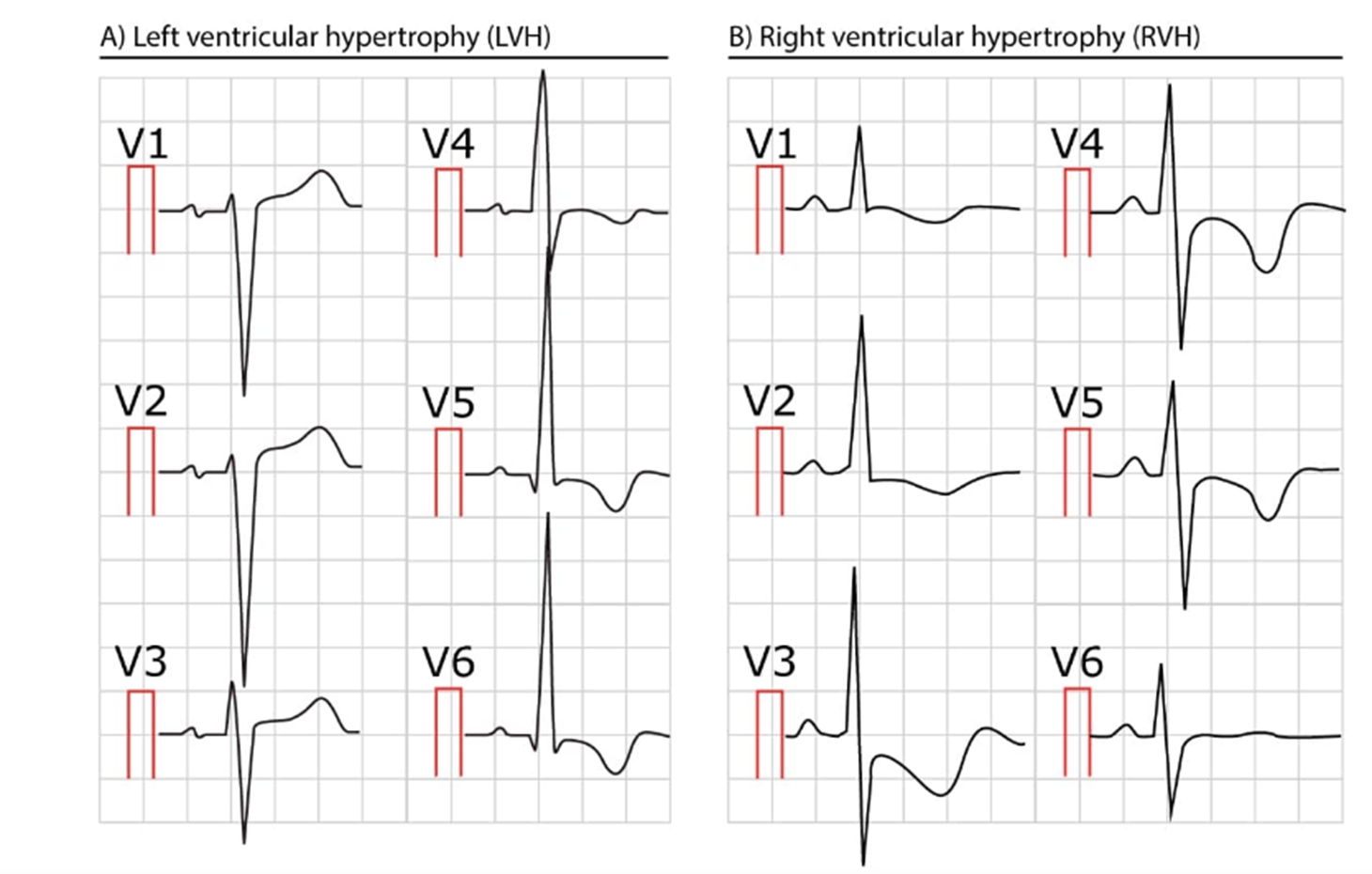
Ventricular Hypertrophy
Left Ventricular: Tall left precordial R waves and deep right precordial S waves. Repolarization abnormalities (ST depression with T-wave inversions) in leads with prominent R waves.
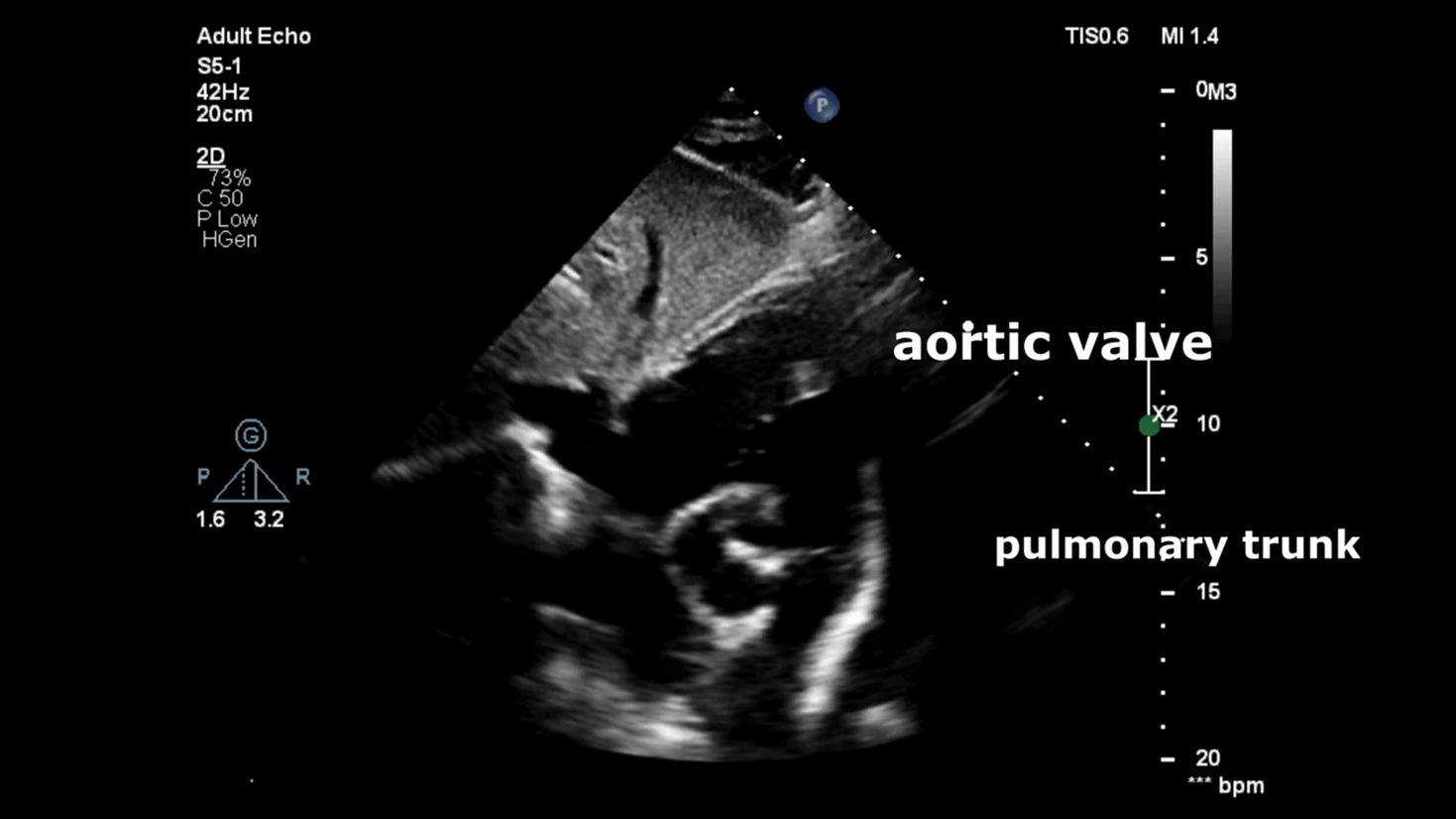
Echocardiography
Echocardiography: 6 mm peri membranous ventricular septal defect with left to right shunting dilated left atrium and left ventricle normal systolic function
Management of VSD
Medical
Surgical:
Temporarily
Permanently
Antibiotics for Bacterial Pneumonia
The patient was commenced on respiratory supportive care and on intravenous antibiotics such as ceftriaxone and azithromycin to target potential bacterial pathogens.
Mechanism of Action of Beta-Lactams
Beta-lactams inhibit bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs), preventing peptidoglycan cross-linking.
Cephalosporins
Anti-MRSA cephalosporins: Effective against MRSA (e.g., ceftaroline).
Fourth generation: Expand on the gram-negative activity of the third generation to provide useful activity against Pseudomonas.
First generation: Good activity against gram-positive bacteria and modest activity against gram-negative microorganisms.
Mechanism of Action of Vancomycin
Vancomycin binds to the D-Ala-D-Ala terminus of peptidoglycan precursors, inhibiting cell wall synthesis.
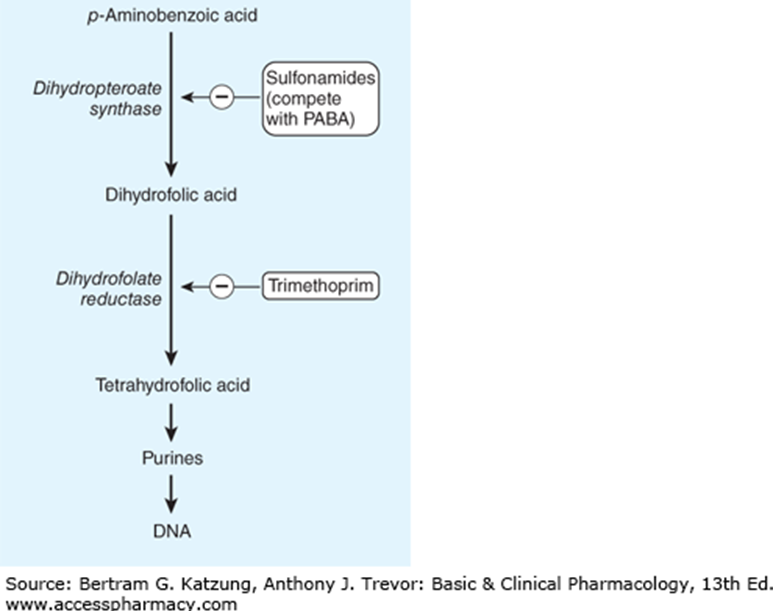
Mechanism of Action of Cotrimoxazole
Cotrimoxazole inhibits folate synthesis by inhibiting dihydrofolate reductase and dihydropteroate synthase.
Mechanism of Action of Azithromycin
Macrolides bind to the 50S ribosomal subunit, inhibiting bacterial protein synthesis.
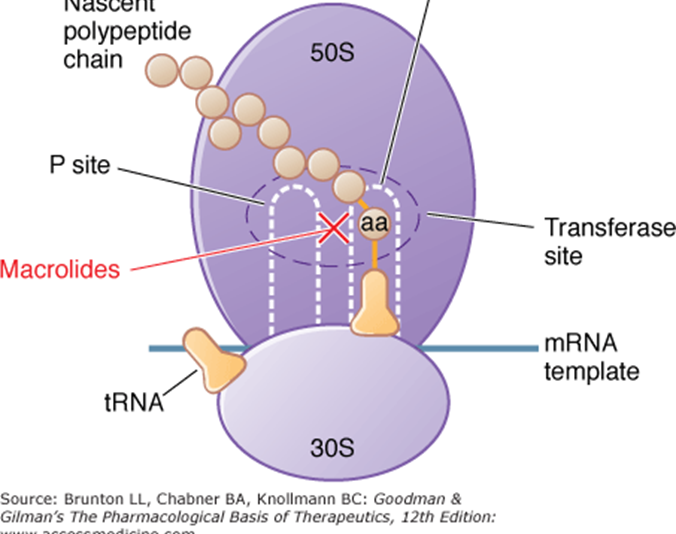
Congestive Heart Failure Treatment
Congestive heart failure in the infant is mainly treated with:
Diuretics
Afterload reduction (Angiotensin converting enzyme inhibitors)
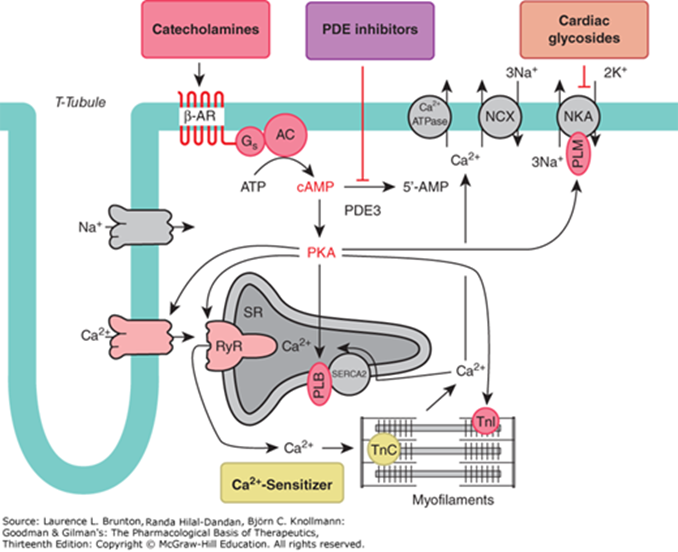
Digoxin
Benefits of Afterload Reduction
Reduction in afterload allows the heart to expel blood against a lower output resistance. This increases stroke volume.
These mechanisms explain why vasodilators (which have no inotropic efficacy and lower blood pressure) increase stroke volume.
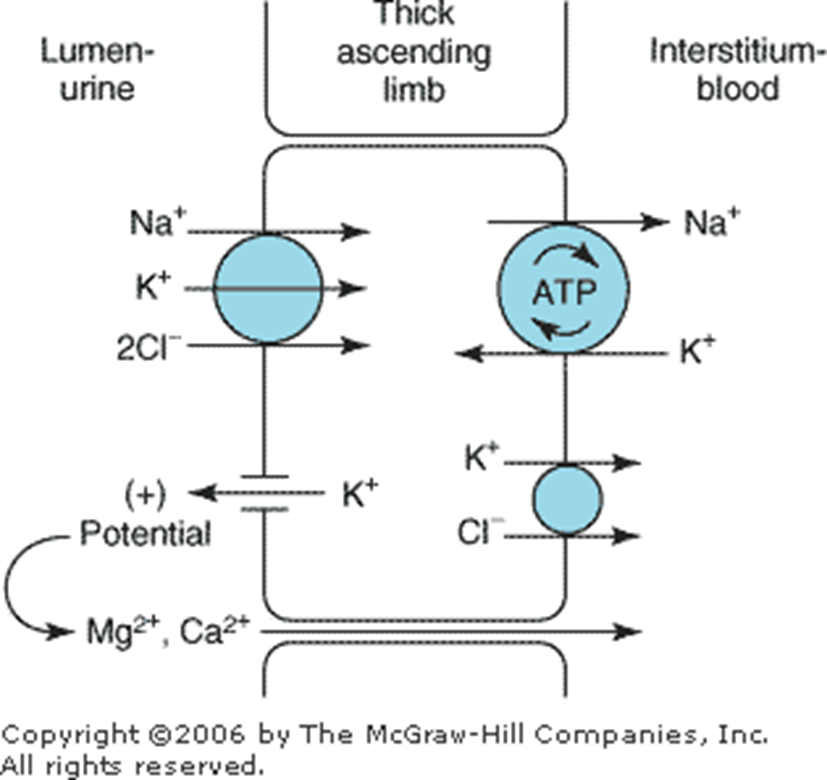
Furosemide
Discharge medications included furosemide. Furosemide is a loop diuretic that inhibits the Na+/K+/2Cl− cotransporter in the thick ascending limb of the loop of Henle.
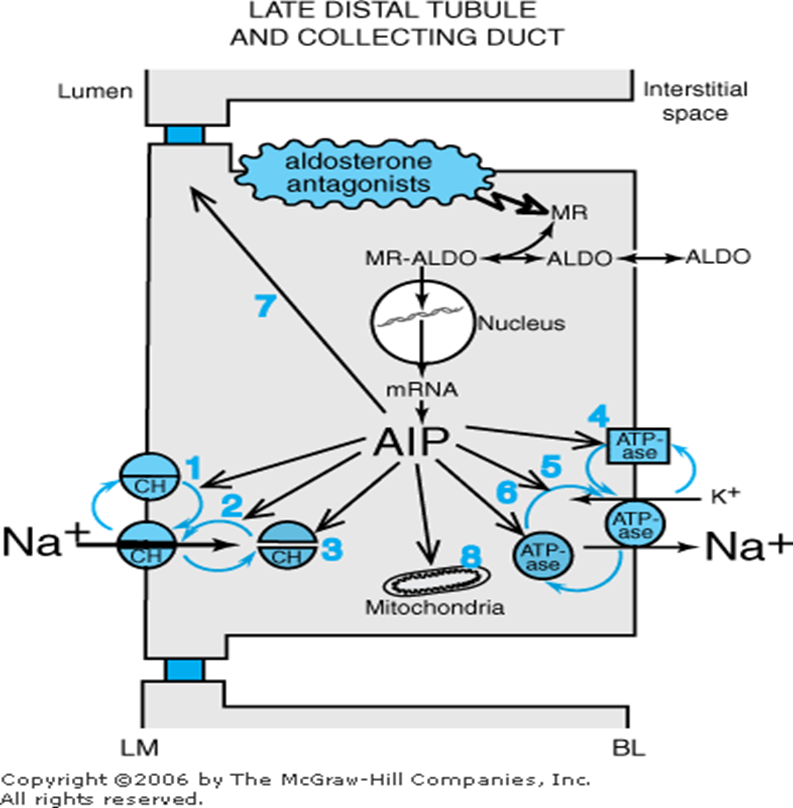
Spironolactone
Discharge medications included spironolactone. Spironolactone is an aldosterone antagonist that blocks aldosterone receptors in the late distal tubule and collecting duct.
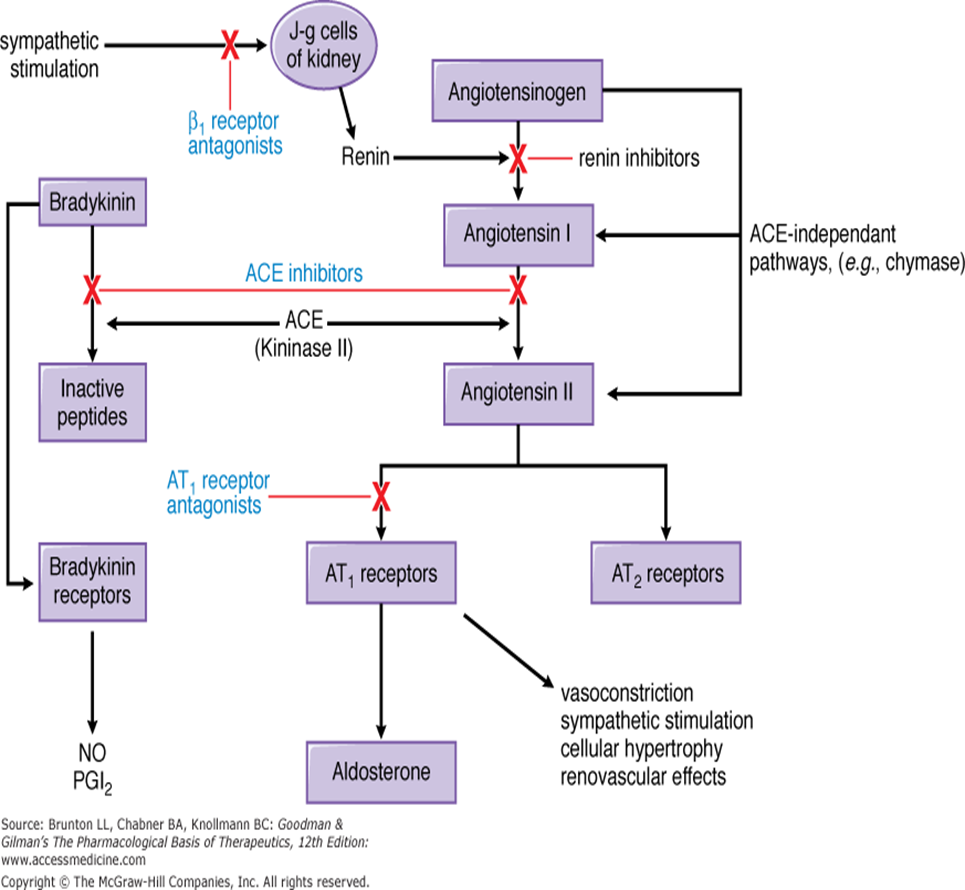
Captopril
Reduces salt and water retention by reducing aldosterone secretion reduce preload.
Reduces peripheral resistance â reduce afterload.
Reduces sympathetic activity
Enhance long-term remodeling of the heart and vessels R eduction in mortality and morbidity.
side effects of captopril?
Cough: A persistent dry cough is a common side effect.
Hyperkalemia: Elevated potassium levels can occur, requiring monitoring.
Angioedema: Swelling of the face, lips, or throat may take place, which can be serious.
Hypotension: A significant drop in blood pressure can occur, especially after the first dose.
Bradycardia: A slower than normal heart rate may be observed, necessitating further evaluation.
Digoxin Side Effects
Altered color perception
Nausea, vomiting and abdominal pain
Arrhythmias
Headache, fatigue, and sleeplessness
Gynecomastia
Oxygen Therapy
Hypoxemia
Hypoxaemia: Low levels of oxygen in the blood.
Hypoxia: Inadequate oxygen in tissues for normal cell and organ function.
SaO2: Arterial oxygen saturation measured by gas analysis.
SpO2: Arterial oxygen saturation measured by pulse oximetry.
Newborn infants have lower oxygen saturation immediately after delivery.
Causes of Hypoxemia
Upper airway obstruction
Lower respiratory tract infection (severe pneumonia or bronchiolitis)
Severe asthma
Common neonatal conditions like birth asphyxia and in respiratory distress syndrome
Severe sepsis
Heart failure or cardiac arrest
Anemia
Trauma
Carbon monoxide poisoning
Oxygen Delivery Devices
Non-Invasive:
Low flow Nasal cannula
High flow Nasal cannula
Face mask
Contanous positive airway pressure (CPAP)
Bi-PAP
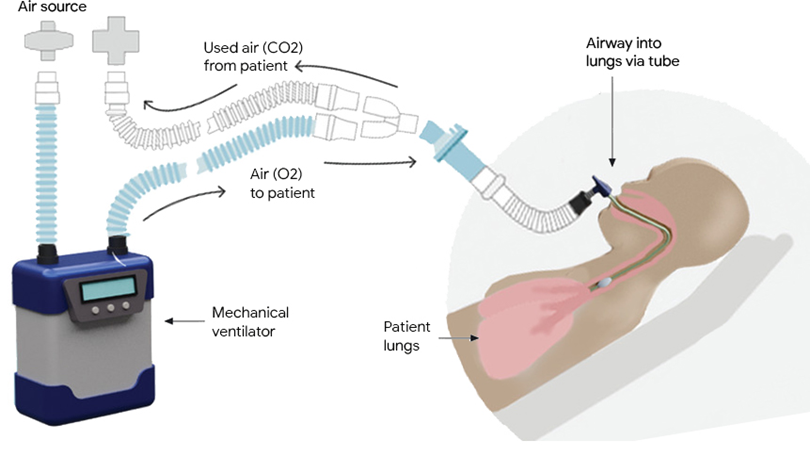
Invasive:
Intubation and mechanical ventilation
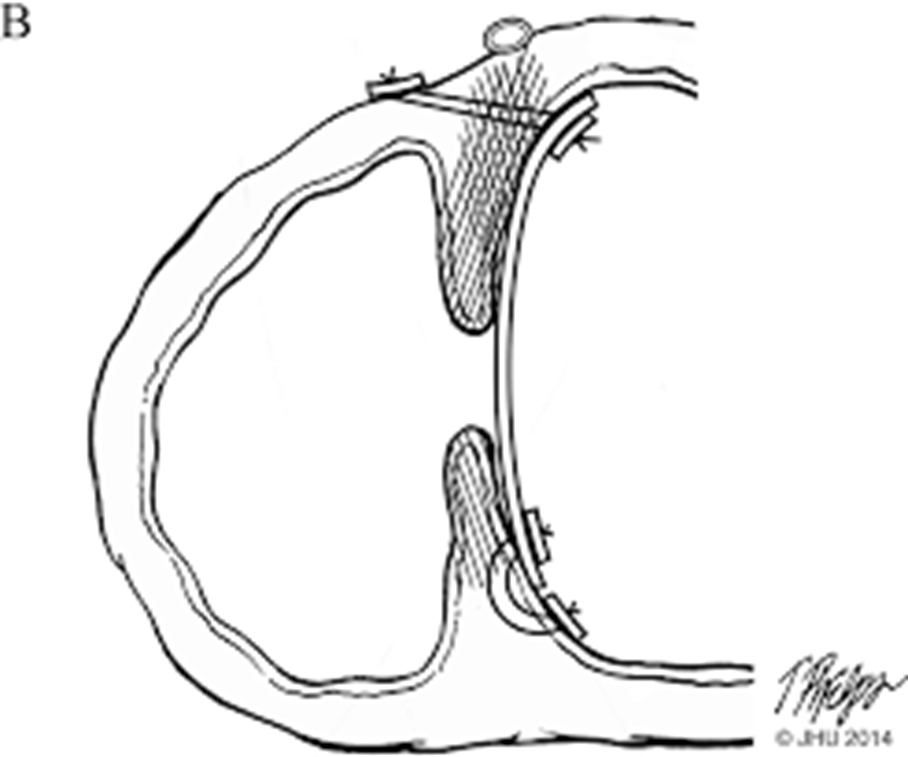
Surgical Interventions
Temporarily: Pulmonary artery banding
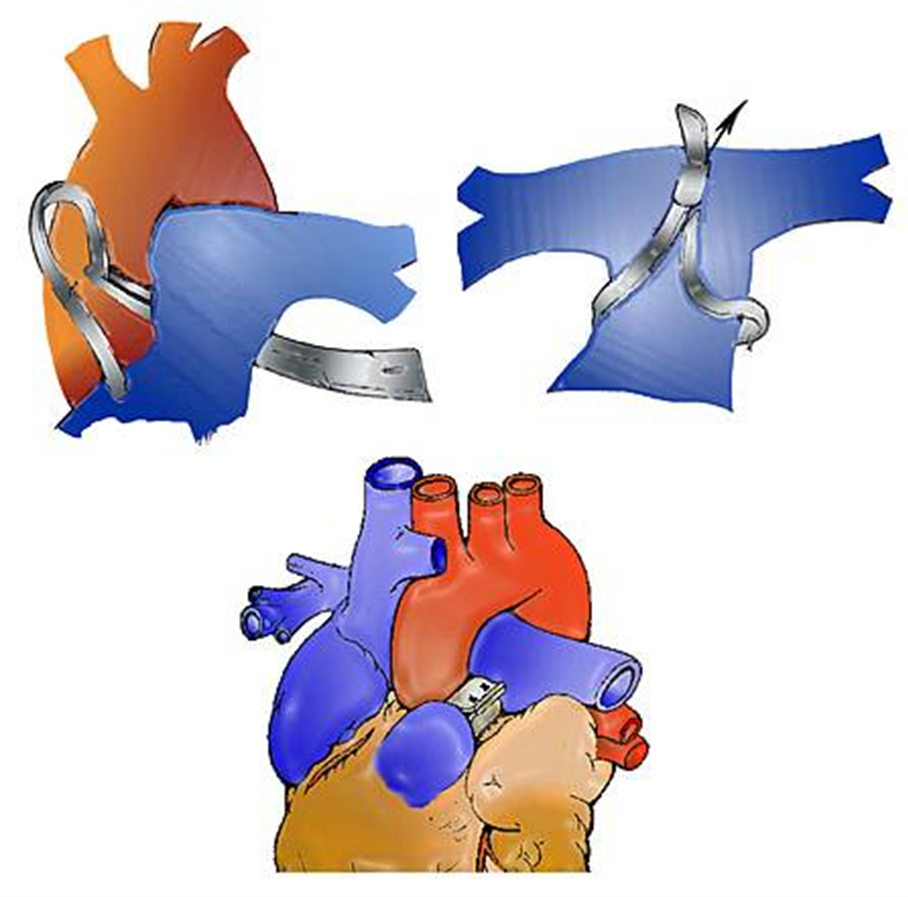
Permanently:
Device closure (VSD)
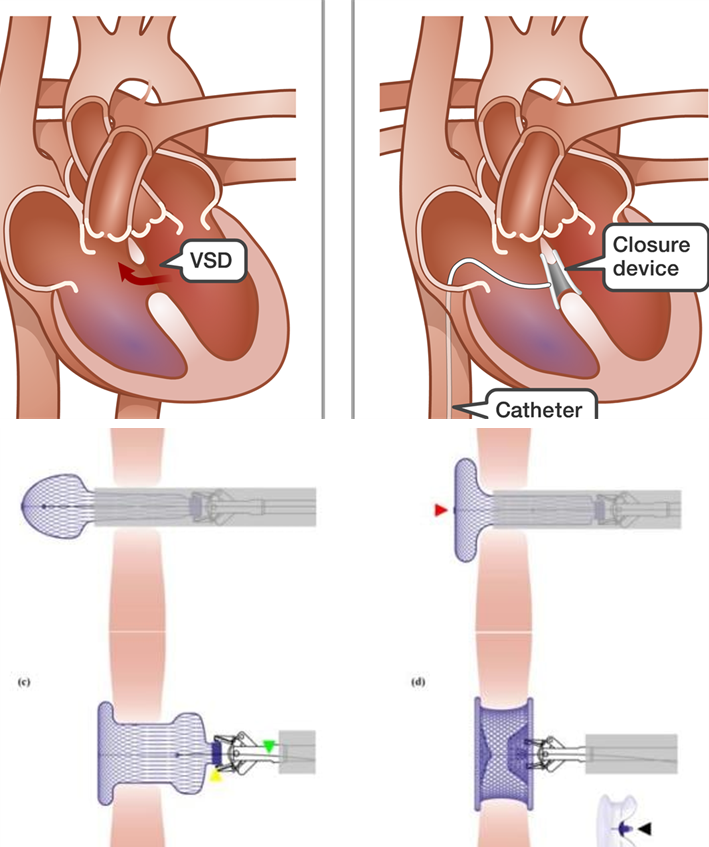
Surgical repair